Abstract
The study was conducted to evaluate the efficacy of different forms of trivalent chromium (Cr) supplementation on tissue chromium deposition in finishing pigs. A total of 96 pigs with an initial average body mass 65.57 ± 1.05 kg were blocked by body mass and randomly assigned to four treatments with three replicates. Pigs were offered one of four diets including a control diet or the control diet supplemented with 200 μg/kg chromium from either chromium chloride (CrCl3), chromium picolinate (CrPic) or chromium nanocomposite (CrNano) for 40 days. During the trial, all pigs were given free access to feed and water. After feeding trial, eight pigs from each treatment were slaughtered for samples collection. The results showed that supplemental CrNano increased Cr content in blood, longissimus muscle, heart, liver, kidney, jejunum, and ileum (P < 0.05). Supplemental Cr from three sources increased Cr excretion from all feces (P < 0.05). Urinary Cr excretion was increased by CrNano or CrPic supplementation significantly. These results suggested that chromium nanocomposite exhibited more effective on tissue Cr deposition in pigs, which indicated higher absorption compared with CrCl3 and CrPic.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Trivalent chromium [Cr (III)] was proposed to be an essential trace element over 50 years ago and has been accepted as an essential element for over 30 years [1]. There have been a number of studies on the effect of Cr (III) supplementation on growth performance, carcass characteristics, pork quality, reproduction and tissue deposition in domestic animals [2–5]. However, Di Bona et al. [6] reported Cr (III) appeared not to be an essential element, and its potential effects on domestic animals might be considered pharmacological.
The factors controlling intestinal absorption of particles are now better known. Size, nature of the polymer, zeta potential, and vehicle have been determined as critical factors influencing particles uptake [7]. It is postulated that the absorption and utilization of Cr is dependent on its status in the gastrointestinal tract. Nanocomposite, which possesses new electrical, magnetic, mechanical and biological properties associated with its reduced dimension and the high surface area, was investigated in various biomedical applications [8] and was shown to exhibit a high rate of absorption in the gastrointestinal tract [9, 10]. In previous work at our laboratory, chromium nanocomposite (CrNano) was shown to produce beneficial effects on carcass characteristics, pork quality and individual skeletal muscle weight compared to the control group, which implicated higher bioavailability [11]. Further studies in rats showed that dietary supplementation of Cr as CrNano exhibited considerably higher absorption efficiency than both chromium picolinate (CrPic) and chromium chloride (CrCl3) [12]. The objective of the present investigation was to assess the efficacy of three different sources of chromium as CrCl3, CrPic, and CrNano on tissue Cr deposition in finishing pigs.
Materials and Methods
Animals and Experimental Design
The protocol of this study was approved by the Institution Animal Care and Use Committee at Zhejiang University and was conducted in accordance with the National Institutes of Health guidelines for the care and use of experimental animals. The feeding trial was carried out in Anji Zhengxin Breeding Farm. A total of 96 crossbred pigs (Duroc × Landrace × Yorkshire) with an average body mass of 65.57 kg (SD = 1.05) were blocked by initial mass and equalized for sex and ancestry, randomly allotted to one of the following dietary treatments: control and control diet supplemented with 200 μg/kg chromium from either CrCl3, CrPic, or CrNano, with three replicate pens per treatment and eight pigs per pen. The CrCl3 was purchased from Sangon Co. Ltd. The CrPic was provided by Zhejiang Huangyan Kanda Animal Health Co., Ltd., China. CrNano (Nanocomposite of CrCl3, size ranges from 40 to 70 nm) was provided by the Key Laboratory of Molecular Animal Nutrition, Ministry of Education, China. All experimental treatments used the same corn–soybean meal basal diet formulated met or exceeded NRC (1998) recommendations for nutrients except digestible energy (Table 1).
The pigs were penned in 3.25 × 5.25-m, with a nipple drinker and feeder to allow pigs ad libitum access to feed and water. The duration of the feeding trial was 40 days. Preceding the study, pigs were allowed a 7-day adaptation period, during which they were offered basal diet for ad libitum consumption.
Samples Collection
At the end of the feeding trial, after 1 h of feed removal, eight pigs from each treatment were selected and slaughtered by exsanguination after electrical stunning. Blood samples were collected at slaughter, kept in heparinized tubes and stored at −20°C. The heart, liver, kidney spleen, pancreas, lung and intestines were harvested with excess fat and veins carefully stayed away. The samples of hair, longissimus muscle, and fat in the tenth rib, heart, liver, kidney spleen, pancreas, lung, and intestines (duodenum, jejunum, ileum, cecum, colon) were collected from the same position of the organs, packed and stored at −20°C. Feces from the end of rectum and urine from bladders were also collected and stored at −20°C
Assay of Chromium Residue
Tissue Cr content in blood, hair, faces, urine, longissimus muscle, fat, sampled organs, and intestines were determined by the method of Anderson et al. [13] with atomic absorption spectrometry (AA6510, Shimadzu, Japan).
Statistical Analysis
Data are presented as mean value. The general linear model procedure of SAS software [14] was used to determine treatment effects using one way analysis of variance. For all data, the model included treatment as main effect. A probability of P < 0.05 was considered significant.
Results
Cr Contents in Muscle and Fat
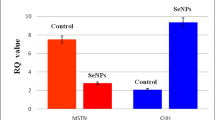
Cr content in longissimus muscle was increased by 119.06% (P < 0.05) with the supplementation of CrNano, and was 89.45% (P < 0.05) higher than that in CrCl3 added group (Fig. 1). There was no significant difference of Cr content in fat from the tenth rib with the dietary Cr supplementation from CrCl3, CrPic, or CrNano.
Cr Contents in Selected Organs
Supplemental Cr from CrNano increased Cr content in heart, liver, kidney by 87.10% (P < 0.05), 114.60% (P < 0.05), 94.76% (P < 0.05; Table 2), and supplemental Cr from CrPic also increased Cr content in heart, liver, and kidney by 58.41% (P < 0.05), 86.45% (P < 0.05), 67.55% (P < 0.05), respectively (Table 2). No significant difference was found in Cr content in spleen, lung, and pancreas of pigs fed Cr from different sources.
Cr Contents in Intestinal Tissues
Table 3 showed that Cr incorporation into the jejunum was the greatest, followed by ileum and colon, rectum was the lowest. Supplemental Cr from CrNano increased Cr content in jejunum and ileum by 68.95% (P < 0.05), 61.27% (P < 0.05). CrPic also increased Cr content in jejunum and ileum by 54.43% (P < 0.05), 58.06% (P < 0.05), respectively.
Cr Contents in Blood, Hair, Feces, and Urine
Table 4 showed that supplemental Cr from CrNano and CrPic increased blood Cr by 134.83% (P < 0.05), 86.06% (P < 0.05) respectively. Feces Cr was significantly increased with the supplementation of Cr, which was increased by 43.59% (P < 0.05), 30.22% (P < 0.05), 29.15% (P < 0.05) in CrNano, CrPic and CrCl3 added group respectively. Urine Cr was also increased by 37.62% (P < 0.05), 36.54% (P < 0.05) with the supplementation of CrNano or CrPic. No significant difference was found in hair Cr content.
Relative Mass of Selected Organs
Table 5 showed that there were no significant differences in relative mass of heart, liver, spleen, kidney, and pancreas of pigs fed supplemental Cr from CrCl3, CrPiC, or CrNano.
Discussion
It is well known that different chemical forms of Cr (III) have diverse rates of absorption. Inorganic Cr (III) (e.g. chromium chloride, CrCl3) is very poorly absorbed (0.5–2%) [15]. Organic Cr (III) is supposed to have greater biological availability than inorganic Cr (III) [16]. However, some studies have shown that the absorption rate of CrPic is similar to CrCl3 and Cr nicotonate [17, 18], and the recent study of comparative absorption even indicated that CrCl3 is possibly absorbed better [19]. Some Cr supplements have been shown to have much higher absorption (40–60 or higher) [20]. Therefore, it is of great significance to develop novel forms of Cr (III) with enhanced intestinal absorption efficiency and high oral bioavailability.
Nanotechnology, a multidisciplinary scientific undertaking, involves creation and utilization of materials, devices or systems on the nanometer scale (range in size from 1 to 100 nm). Nanoparticle has already been investigated as drug carrier system for gastrointestinal (GI) delivery of therapeutic agents [21–23] to enhance drug absorption, improve bioavailability [24], and target therapeutic agents to particular organs [25, 26]. It has been shown that nanoparticle was able to exhibit high rate of uptake in the GI tract [10]. CrNano, a novel form of Cr (III) constructed previously based on the nanotechnology, was shown to greatly increase tissue chromium retention in selected muscles and organs when supplemented to the diets of pigs and rats, and it was postulated that CrNano could improve the absorption of Cr (III) in the GI tract [11, 12]. The transportation and absorption of CrNano via Caco-2 cell monolayers in comparison to CrPic and chromium chloride (CrCl3) was investigated, and it was found that epithelial transport of CrNano, CrPic and CrCl3 across the Caco-2 cell monolayers mainly via passive transport pathways, and CrNano exhibited considerably higher absorption efficiency than both CrPic and CrCl3 in Caco-2 cell monolayers [27].
Supplemental Cr (III) from CrNano greatly elevated muscle Cr content in the current study, which is consistent with previous study in pigs [11]. Normally Cr content in muscle is relatively stable, and was found increased only when fed very high dosage in some animals. Liu et al. [28] demonstrated higher Cr content in breast muscle of laying hens, when fed 5000 or 50,000 μg/kg Cr from CrCl3. In the current study, pigs were fed the same dosage (200 μg/kg) Cr supplementation from different Cr source, only CrNano supplementation group exhibited higher muscle Cr content, which might due its higher absorption rate [27].
In the present study, Cr concentrations in liver and kidney were greatly increased with the supplementation of CrNano in the diet, which is consistent with previous result in pigs and rats [11, 12]. Significant increases of Cr concentration in kidney and liver were also found in CrPic supplemented group, which is in agreement with Anderson et al. [18], who measured the incorporation of Cr in the tissues of rats fed nine different forms of Cr, and reported Cr incorporation into the kidney was greatest for Cr dinicotinic acid diglycine cysteine glutamic acid (850 ng/g dry weight) followed by Cr potassium sulfate (407 ng/g), Cr acetate (397 ng/g), Cr dinicotinic acid dihistidine (394 ng/g), CrPic (368 ng/g), Cr glycine (343 ng/g), CrNic (166 ng/g), CrCl3 (74 ng/g), Cr trihistidine (49 ng/g) and the control (23 ng/g). Their results also showed that increases in tissue Cr following Cr supplementation were greatest for the kidney, followed by the liver, with smaller changes in the heart, which is in line with our current results. The Cr concentrations in spleen and pancreas exhibited a climbing trend with the supplementation of Cr, though no statistically significant difference was found. Among the supplemental Cr-treated groups, Cr residue level in the selected organs was highest in CrNano, followed by CrPic, then CrCl3, which implicated their differences in bioavailability.
The effect of Cr supplementation on Cr deposition in small intestine (duodenum, jejunum, ileum) and large intestine (cecum, colon, rectum) suggested that supplemental Cr only resulted in Cr incorporation in jejunum and ileum, which indicated that jejunum and ileum was the main absorption site in pigs. Research in rats also confirmed that jejunum was the main absorption site of Cr [29].
The effect of supplemental Cr on Cr content in blood, feces, and urine suggested that only part of the supplemental Cr was absorbed. Absorbed Cr is excreted mainly in the urine [30], which might be the reason for the highest Cr residue in the kidney. Higher Cr in blood and urine and lower Cr in feces of pigs fed CrNano indicated that CrNano exhibited higher absorption rate. The clear rate of the high bioavailable Cr as CrNano in the body and long-term safety evaluation remain to be further studied.
In the current study, there were no significant differences in relative mass of heart, liver, spleen, kidney and pancreas of pigs fed supplemental Cr from different sources. Effects of supplemental Cr on organs mass varies in different researches. Page et al. [2] reported that CrPic exhibited no effect on heart mass in pigs. However, CrPic supplementation resulted increased heart mass in lamb [31] and rats [32]. Boleman et al. [33] reported that mass of liver and kidney were decreased with the supplementation in pigs.
There are many concerns over the use of the most popular Cr dietary supplement, CrPic. Isolated incidents of deleterious of CrPic supplementation have been reported [34, 35]. Sterns et al. [36] reported the complex generated chromosome damage in a Chinese hamster ovary (CHO) cell model. CrPic at concentrations range from 50 μM to 1.0 mM generated three- to 18-fold more chromosome damage than found in controls. CrNic and CrCl3 did not produce damage at equivalent nontoxic doses. In contrast, Anderson et al. [37] fed rats diets containing up to 100 mg Cr as CrPic/kg of diet for 24 weeks, no acute toxic effects were observed. The results of the effects of CrNano on tissue Cr reported here are with an observation period of only 40 days, which is a relatively short-term study. The accumulation of Cr in the tissue could be a concern if pigs are fed dietary CrNano for a long period, which could be lead to toxic. The effects of long-term use of CrNano, positive or negative remain to be examined further. The effects of long-term use of CrNano, positive or negative remain to be examined further.
Conclusions
The results of the present study suggested that chromium nanocomposite were exhibited to be more effective on tissue Cr deposition in pigs, which indicated higher absorption compared with CrCl3 and CrPic.
References
Chen Y, Watson HM, Gao JJ, Sinha SH, Cassady CJ, Vincent JB (2011) Characterization of the organic component of low-molecular-weight chromium-binding substance and its binding of chromium. J Nutr 141:1225–1232
Page TG, Southern LL, Ward TL, Thompson DL Jr (1993) Effect of chromium on growth and serum and carcass traits of growing-fishing pigs. J Anim Sci 71:656–662
Lindermann MD, Wood CM, Harper AF, Kornegay ET, Anderson RA (1995) Dietary chromium picolinate additions improve gain: feed and carcass characteristics in growing–finishing pigs and increase litter size in reproducing sows. J Anim Sci 73:457–465
Mooney KW, Cromwell GL (1997) Efficacy of chromium picolinate and chromium chloride as potential carcass modifiers in swine. J Anim Sci 75:2661–2671
Shelton JL, Payne RL, Johnston SL, Bidner TD, Southern LL, Odgaard RL, Page TG (2003) Effect of chromium propionate on growth, carcass traits, pork quality, and plasma metabolites in growing-finishing pigs. J Anim Sci 81:2515–2524
Di Bona KR, Love S, Rhodes NR, McAdory D, Sinha SH, Kern N, Kent J, Strickland J, Wilson A, Beaird J, Ramage J, Rasco JF, Vincent JB (2011) Chromium is not an essential trace element for mammals: effects of a “low-chromium” diet. J Biol Inorg Chem 16:381–390
Florance D (1998) Evaluation of nano- and microparticle uptake by the gastrointestinal tract. Adv Drug Deliv Rev 34:221–233
Sahoo SK, Labhasetwar V (2003) Nanotech approaches to drug delivery and imaging. Drug Discov Today 8(24):1112–1120
Desai MP, Labhasetwar V, Amidon GL, Levy RJ (1996) Gastrointestinal uptake of biodegradable microparticles: effect of particle size. Pharm Res 13:1838–1845
Hussain N, Jaitley V, Florence AT (2001) Recent advances in the understanding of uptake of microparticles across the gastrointestinal lymphatics. Adv Drug Deliv Rev 50:107–142
Wang MQ, Xu ZR (2004) Effect of chromium nanoparticle on growth performance, carcass characteristics, pork quality and tissue chromium in finishing pigs. Asian-Aust J Anim Sci 17:1118–1122
Zha LY, Wang MQ, Xu ZR, Gu LY (2007) Efficacy of chromium (III) supplementation on growth, body composition, serum parameters, and tissue chromium in rats. Biol Trace Elem Res 119:42–50
Anderson RA, Bryden NA, Polansky MM, Gautschi K (1996) Dietary chromium effects on tissue chromium concentrations and chromium absorption in rats. J Trace Elem Exp Med 9:11–19
SAS (Statistical Analysis System Inc.) (1989) SAS/STAT User’s Guide, Version 6. SAS Institute, Cary, NC
Mertz W (1969) Chromium occurrence and function in biological systems. Physiol Rev 49:163
Lukaski HC (1999) Chromium as a supplement. Annu Rev Nutr 19:279–302
Olin KL, Sterns DM, Armstrong WH, Keen CL (1994) Comparativer retention absorption of chromium-51 (Cr-51) from Cr-51 chloride, Cr-51 nicotinate and Cr-51 picolinate in a rat model. Trace Elem Electrolytes 11:182–186
Anderson RA, Bryden NA, Polansky MM, Gautschi K (1996) Dietary chromium effects on tissue chromium concentrations and chromium absorption in rats. J Trace Elem Med 9:11–25
Kottwitz K, Laschinsky N, Fischer R, Nielsen P (2009) Absorption, excretion and retention of (51) Cr from labelled Cr-(III)-picolinate in rats. Biometals 22:289–295
Clodfelder BJ, Chang C, Vincent JB (2004) Absorption of the biomimetic chromium cation triaqua-mu(3)-oxo-mu-hexapropionatotrichromium (III) in rats. Biol Trace Elem Res 98:159–170
Florence AT, Hillery AM, Hussain N, Jani PU (1995) Nanoparticles as carriers for oral peptide absorption: studies on particle uptake and fate. J Control Release 36:39–46
Kreuter J (1996) Nanoparticles and microparticles for drug and vaccine delivery. J Anat 189:503–505
Anderson KE, Stevenson BR, Rogers JA (1999) Folic acid-PEO-labeled liposomes to improve gastrointestinal absorption of encapsulated agents. J Control Release 60:189–198
Maincent P, Verge RL, Sado P, Couvreur P, Devissaguet JP (1986) Disposition kinetics and oral bioavailability of vincamine-loaded polyalkyl cyanoacrylate nanoparticles. J Pharm Sci 75:955–958
David SS (1997) Biomedical applications of nanotechnology implications for drug targeting and gene therapy. Trends Biotechnol 15:217–224
Langer R (1998) Drug delivery and targeting. Nature 392:5–10
Zha LY, Xu ZR, Wang MQ, Gu LY (2007) Chromium nanoparticle exhibits higher absorption efficiency than chromium picolinate and chromium chloride in Caco-2 cell monolayers. Biol Trace Elem Res 119:42–50
Liu PX, Chen LJ, Xie DB, Xiong XM (1997) Effects of dietary chromium on the productivity of laying hens and the distribution of chromium. Acta Agriculturae Universitatis Jiandxiensis 21(4):564–568
Chen NS, Tsai A, Dyer IA (1973) Effect of chelating agents on chromium absorption in rats. J Nutr 103:1182–1186
Borel JS, Anderson RA (1984) Chromium. In: Frieden E (ed) Biochemistry of the essential ultratrace elements. Plenum, NY, pp 175–199
Kitchalong L, Fernandez JM, Bunting LD (1993) Chromium picolinate supplementation in lamb rations: effects on performance, nitrogen balance, endocrine and metabolic parameters. J Anim Sci 71(suppl1):291, Abstract
Hasten DL, Hegsted M, Keenan MJ (1996) Effects of various forms of dietary chromium on growth and body composition in the rat. Nutr Res 17(2):283–294
Boleman SL, Boleman SJ, Bidner TD, Southern LL, Ward TL, Pontif JE, Pike MM (1995) Effect of chromium picolinate on growth, body composition, and tissue accretion in pigs. J Anim Sci 73:2033–2042
Cerulli J, Grabe DW, Gauthier I, Malone M, McGoldrick MD (1998) Chromium picolinate toxicity. Ann Pharmacother 32:428–431
Martin WR, Fuller RE (1998) Suspected chromium picolinate-induced rhabdomyolysis. Pharmacotherapy 18:860–862
Stearns DM, SrJP W, Patierno SR, Wetterhahn KE (1995) Chromium(III) picolinate produces chromosome damage in Chinese hamster ovary cells. FASEB J 9:1643–1648
Anderson RA, Bryden NA, Polansky MM (1997) Lack of toxicity of chromium chloride and chromium picolinate in rats. J Am Coll Nutr 16:273–279
Acknowledgements
This article was financially supported by National Natural Science Foundation of China (30972106).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, MQ., Li, H., He, YD. et al. Efficacy of Dietary Chromium (III) Supplementation on Tissue Chromium Deposition in Finishing Pigs. Biol Trace Elem Res 148, 316–321 (2012). https://doi.org/10.1007/s12011-012-9369-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-012-9369-x