Abstract
Background
Innate defense regulator peptide-1018 (IDR-1018) is a 12-amino acid, synthetic, immunomodulatory host defense peptide that can reduce soft tissue infections and is less likely to induce bacterial resistance than conventional antibiotics. However, IDRs have not been tested on orthopaedic infections and the immunomodulatory effects of IDR-1018 have only been characterized in response to lipopolysacharide, which is exclusively produced by Gram-negative bacteria.
Questions/purposes
We sought (1) to more fully characterize the immunomodulatory effects of IDR-1018, especially in response to Staphylococcus aureus; and (2) to determine whether IDR-1018 decreases S aureus infection of orthopaedic implants in mice and thereby protects the implants from failure to osseointegrate.
Methods
In vitro effects of IDR-1018 on S aureus were assessed by determining minimum inhibitory concentrations in bacterial broth without and with supplementation of physiologic ion levels. In vitro effects of IDR-1018 on macrophages were determined by measuring production of monocyte chemoattractant protein-1 (MCP-1) and proinflammatory cytokines by enzyme-linked immunosorbent assay. In vivo effects of IDR-1018 were determined in a murine model of S aureus implant infection by quantitating bacterial burden, macrophage recruitment, MCP-1, proinflammatory cytokines, and osseointegration in nine mice per group on Day 1 postimplantation and 20 mice per group on Day 15 postimplantation.
Results
IDR-1018 demonstrated antimicrobial activity by directly killing S aureus even in the presence of physiologic ion levels, increasing recruitment of macrophages to the site of infections by 40% (p = 0.036) and accelerating S aureus clearance in vivo (p = 0.008) with a 2.6-fold decrease in bacterial bioburden on Day 7 postimplantation. In vitro immunomodulatory activity of IDR-1018 included inducing production of MCP-1 in the absence of other inflammatory stimuli and to potently blunt excess production of proinflammatory cytokines and MCP-1 induced by lipopolysaccharide. Higher concentrations of IDR-1018 were required to blunt production of proinflammatory cytokines and MCP-1 in the presence S aureus. The largest in vivo immunomodulatory effect of IDR-1018 was to reduce tumor necrosis factor-α levels induced by S aureus by 60% (p = 0.006). Most importantly, IDR-1018 reduced S aureus-induced failures of osseointegration by threefold (p = 0.022) and increased osseointegration as measured by ultimate force (5.4-fold, p = 0.033) and average stiffness (4.3-fold, p = 0.049).
Conclusions
IDR-1018 is potentially useful to reduce orthopaedic infections by directly killing bacteria and by recruiting macrophages to the infection site.
Clinical Relevance
These findings make IDR-1018 an attractive candidate to explore in larger animal models to ascertain whether its effects in our in vitro and mouse experiments can be replicated in more clinically relevant settings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Orthopaedic implant infections are difficult to manage and often result in repeated surgical interventions and long-term treatment with intravenous antibiotics [24]. More than 100,000 orthopaedic implant infections occur in the United States each year and more than 80% are caused by Staphylococcus aureus [13]. Treatment of these infections is complicated by the fact that the surfaces of the orthopaedic plates, screws, and intramedullary nails used for fixation of the fractures provide ideal surfaces for formation of bacterial biofilms [18]. Moreover, prolonged infection and progressive inflammation result in osteolysis around the orthopaedic implants and, therefore, impair implant osseointegration and contribute to loss of fracture fixation [27]. Antibiotic resistance and the relative lack of new therapies in development further complicate treatment of orthopaedic implant infections, which are often associated with implant loosening, loss of fracture fixation, and fracture nonunion [31, 33, 41]. Additionally, increased incidence of methicillin-resistant S aureus infections is a major concern in both civilian and military orthopaedics [9, 29, 31, 45], because therapies for methicillin-resistant S aureus infections are limited.
Host-defense peptides (HDPs) represent a promising new approach to combating infection [6, 14, 15, 19]. HDPs were originally identified as small antimicrobial peptides produced by a wide variety of mammalian cell types. More recently, evidence has accumulated that the antiinfective effects of HDPs are also the result of their immunomodulatory properties such as upregulation of expression of chemokines that, in turn, recruit and activate host immune cells [6, 14, 15, 19, 28, 38]. HDPs can also modulate production of pro– and antiinflammatory cytokines to control the inflammatory response [14, 15, 19, 28, 38] and can inhibit osteoclast differentiation [20, 40] and thereby potentially can reduce inflammatory osteolysis and the lethal consequences of septic shock. As a result of their unique immunomodulatory mechanisms of action, HDPs are also less likely to induce bacterial resistance than are conventional antibiotics [14, 15]. Innate defense regulator peptide-1018 (IDR-1018) is a synthetic 12-amino acid derivative of bactenecin, a bovine HDP [42]. IDR-1018 can directly kill bacteria [42], can modulate differentiation and activation of macrophages and neutrophils, thereby regulating their production of chemokines and cytokines [1, 26, 30, 32, 37, 42], can reduce soft tissue infections [1, 37], and can disrupt bacterial biofilms [25, 35]. IDR-1018 was chosen as the focus of this study because it is the most potent IDR that has been described [37] and is therefore an attractive agent to potentially reduce orthopaedic infections and inflammatory osteolysis around orthopaedic implants. However, orthopaedic infections can be especially difficult to combat [18, 24] and IDRs have not been tested in that setting. Moreover, the immunomodulatory effects of IDR-1018 have only been characterized in response to lipopolysacharide [32, 42], which is exclusively produced by Gram-negative bacteria.
The purposes of our study were therefore (1) to more fully characterize the immunomodulatory effects of IDR-1018, especially in response to S aureus; and (2) to determine whether IDR-1018 decreases S aureus infection of orthopaedic implants in mice and thereby protects the implants from failure to osseointegrate.
Material and Methods
IDR-1018 (VRLIVAVRIWRR-NH2) was synthesized, purified to 98.4% by high-performance liquid chromatography, and validated by mass spectrometry (Biomatik, Cambridge, Canada). IDR-1018 was dissolved in sterile water (10 mg/mL) and stored at −80° C.
IDR-1018 has been reported to potently kill S aureus in bacterial broth [42]. However, physiologic ion levels block bactericidal effects of many HDPs [6, 19, 43]. We therefore determined the effect of physiologic ion levels on IDR-1018 minimum inhibitory concentrations using a modified broth microdilution method as recommended [11, 42, 43]. Thus, overnight cultures of bioluminescent, methicillin-sensitive S aureus-Xen36 (Caliper Life Sciences, Hopkinton, MA, USA) were diluted 100-fold in Mueller-Hinton broth (MHB; Difco, Franklin Lakes, NJ, USA) and incubated at 37° C until early log phase (A600/0.1 cm light path = 0.05) was reached (Nanodrop 1000; Fisher Scientific, Fair Lawn, NJ, USA). Bacterial suspensions were diluted to 5 × 105 colony-forming units (CFUs)/mL in MHB without or with physiologic ion supplementation (Table 1) and cultured overnight at 37 °C with 1/10 volume of 0.01% acetic acid containing 0.2% bovine serum albumin plus various concentrations of IDR-1018. Aliquots of the cultures were spotted on MHB agar plates, incubated overnight at 37° C, and the qualitative presence or absence of bacterial growth assessed visually as recommended [11, 42, 43].
To characterize the immunomodulatory effects of IDR-1018, production of cytokines and monocyte chemoattractant protein-1 (MCP-1) by RAW264.7 murine macrophages (ATCC, Manassas, VA, USA) was measured. RAW264.7 macrophages were maintained in minimum essential medium (Hyclone, Logan, UT, USA) with 10% fetal bovine serum (Hyclone), nonessential amino acids (Cellgro, Manassas, VA, USA), L-glutamine (Cellgro), and penicillin/streptomycin (Cellgro) at 37 °C in 5% CO2. RAW264.7 macrophages (1 × 105 cells in 2 mL of media/2-cm2 well) were incubated with IDR-1018 (7−700 μg/mL) for 45 minutes and then incubated for 24 hours in the absence or presence of 2 × 107 CFU/mL heat-killed S aureus or ultrapure lipopolysaccharide (LPS) from Escherichia coli 0111:B4 (InvivoGen, San Diego, CA, USA). Culture media were centrifuged (9000 g, 15 minutes) and supernatants stored at −20° C. MCP-1, tumor necrosis factor-α (TNF-α), interleukin (IL)-6, and IL-1β were quantified by enzyme-linked immunosorbent assay (R&D Systems, Minneapolis, MN, USA).
Murine Model of Implant Infection
Our study was approved by our Institutional Animal Care and Use Committee. In vivo experiments focused on 1 day or 15 days after implantation (Fig. 1), respectively, because higher levels of cytokines, chemokines, and macrophage recruitment occur at the early time point and because preliminary studies showed that S aureus infection blocks osseointegration at 15 days. Female mice matched for age and strain were randomized to receive saline or IDR-1018 (200 μL, diluted to 1 mg/mL in saline; Henry Schein, Melville, NY, USA) intraperitoneally 4 hours before and 24 and 48 hours after implant insertion (Fig. 1). Each group contained nine mice in the 1-day experiments and 20 mice in the 15-day experiments. Titanium alloy screw-shaped implants (0.8 mm diameter, 3.5 mm length; Ti-6Al-4 V; Antrin Miniature Specialties Inc, Fallbrook, CA, USA) were rigorously cleaned with five cycles of alternating treatments in alkali ethanol (0.1 N NaOH and 95% ethanol at 32 °C) and 25% nitric acid [5]. Overnight cultures of bioluminescent S aureus-Xen36 were diluted 100-fold in lysogeny broth (LB; Fisher Scientific) and incubated at 37° C until early log phase was reached. Bacterial suspensions were centrifuged (1500 g, 5 minutes) and resuspended in 1/30 volume of LB (3 × 1010 CFUs/mL). Rigorously cleaned implants were incubated with 100 μL of the bacterial suspensions for 20 minutes at room temperature with gentle shaking, rinsed three times in phosphate-buffered saline (PBS), and immediately implanted into pilot holes in the middiaphysis of the femur [4, 5] of 6- to 8-week-old female C57BL/6 J or macrophage Fas-induced apoptosis (MAFIA) mice. Macrophage recruitment was measured in the MAFIA mice, in which the monocyte/macrophage-specific colony-stimulating factor receptor promoter drives both expression of enhanced green fluorescent protein and a Fas apoptosis system activated by the dimerization drug, AP20187 [8, 10]. MAFIA mice are on the C57BL/6 J background and have a normal phenotype unless exposed to AP20187. Macrophage recruitment was therefore quantified by fluorescence imaging with the Maestro imaging system (Perkin Elmer, Waltham, MA, USA) 1 day postimplantation after exposing the femurs, implants, and surrounding soft tissues by dissection. Fluorescence imaging signals from green fluorescent protein were defined by automatic spectral segmentation and were quantified in automatically selected regions of interest (ROIs) encompassing both the femurs and surrounding soft tissues.
Diagrams depict time lines for in vivo experiments. Mice were euthanized on (A) Day 1 or (B) Day 15 postimplantation. Mice received either saline or IDR-1018 (200 μg in 200 μL) intraperitoneally 4 hours before and 24 and 48 hours after implant insertion. Bioluminescence imaging was performed 1, 3, 7, 10, and 14 days postimplantation.
Osseointegration failures were defined as implants that were not fixed in the femurs at the time of euthanasia and therefore could not be assessed by biomechanical pullout testing. Those implants were assigned values of 0 for the biomechanical measurements. Biomechanic pullout testing was performed immediately after euthanasia at a displacement rate of 1 mm/min as described previously [5]. Ultimate force, average stiffness, and work to maximum load were determined from the resultant load displacement curves according to ASTM standards (F543-07). To reduce preloading variability, calculations of stiffness and work began when ultimate force equaled 0.1 N.
S aureus-Xen36 contains a stable copy of the bacterial lux ABCDE operon at a single integration site and therefore emits bioluminescent signal without addition of exogenous substrate as long as the bacteria are viable [34]. Bioluminescence imaging was performed longitudinally on live mice using a Xenogen IVIS 200 system (Perkin Elmer/Caliper Life Sciences, Hopkinton, MA, USA) and data were analyzed using Xenogen Living Image 2.5 (Perkin Elmer/Caliper Life Sciences). Oval ROIs of the same size were placed on the femoral region encompassing the bioluminescence imaging signal for each mouse. Bioluminescence imaging signal intensity was quantified within each ROI on the indicated days postimplantation.
The numbers of CFUs and bacterial gene copies on the implants and in the surrounding femurs were quantified after pullout testing. Implants were sonicated for 10 minutes (50 W, 43,000 Hz) in 1 mL of PBS with 0.3% Tween-80 (Fisher Scientific, Fair Lawn, NJ, USA) followed by vortexing for 5 minutes [3]. Femurs were homogenized (Pro200H Series homogenizer; Pro Scientific, Oxford, CT, USA) in 500 μL of PBS and diluted with another 500 μL of PBS [3, 34]. CFUs in sonicates and homogenates were counted on LB agar plates. DNA was extracted from sonicates and homogenates (Power Biofilm™ DNA isolation kit; MoBio, Carlsbad, CA, USA). Real-time polymerase chain reaction assays with forward (5′–GACTTTCGCGTATTCGGCAC-3′) and reverse (5′-ATTGAGCAGCCCACTCAGTC-3′) primers that target the S aureus-Xen36 luxA gene (Primer-BLAST, National Center for Biotechnology Information) were performed (Applied Biosystems 7500; Applied Biosystems, Foster City, CA, USA) using the standard curve method for quantification as we have done previously [12].
Femur homogenates were centrifuged (9000 g, 10 minutes) and supernatants stored at –20 °C. MCP-1, TNF-α, IL-6, and IL-1β were measured by enzyme-linked immunosorbent assay (R&D Systems, Minneapolis, MN, USA).
Statistical Analysis
All error bars in the figures and tables show SDs. Statistical significance was determined by t-tests or by one-way analysis of variance (ANOVA) followed by Bonferroni post hoc tests in experiments with multiple groups. Nonparametric Mann-Whitney tests, Kruskal-Wallis ANOVA followed by Newman-Kuels post hoc tests, or van Elteren’s two-way tests were applied to data sets that were not normally distributed or were not of equal variance. Differences were reported as significant if the p value was < 0.05.
Results
Immunomodulatory Effects of IDR-1018
In vitro experiments demonstrated that, in the absence of inflammatory stimuli, high concentrations of IDR-1018 primarily act on macrophages to induce chemokine production and thereby recruit immune cells. On the other hand, in the presence of inflammatory stimuli such as occurs during bacterial infection, IDR-1018 primarily acted on macrophages to blunt excessive production of both proinflammatory cytokines and chemokines. Thus, relatively high concentrations (70–200 μg/mL) of IDR-1018 dose-dependently induced production of the chemokine MCP-1 (also known as CCL2 [black symbols in Figs. 2A, 2D]), but IDR-1018 did not induce production of TNF-α or IL-6 in the absence of other stimuli (Figs. 2B–C, 2E–F). Low concentrations (7–70 μg/mL) of IDR-1018 potently blunted LPS-induced production of MCP-1, TNF-α, and IL-6 (gray symbols in Fig. 2D–F) but higher concentrations (200–700 μg/mL) were required to blunt their production when induced by S aureus (open symbols in Fig. 2A–C). IL-1β levels in all of the groups were less than the lowest standard (3.9 pg/mL; data not shown), likely attributable to the absence of a signal 2 to activate inflammasome processing of pro–IL–1β to mature IL-1β [16]. LPS dose–response experiments showed that 20 μg/mL of IDR-1018 blunted the effects of low concentrations of LPS (10 ng/mL; gray symbols in Fig. 2G–I) but that 200 μg/mL of IDR-1018 was required to blunt the effects of a higher concentration of LPS (103–104 ng/mL; black symbols in Fig. 2G–I).
IDR-1018 has in vitro immunomodulatory effects on macrophages. RAW264.7 macrophages were treated with the indicated concentrations of IDR-1018 45 minutes before stimulation with (A–C) 2 × 107 CFU/mL heat-killed S aureus; (D–F) ultrapure LPS at 10 ng/mL; or (G–I) ultrapure LPS at the indicated concentrations. Control wells received 40 μL of sterile water as vehicle controls for IDR-1018 and LPS. Cytokines and MCP-1 were measured by enzyme-linked immunosorbent assay in the culture media after 24-hour incubation. #p < 0.05 compared with control group without IDR-1018 and without other stimulus (A–F). *p < 0.05 compared with control group without IDR-1018 but with either S aureus (A–C) or LPS (D–F). ¶p < 0.05 for overall effect of IDR-1018 (20 or 200 μg/mL; G–I). For data sets that were normally distributed and were of equal variance (white symbols in A and C, gray symbols in D, and black symbols in A), statistical analysis was by one-way ANOVA followed by Bonferroni versus control, post hoc tests. Otherwise, statistical analysis was by nonparametric Kruskal-Wallis ANOVA followed by Dunnett versus control, post hoc tests (white symbols in B, gray symbols in E and F, and black symbols in B–F) or by van Elteren’s two-way tests (G–I). For A−F, n = 3 culture wells in each group; for G–I, n = 4 culture wells in each group. All results are representative of three to four experiments of each type and are reported as means ± SD.
The in vivo immunomodulatory effects of IDR-1018 were best observed at an early time point when higher levels of cytokines, chemokines, and macrophage recruitment occur (Fig. 1A). Thus, 1 day postimplantation, IDR-1018 enhanced recruitment of macrophages to the infected implant site by 40% (p = 0.036) without affecting macrophage levels at the contralateral femur (Fig. 3A–B). Consistent with the in vitro effects of IDR-1018 (Fig. 2), there was a 60% (p = 0.006) decrease in TNF-α level 1 day postimplantation (Fig. 3C), although there was no significant difference with the available number of samples in levels of MCP-1, IL-6, or IL-1β 1 day postimplantation (Fig. 3D–F). The levels of MCP-1 and the proinflammatory cytokines were relatively low on Day 15 and were unaffected by IDR-1018 (Fig. 3G–J).
IDR-1018 has in vivo immunomodulatory effects. IDR-1018 or saline was injected intraperitoneally 4 hours before and 24 and 48 hours after insertion of implants with adherent S aureus-Xen36. (A–B) Macrophage recruitment was measured by fluorescence imaging 1 day postimplantation. MCP-1, TNF-α, IL-6, and IL-1β were measured in the surrounding bones on (C–F) Day 1 or (G–J) Day 15 postimplantation. All data sets were normally distributed and were of equal variance; thus, black bars denote means and statistical analysis was performed using t-tests. For A and C–J, n = 9–11 mice per group; for B, n = 5 mice per group. NS = nonsignificant; FLI = fluorescence imaging.
Antibacterial Effects and Osseointegration
IDR-1018 killed S aureus in vitro and decreased S aureus infection of orthopaedic implants in mice. In vitro, IDR-1018 potently killed S aureus with similar minimum inhibitory concentration values both in the absence and presence of physiologic ion levels (Table 1). One day postimplantation, IDR-1018 did not affect the in vivo bacterial burden (Fig. 4A–B). Consistent with those findings, longitudinal bioluminescence imaging demonstrated that IDR-1018 did not affect the initial bacterial burden but accelerated (p = 0.008) bacterial clearance between Days 1 and 7 postimplantation with a 2.6-fold difference in bioluminescence imaging on Day 7 (Fig. 4C). Bacterial clearance continued for a longer period of time in the absence of IDR-1018 such that the bacterial burdens were indistinguishable by Day 14 postimplantation (Fig. 4C–G).
IDR-1018 decreases implant infection. IDR-1018 or saline was injected intraperitoneally 4 hours before and 24 and 48 hours after insertion of implants with adherent S aureus-Xen36. Bacterial burden was determined on Day 1 postimplantation by measuring CFUs (A) adherent to the implants or (B) in the surrounding bones. Bacterial burden was determined (C) longitudinally by bioluminescence imaging. Bacterial burden was determined on Day 15 postimplantation by measuring (D–E) CFUs and (F–G) the S aureus-Xen36 luxA gene adherent to the implants (D and F) and in the surrounding bones (E, G). For data sets that were normally distributed and were of equal variance (A–B), black bars denote means with statistical analysis performed using t-tests. Otherwise, statistical analysis was by nonparametric, two-way van Elteren tests (C) or by nonparametric Mann-Whitney tests and black bars denote medians (D–G). For A–B, n = 9 mice per group; for C–G, n = 20 mice per group. NS = nonsignificant.
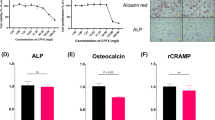
IDR-1018 protected the implants from failure to osseointegrate. These experiments focused on 15 days after implantation (Fig. 1B) because preliminary studies showed that S aureus infection blocks osseointegration during that timeframe. IDR-1018 led to a threefold (p = 0.022) decrease in the frequency of the gross failures of osseointegration that are caused by S aureus infection in the murine model (Table 2). IDR-1018 also modestly increased biomechanic measures of osseointegration. Thus, ultimate force was increased by 5.4-fold (p = 0.033; Fig. 5A) and average stiffness was increased by 4.3-fold (p = 0.049; Fig. 5B), whereas work was not detectably affected (Fig. 5C). In contrast, gross osseointegration failures were never observed 1 day postimplantation and the biomechanic measures of osseointegration in the presence of S aureus infection were higher on Day 1 than on Day 15 (compare Fig. 5D–F with Fig. 5A–C).
IDR-1018 preserves osseointegration at 15 days after implant insertion. IDR-1018 or saline was injected intraperitoneally 4 hours before and 24 and 48 hours after insertion of implants with adherent S aureus-Xen36. Osseointegration was measured by biomechanic pullout testing on (A–C) Day 15 or (D–F) Day 1 postimplantation. For comparison, biomechanic measures of sterile implants at Day 15 after insertion were 11.6 ± 2.2 Newtons (N) for ultimate force, 18.5 ± 3.8 N/mm for average stiffness, and 6.1 ± 1.6 × 10−4 joules (J) for work to the maximum force point. For data sets that were normally distributed and were of equal variance (D–F), black bars denote means with statistical analysis performed using t-tests. Otherwise, statistical analysis was by nonparametric Mann-Whitney tests and black bars denote medians (A–C). For A–C, n = 20 mice per group; for D–F, n = 9 mice per group. NS = nonsignificant.
Discussion
Orthopaedic infections are difficult to manage because of the formation of bacterial biofilms on implant surfaces [18], the inflammatory osteolysis that causes osseointegration failures and loosening of infected implants [27], and antibiotic resistance [9, 23, 29, 31, 33, 41, 45]. HDPs are promising new agents [6, 14, 15, 19] that, owing to their unique immunomodulatory mechanisms of action, are less likely to induce bacterial resistance than are conventional antibiotics [14, 15]. Moreover, HDPs can also modulate cytokine production to control the inflammatory response [14, 15, 19, 28, 38] and thereby may reduce the risk of developing inflammatory osteolysis. IDR-1018 is one of the most attractive of the HDPs [1, 26, 30, 32, 37, 39, 42]. The purposes of our study were therefore (1) to more fully characterize the immunomodulatory effects of IDR-1018, especially in response to S aureus; and (2) to determine whether IDR-1018 decreases S aureus infection of orthopaedic implants in mice and thereby protects the implants from failure to osseointegrate.
A major limitation of our study is that IDR-1018 only partially reduced the bacterial burden, proinflammatory cytokine production, and osseointegration failures induced by S aureus. The partial effects might be explained by partial effects on free-living bacteria or by production of biofilms that protected the bacteria from the antimicrobial and immunologic effects of IDR-1018 [18]. In that regard, a limitation of our study is that the longitudinal bacterial bioluminesce data does not distinguish between free-living bacteria and bacteria within biofilms. Another limitation is that our study only assessed a single dose of IDR-1018, which was chosen based on its effectiveness in reducing soft tissue infections [1]. Nonetheless, IDR-1018 might be more effective if administered more often, at a higher concentration, or locally, at the infection site. IDR-1018 might also be more effective in combination with antibiotics and implant removal, which is the most likely clinical application. Another major limitation is that we only assessed effects of IDR-1018 on a single strain of S aureus and the results with other strains or other bacterial species could be different. We also only examined female mice, which were chosen because the previous study on IDR-1018 and soft tissue S aureus infections was performed on female mice [1]. It is also always difficult to directly predict translational relevance in patients from results in rodents.
One primary purpose of our study was to more fully characterize the immunomodulatory effects of IDR-1018, especially in response to S aureus. We found that IDR-1018 was bactericidal even in the presence of physiologic ion levels, induced production of MCP-1, recruited macrophages to the site of infections, and blunted excess production of proinflammatory cytokines and MCP-1 induced by LPS in vitro or by in vivo infection with S aureus. However, the immunomodulatory effects of IDR-1018 were more complex than previously appreciated [1, 32, 42]. For example, our study was the first demonstration that IDR-1018 can blunt production of MCP-1. Moreover, IDR-1018 had relatively equivalent effects on production of cytokines and MCP-1 by macrophages in vitro, whereas in vivo, IDR-1018 had a greater effect on TNF-α levels, an intermediate effect on MCP-1, and little or no effect on IL-6. The difference between the in vitro and in vivo effects might be explained by macrophages being primarily responsible for TNF-α production in vivo [7], whereas IL-6 is primarily produced by other cell types [36] that are less affected by IDR-1018, and MCP-1 is produced by both macrophages and other cell types [44]. Consistent with this possibility, the relative in vitro cytokine levels in macrophage cell cultures compared with in vivo are highest for TNF-α, intermediate for MCP-1, and lowest for IL-6.
The other primary purpose of our study was to determine whether IDR-1018 decreases S aureus infection of orthopaedic implants in mice, thereby protecting the implants from failure to osseointegrate. Importantly, we found that IDR-1018 accelerated bacterial clearance and partially reduced both inflammatory osteolysis and infection-induced failure to osseointegrate. It is likely that decreased TNF-α levels, in response to treatment with IDR-1018, contributed to the improved osseointegration because the TNF-α levels were more strongly reduced than were the levels of other cytokines and chemokines that we measured. Moreover, TNF-α potently induces osteolysis [21] and contributes to osteolysis in other models of infection [2].
In conclusion, our results showed that IDR-1018 is potentially useful in reducing orthopaedic infections by directly killing bacteria, recruiting macrophages to the infection site, blunting excess cytokine production, and reducing osseointegration failures. IDR-1018 might therefore also reduce other examples of inflammatory osteolysis, including aseptic loosening of orthopaedic implants as a result of macrophage activation by wear particles [17, 22]. These findings make IDR-1018 an attractive candidate to explore in larger animal models to ascertain whether its effects in our in vitro and mouse experiments can be replicated in more clinically relevant settings. Additional studies to test and optimize possible strategies to increase the effectiveness of IDR-1018 are warranted. Future studies should also distinguish between effects of IDR-1018 on free-living bacteria and bacteria within biofilms and determine the effect of more potent IDRs as they are developed.
References
Achtman AH, Pilat S, Law CW, Lynn DJ, Janot L, Mayer ML, Ma S, Kindrachuk J, Finlay BB, Brinkman FS, Smyth GK, Hancock RE, Schofield L. Effective adjunctive therapy by an innate defense regulatory peptide in a preclinical model of severe malaria. Sci Transl Med. 2012;4:135ra164.
Assuma R, Oates T, Cochran D, Amar S, Graves DT. IL-1 and TNF antagonists inhibit the inflammatory response and bone loss in experimental periodontitis. J Immunol. 1998;160:403–409.
Bernthal NM, Stavrakis AI, Billi F, Cho JS, Kremen TJ, Simon SI, Cheung AL, Finerman GA, Lieberman JR, Adams JS, Miller LS. A mouse model of post-arthroplasty Staphylococcus aureus joint infection to evaluate in vivo the efficacy of antimicrobial implant coatings. PLoS One. 2010;5:e12580.
Bonsignore LA, Anderson JR, Lee Z, Goldberg VM, Greenfield EM. Adherent lipopolysaccharide inhibits the osseointegration of orthopedic implants by impairing osteoblast differentiation. Bone. 2013;52:93–101.
Bonsignore LA, Colbrunn RW, Tatro JM, Messerschmitt PJ, Hernandez CJ, Goldberg VM, Stewart MC, Greenfield EM. Surface contaminants inhibit osseointegration in a novel murine model. Bone. 2011;49:923–930.
Bowdish DM, Davidson DJ, Hancock RE. A re-evaluation of the role of host defence peptides in mammalian immunity. Curr Protein Pept Sci. 2005;6:35–51.
Bradley JR. TNF-mediated inflammatory disease. J Pathol. 2008;214:149–160.
Burnett SH, Kershen EJ, Zhang J, Zeng L, Straley SC, Kaplan AM, Cohen DA. Conditional macrophage ablation in transgenic mice expressing a Fas-based suicide gene. J Leukoc Biol. 2004;75:612–623.
Calhoun JH, Murray CK, Manring MM. Multidrug-resistant organisms in military wounds from Iraq and Afghanistan. Clin Orthop Relat Res. 2008;466:1356–1362.
Chinnery HR, Carlson EC, Sun Y, Lin M, Burnett SH, Perez VL, McMenamin PG, Pearlman E. Bone marrow chimeras and c-fms conditional ablation (Mafia) mice reveal an essential role for resident myeloid cells in lipopolysaccharide/TLR4-induced corneal inflammation. J Immunol. 2009;182:2738–2744.
Clinical and Laboratory Standards Institute. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; Approved Standard. 8th ed. Wayne, PA, USA: Clinical and Laboratory Standards Institute; 2009.
Dai JC, He P, Chen X, Greenfield EM. TNFalpha and PTH utilize distinct mechanisms to induce IL-6 and RANKL expression with markedly different kinetics. Bone. 2006;38:509–520.
Darouiche RO. Treatment of infections associated with surgical implants. N Engl J Med. 2004;350:1422–1429.
Diamond G, Beckloff N, Weinberg A, Kisich KO. The roles of antimicrobial peptides in innate host defense. Curr Pharm Des. 2009;15:2377–2392.
Finlay BB, Hancock RE. Can innate immunity be enhanced to treat microbial infections? Nat Rev Microbiol. 2004;2:497–504.
Franchi L, Munoz-Planillo R, Nunez G. Sensing and reacting to microbes through the inflammasomes. Nat Immunol. 2012;13:325–332.
Greenfield E. Particulate matter and host reactions. In: Wnek G, Bowlin G, eds. Encyclopedia of Biomaterials and Biomedical Engineering. New York, NY, USA: Taylor Francis; 2006:1–8.
Gristina AG, Costerton JW. Bacterial adherence to biomaterials and tissue. The significance of its role in clinical sepsis. J Bone Joint Surg Am. 1985;67:264–273.
Hancock RE, Sahl HG. Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat Biotechnol. 2006;24:1551–1557.
Horibe K, Nakamichi Y, Uehara S, Nakamura M, Koide M, Kobayashi Y, Takahashi N, Udagawa N. Roles of cathelicidin-related antimicrobial peptide in murine osteoclastogenesis. Immunology. 2013;140:344–351.
Horowitz M, Lorenzo J. Local regulators of bone: IL-1, TNF, lymphotoxin, interferon-γ, the LIF/IL-6 family and additional cytokines. In: Bilezikian JP, Raisz LG, Martin TJ, eds. Principles of Bone Biology. 3rd ed. Amsterdam, The Netherlands: Academic Press; 2008:1209–1234.
Ingham E, Fisher J. The role of macrophages in osteolysis of total joint replacement. Biomaterials. 2005;26:1271–1286.
Johnson EN, Burns TC, Hayda RA, Hospenthal DR, Murray CK. Infectious complications of open type III tibial fractures among combat casualties. Clin Infect Dis. 2007;45:409–415.
Kim BN, Kim ES, Oh MD. Oral antibiotic treatment of staphylococcal bone and joint infections in adults. J Antimicrob Chemother. 2014;69:309–322.
Mansour SC, de la Fuente-Nunez C, Hancock RE. Peptide IDR-1018: modulating the immune system and targeting bacterial biofilms to treat antibiotic-resistant bacterial infections. J Pept Sci. 2014 Oct 31 [Epub ahead of print].
Mayer ML, Blohmke CJ, Falsafi R, Fjell CD, Madera L, Turvey SE, Hancock RE. Rescue of dysfunctional autophagy attenuates hyperinflammatory responses from cystic fibrosis cells. J Immunol. 2013;190:1227–1238.
Montanaro L, Testoni F, Poggi A, Visai L, Speziale P, Arciola CR. Emerging pathogenetic mechanisms of the implant-related osteomyelitis by Staphylococcus aureus. Int J Artif Organs. 2011;34:781–788.
Mookherjee N, Brown KL, Bowdish DM, Doria S, Falsafi R, Hokamp K, Roche FM, Mu R, Doho GH, Pistolic J, Powers JP, Bryan J, Brinkman FS, Hancock RE. Modulation of the TLR-mediated inflammatory response by the endogenous human host defense peptide LL-37. J Immunol. 2006;176:2455–2464.
Murray CK, Yun HC, Griffith ME, Thompson B, Crouch HK, Monson LS, Aldous WK, Mende K, Hospenthal DR. Recovery of multidrug-resistant bacteria from combat personnel evacuated from Iraq and Afghanistan at a single military treatment facility. Mil Med. 2009;174:598–604.
Niyonsaba F, Madera L, Afacan N, Okumura K, Ogawa H, Hancock RE. The innate defense regulator peptides IDR-HH2, IDR-1002, and IDR-1018 modulate human neutrophil functions. J Leukoc Biol. 2013;94:159–170.
Patel A, Calfee RP, Plante M, Fischer SA, Arcand N, Born C. Methicillin-resistant Staphylococcus aureus in orthopaedic surgery. J Bone Joint Surg Br. 2008;90:1401–1406.
Pena OM, Afacan N, Pistolic J, Chen C, Madera L, Falsafi R, Fjell CD, Hancock RE. Synthetic cationic peptide IDR-1018 modulates human macrophage differentiation. PLoS One. 2013;8:e52449.
Perez F, Hujer AM, Hujer KM, Decker BK, Rather PN, Bonomo RA. Global challenge of multidrug-resistant Acinetobacter baumannii. Antimicrob Agents Chemother. 2007;51:3471–3484.
Pribaz JR, Bernthal NM, Billi F, Cho JS, Ramos RI, Guo Y, Cheung AL, Francis KP, Miller LS. Mouse model of chronic post-arthroplasty infection: noninvasive in vivo bioluminescence imaging to monitor bacterial burden for long-term study. J Orthop Res. 2011;30:335–340.
Reffuveille F, de la Fuente-Nunez C, Mansour S, Hancock RE. A broad-spectrum antibiofilm peptide enhances antibiotic action against bacterial biofilms. Antimicrob Agents Chemother. 2014;58:5363–5371.
Rincon M. Interleukin-6: from an inflammatory marker to a target for inflammatory diseases. Trends Immunol. 2012;33:571–577.
Rivas-Santiago B, Castaneda-Delgado JE, Rivas Santiago CE, Waldbrook M, Gonzalez-Curiel I, Leon-Contreras JC, Enciso-Moreno JA, del Villar V, Mendez-Ramos J, Hancock RE, Hernandez-Pando R. Ability of innate defence regulator peptides IDR-1002, IDR-HH2 and IDR-1018 to protect against Mycobacterium tuberculosis infections in animal models. PLoS One. 2013;8:e59119.
Scott MG, Dullaghan E, Mookherjee N, Glavas N, Waldbrook M, Thompson A, Wang A, Lee K, Doria S, Hamill P, Yu JJ, Li Y, Donini O, Guarna MM, Finlay BB, North JR, Hancock RE. An anti-infective peptide that selectively modulates the innate immune response. Nat Biotechnol. 2007;25:465–472.
Steinstraesser L, Hirsch T, Schulte M, Kueckelhaus M, Jacobsen F, Mersch EA, Stricker I, Afacan N, Jenssen H, Hancock RE, Kindrachuk J. Innate defense regulator peptide 1018 in wound healing and wound infection. PLoS One. 2012;7:e39373.
Supanchart C, Thawanaphong S, Makeudom A, Bolscher JG, Nazmi K, Kornak U, Krisanaprakornkit S. The antimicrobial peptide, LL-37, inhibits in vitro osteoclastogenesis. J Dent Res. 2012;91:1071–1077.
Talbot GH, Bradley J, Edwards JE Jr, Gilbert D, Scheld M, Bartlett JG. Bad bugs need drugs: an update on the development pipeline from the Antimicrobial Availability Task Force of the Infectious Diseases Society of America. Clin Infect Dis. 2006;42:657–668.
Wieczorek M, Jenssen H, Kindrachuk J, Scott WR, Elliott M, Hilpert K, Cheng JT, Hancock RE, Straus SK. Structural studies of a peptide with immune modulating and direct antimicrobial activity. Chem Biol. 2010;17:970–980.
Wiegand I, Hilpert K, Hancock RE. Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nat Protoc. 2008;3:163–175.
Yadav A, Saini V, Arora S. MCP-1: chemoattractant with a role beyond immunity: a review. Clin Chim Acta. 2010;411:1570–1579.
Yun HC, Branstetter JG, Murray CK. Osteomyelitis in military personnel wounded in Iraq and Afghanistan. J Trauma. 2008;64(Suppl):S163–168; discussion S168.
Acknowledgments
We thank Ozan Akkus for advice on biomechanical testing; Nicholas M. Bernthal and Lloyd S. Miller for advice on murine implant infection models; David J. Corn for assistance with bioluminescence imaging and fluorescence imaging; Hani A. Essber, Sona Haku, and Jenifer Mikulan for assistance with the mouse surgeries; Susan W. Farmer and Robert E. W. Hancock for advice on handling IDR-1018; Bryan S. Hausman for assistance with both polymerase chain reaction primer design and mouse surgery; Steve H. Marshall for advice on microbiology methods; Eric Pearlman for providing the MAFIA transgenic mice; Mark D. Schluchter for statistical assistance; and Jack Wilber for suggestions on clinical relevance. We would like to acknowledge the support of the Case Center for Imaging Research which is a shared resource for Case Western Reserve University and the Case Comprehensive Cancer Center.
Author information
Authors and Affiliations
Corresponding author
Additional information
One or more of the authors has received research funding from the Department of Defense Peer Reviewed Orthopaedic Research Program Idea Development Award (EMG), the Harry E. Figgie III MD Professorship (EMG), the Mochida Memorial Foundation for Medical and Pharmaceutical Research (HC), the Cleveland Department of Veterans Affairs (RAB), the Veterans Affairs Merit Review Program award number 1I01BX001974 (RAB), the Geriatric Research Education and Clinical Center VISN 10 (RAB), the National Institute of Allergy and Infectious Diseases of the National Institutes of Health awards R01AI063517 and R01AI10056 (RAB), and the Antibiotic Resistance Leadership Group under National Institutes of Health award UM1AI104681 (RAB).
The content is solely the responsibility of the authors and does not represent the official views of the Department of Defense, the Mochida Memorial Foundation for Medical and Pharmaceutical Research, the National Institutes of Health, or the Department of Veterans Affairs.
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research ® editors and board members are on file with the publication and can be viewed on request.
Clinical Orthopaedics and Related Research ® neither advocates nor endorses the use of any treatment, drug, or device. Readers are encouraged to always seek additional information, including FDA-approval status, of any drug or device prior to clinical use.
Each author certifies that his or her institution approved the animal protocol for this investigation and that all investigations were conducted in conformity with ethical principles of research.
About this article
Cite this article
Choe, H., Narayanan, A.S., Gandhi, D.A. et al. Immunomodulatory Peptide IDR-1018 Decreases Implant Infection and Preserves Osseointegration. Clin Orthop Relat Res 473, 2898–2907 (2015). https://doi.org/10.1007/s11999-015-4301-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11999-015-4301-2