Abstract
Patterns of motion in the native knee show substantial variability. Guided motion prosthetic designs offer stability but may limit natural variability. To assess these limits, we therefore determined the in vivo kinematic patterns for patients having a cruciate-substituting TKA of one design and determined the intersurgeon variability associated with a guided-motion prosthetic design. Three-dimensional femorotibial contact positions were evaluated for 86 TKAs in 80 subjects from three different surgeons using fluoroscopy during a weightbearing deep knee bend. The average posterior femoral rollback of the medial and lateral condyles for all TKAs from full extension to maximum flexion was −14.0 mm and −23.0 mm, respectively. The average axial tibiofemoral rotation from full extension to maximum flexion for all TKAs was 10.8°. The average weightbearing range of motion (ROM) was 109º (range, 60º–150º; standard deviation, 18.7º). Overall, the TKA showed axial rotation patterns similar to those of the normal knee, although less in magnitude. Surgeon-to-surgeon comparison revealed dissimilarities, showing the surgical technique and soft tissue handling influence kinematics in a guided-motion prosthetic design.
Level of Evidence: Level III, therapeutic study. See Guidelines for Authors for a complete description of levels of evidence.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In vivo kinematic patterns in subjects undergoing TKA vary considerably from the normal knee. This is supported with findings from fluoroscopy, studies using in vitro analyses, and those using external markers associated with gait laboratory systems [1, 2, 4, 8–10, 12, 13, 16, 18, 19, 21, 22, 24, 25, 34–37]. With increasing knee flexion, the normal knee reportedly experiences more posterior motion of the lateral condyle leading to internal rotation of the tibia with respect to the femur [18, 22, 25]. In contrast to the normal knee, in vivo kinematic analyses suggest subjects undergoing TKA often experience a motion pattern opposite the normal knee where the condyles slide in the anterior direction with increasing knee flexion [2, 8, 9, 24, 28, 31, 34–37]. Also, in vivo kinematic studies involving patients undergoing TKA have documented reverse rotational patterns [2, 10] and lateral condylar liftoff [13, 21, 34, 37]. There is evidence these abnormal kinematic patterns lead to decreased ROM [3, 11, 38]. In addition, these abnormal kinematic patterns possibly relate to inferior functional performance of daily activities by patients undergoing TKA. Noble et al. [33] reported substantial differences in functional capacities between patients who had successful TKAs and their age-matched peers with normal knees. The activities causing the most trouble in the TKA group were related to loaded flexion and stability: squatting, kneeling, gardening, and turning/cutting. Our current inability to restore physiologic function is multifactorial and related to irreversible damage caused by the arthritic process, surgical damage caused by insertion of the implant, loss of proprioception, and kinematic changes induced by the prosthesis [3, 32, 34].
In the spectrum of kinematic abnormalities, a couple studies suggest subjects having a posterior stabilized (PS) TKA have less abnormal knee kinematics in deeper flexion and greater flexion than subjects having a posterior cruciate-retaining (PCR) TKA [8, 38]. However, measurement in weightbearing conditions typically reduces the observed ROM [11]. PCR and PS TKAs have similar kinematic patterns in early flexion activities such as gait [9, 10].
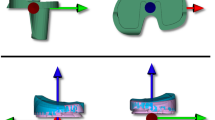
Two important features influence stability and kinematics in the TKA implant: surface geometry of the bearing surfaces and a mechanical interaction between the polyethylene and the femoral component, eg, a cam and post mechanism. In an in vitro study, Bull et al. related the changes in knee kinematics to the articular geometry of a single radius design. They observed only a small tibial posterior translation between 40° and 90° flexion [6]. A cruciate-retaining implant with differing geometries on the lateral and medial condyles and a changing radius of curvature of the femoral condyle showed improved kinematic patterns [26]. Recently, an assumption was made and implemented into a new TKA design that attempts to substitute for the anterior cruciate ligament (ACL) and posterior cruciate ligament (PCL) function by using an asymmetric tibial plateau geometry and a dual-cam substitution attempting to produce more normal-like kinematic patterns [39] (Fig. 1). One would presume that if the implant behaved strictly as a mechanically constraint-guided motion system, one would observe no intersurgeon and intrasurgeon differences.
The dual cam mechanism of the implant substituting for ACL and PCL function is shown. The posterior cam is asymmetric, driving the internal rotation of the tibia with increasing flexion [35].
The aims of our study are twofold: (1) to describe the in vivo kinematics for a TKA that substitutes for the ACL and PCL and attempts to guide the motion using dual-cam constraints function using an in vivo, fluoroscopic analysis during a weightbearing, deep knee bend; and (2) to determine and compare the differences in patient groups operated on by different surgeons.
Materials and Methods
The average age for all subjects included in this study was 66.5 years (range, 40–82 years; standard deviation [SD], 7.6). The averages for height, weight, and body mass index for all subjects in this study were 169.1 cm (range, 150–188 cm; SD, 9.3 cm), 82 kg (range, 56–126 kg; SD, 13.6 kg), and 28.6 kg/m2 (range, 21.2–40.7 kg/m2; SD, 3.9 kg/m2), respectively (Table 1). We assessed in vivo knee kinematics for 80 subjects implanted with 86 Journey Cruciate substituting (BCS) prostheses (Smith and Nephew, Memphis TN) by three surgeons (JB, JV, MN) at three hospitals. Surgeon 1 provided 40 TKAs from 38 patients, Surgeon 2 provided 35 TKAs from 34 patients, and Surgeon 3 provided 11 TKAs from eight patients. All surgeons used an anteromedial surgical exposure and measured resection surgical technique with the same instrument set provided by the manufacturer. We used a list of chronologic consecutive patients with a well-functioning TKA and selected those judged clinically successful (Hospital for Special Surgery scores greater than 90) [20] to contact for their consent to be included in this study. The list was taken from a prospective database containing followup of all patients undergoing TKAs. Ethical Committee or Institution Review Board approval was obtained for each of the three centers involved and informed consent for all patients participating in the study.
Fluoroscopic examinations were performed in Pellenberg, Belgium (Surgeon 1), Brugge, Belgium (Surgeon 2), and Knoxville, TN (Surgeon 3). We asked each subject to perform successive deep knee bends to maximum weightbearing flexion. Patients were examined using a C-arm-type fluoroscopic unit after an initial trial squat without fluoroscopy. As soon as good imaging was obtained from the full squat, the exercise was finished. The fluoroscopic images were stored on a digital video recorder for subsequent analysis.
Using a three-dimensional (3D) model-fitting approach, we determined the relative position of knee implant components in 3D from a single-perspective fluoroscopic image by manipulating a CAD model in 3D space using a previously described model-fitting process that was validated [29, 30]. Individual fluoroscopic frames were captured and analyzed at full extension (0°), 30°, 60°, 90° (if obtained), 120° (if obtained), and maximum (Max) knee flexion. The correct 3D fit was achieved when the silhouettes of the femoral and tibial implant components best matched the corresponding components in the fluoroscopic image. We determined the anteroposterior (AP) contact positions for the medial and lateral condyles, axial rotation of the femoral component relative to the tibial component, condylar liftoff, and weightbearing ROM. The femorotibial contact positions were determined by finding the lowest point on the femoral component relative to the tibial component for the medial and lateral sides. The AP position was measured as the orthogonal distance from these points to the midline of the tibial component. Positive values indicated the position anterior to the midline; negative values indicated position posterior to the midline.
The insert design of the implant studied has unequal condylar thicknesses on the medial and lateral sides of the femoral component and the tibial insert in an attempt to recreate a physiologic joint line in conjunction with maintaining perpendicular bone cuts. To accurately determine condylar liftoff, the 3D CAD model of the polyethylene insert was used in the analysis where it was transparent during the overlay process but reappears in the image attached to the tibial tray after the best fit is determined. Then, the algorithm was used to determine the distance from each condyle to the polyethylene insert. The difference between these two measurements was calculated to determine the occurrence of condylar liftoff at any flexion increment. We reported condylar liftoff only when the measured difference was greater than 1.0 and then was visually inspected by the operator to verify this occurrence.
We determined differences in femoral condylar positions for the medial and lateral sides and tibiofemoral axial rotation orientation (angle) at all increments (0°, 30°, 60°, 90°, 120°, and Max) of weightbearing flexion between the groups of patients treated by each of the three surgeons. We also determined differences in femoral condylar movement for the medial and lateral sides and relative tibiofemoral axial rotation among all increments of weightbearing flexion between the three groups. Differences in kinematic data, including orientation at the analyzed increments and the movement between these increments, were tested between the surgeon groups using Student’s t-test and the Tukey-Kramer test using JMP Statistical Discovery Software (SAS Institute, Cary, NC).
Results
All patients in this study undergoing TKAs experienced posterior femoral rollback (PFR) of their medial and lateral condyles from full extension to maximum knee flexion. At full extension, the average medial and lateral condyle contact positions were 5.2 mm and 7.2 mm, respectively (Table 2). At maximum knee flexion, the average medial condyle contact position moved posterior to −8.8 mm and the lateral contact position also moved in the posterior direction to −15.9 mm. Therefore, from full extension to maximum knee flexion, the average amounts of PFR were −14.0 mm medially and −23.0 mm laterally. The most noticeable condylar movement occurred between 0° and 30° knee flexion, in which 66.3% and 68.1% of the medial and lateral condylar movements were experienced by the subjects in this study (Table 3). On average, the TKAs we analyzed experienced a normal-like axial rotation pattern from full extension to maximum flexion. The average amount of axial rotation from full extension to maximum knee flexion was 10.8° (range, −4.2°–24.7°; SD, 6.2°) (Table 4). The average weightbearing ROM for the TKA in this study was 109º (range 60º–150º; SD, 18.7º). Sixty-three (73.3%) of the subjects achieved greater than 100° weightbearing flexion and 25 (29.1%) experienced greater than 120° weightbearing flexion (Table 5). Some patients had near normal kinematics and ROM (Video 1, Supplemental materials are available with the online version of CORR) and some had aberrant kinetics with poor ROM (Video 2, Supplemental materials are available with the online version of CORR).
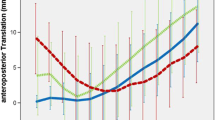
We observed several differences between the patient groups treated by the different surgeons. The medial condylar position at full extension for Surgeon 3’s group was more posterior compared with the groups of Surgeons 1 and 2 (p = 0.007 and p = 0.0008, respectively). We observed no difference in lateral condylar position at full extension. At Max flexion, the mean medial condylar position for Surgeon 1 was more anterior than for Surgeons 2 and 3 (p = 0.0286 and p = 0.002, respectively), and the mean lateral condylar position was more anterior for Surgeon 3 than for Surgeons 1 or 2 (p = 0.0042 and p = 0.0034, respectively) (Figs. 2, 3). The medial AP movement from 0° to maximum flexion for Surgeon 3 was different from Surgeon 1 (p = 0.0139) (Fig. 2). All surgeon groups were similar (p > 0.05) in AP movement on the lateral side from 0° to maximum flexion (Fig. 3). We observed other differences between surgeon groups in AP condylar position and movement. The most consistent difference in movement was on the medial side between Surgeons 1 and 2, which showed differences in movement from 0° to 30° (p = 0.0062), 60° (p = 0.005), and 90° (p = 0.0213). Other differences occurred between Surgeon 3 and the other two groups with respect to AP condylar position for both condyles (Table 6). Although the overall amount of axial rotation was similar, the angles at specific flexion increments were different when compared among surgeon groups. However, when comparing differences in the ranges, the only difference occurred between from 0° and 90° flexion in which Surgeon 2 had more relative axial rotation than the groups of Surgeon 1 (p = 0.0136) and Surgeon 2 (p = 0.0038) (Table 6). Similar to condylar AP movement, the majority of the overall axial rotation occurred in the first 30° knee flexion for all groups (Table 3).
The average medial AP position for all TKAs and groups by surgeon are shown. The error bars indicate one standard deviation for all surgeon groups, showing the differences between these groups. The medial AP movement from 0° to maximum flexion for patients from Surgeon 3 (green) was different from patients from Surgeon 1 (blue) (p = 0.0139).
The average lateral AP positions for all TKAs and groups by surgeon including error bars for the patient group from Surgeon 3 are shown. All surgeon groups were similar (p > 0.05) in AP movement on the lateral side from 0° to maximum flexion. At maximum flexion, the lateral condylar position of Surgeon’s 3 group was more anterior than for the groups of Surgeons 1 and 2 (p = 0.0042 and p = 0.0034, respectively).
Discussion
The patterns of motion in the normal knee exhibit substantial variability. Various prosthetic designs with guided motion offer stability but may limit the natural variability. However, if the implant behaved strictly as a mechanically constraint-guided motion system, one would presume there would be no intersurgeon and intrasurgeon differences. The primary goal of our study therefore was to describe the in vivo kinematics after implantation of a TKA prosthesis based on a guided-motion principle. We also studied intrasurgeon and intersurgeon differences to evaluate the influence of a guided motion prosthesis with dual post-cam constraints on the in vivo kinematics.
The study has some limitations. First, the methods allowed us to detect and describe kinematic differences between groups of patients treated by different surgeons but it does not relate these differences to soft tissue conditions. Because the operations were performed without the use of a surgical navigation system, the medial, lateral, and AP laxity at the end of the procedure eventually was decided by the surgeon’s subjective assessment of joint stability and could not be measured in an accurate and reproducible way. In analogy, fluoroscopic measurements of the weightbearing ROM and kinematics before the operation were not available for comparison with the postoperative results.
Numerous kinematic analyses of the normal knee have documented greater mean posterior motion of the lateral condyle relative to the medial condyle, leading to a mean internal rotation of the tibia with progressive knee flexion [9, 15, 23, 25, 27, 29]. Komistek et al. reported the lateral condyle achieved more posterior motion than the medial condyle, 19.2 mm and 3.4 mm, respectively, with increasing knee flexion during a deep knee bend [25]. They also reported the occurrence of intersubject variability. From full extension to maximum knee flexion, the medial condyle translation ranged from +3 mm anterior motion to −4.6 mm posterior motion. In comparison, the lateral condyle movement was only posterior, ranging from −5.8 to −24.7 mm. The average tibiofemoral rotation during flexion was 16.8° (range, 2.1°–27.1°). Bank’s group used computed tomography derived bone models for model registration and added MRI-derived articular surfaces for obtaining higher accuracy of the contact areas [29]. They observed the greatest femoral external rotation during the squat activity but reported no posterior subluxation of either femoral condyle in maximum knee flexion. In comparing kneel, squat, and stairclimbing motions, they found considerable variations in knee kinematics depending on the activity. In the native knee, different methods seem to reveal different kinematic patterns: the rotational patterns are variable and may be influenced by the bearing surface forces, further influenced by foot position, body inertia, and muscular activity. A guided motion prosthetic knee design carries the risk of imposing a motion pattern and excessively reducing this natural variability. Our data show subjects experienced PFR of their lateral condyle (mean 23 mm) and a lesser amount of PFR of their medial condyle (mean 14 mm) during a loaded deep knee bend. We observed greater medial condyle PFR than previously reported for the normal knee [23, 25, 29], leading to similar axial rotation patterns, but lower in magnitude than that of the normal knee (10.8° for the patients in this study versus 16.8° in the native knee). This greater medial PFR as compared with the normal knee raises concerns because it potentially can overload the medial structures of the knee. The variable analyzed in this study with the most immediate impact on the patient’s function is the weightbearing ROM. The ROM reported in this study would be considered low when compared with passive ROM. Dennis et al. [11] reported weightbearing flexion can be 20° less, on average, than passive flexion with the same group of patients. We found an average weightbearing flexion of 109º with a maximum flexion of 150º. Sixty-three subjects (73.3%) achieved greater than 100° weightbearing flexion and 25 (29.1%) experienced greater than 120° weightbearing flexion. So far, there is no clinical evidence relating certain kinematic patterns to better outcomes like improved longevity or better wear performance. Dorr even suggested wear is not an issue in TKA as long as prosthetic designs do not try to reproduce normality in kinematics [14]. In contrast, recent in vivo work comparing laboratory data with retrieval specimen analysis supported the use of so-called “high flexion” designs because they improve contact conditions and preserve contact area at high flexion angles [7]. In addition, there is in vitro evidence that multidirectional sliding is detrimental to the polyethylene, giving a theoretical advantage to guided motion [5]. Fregly et al. [17] developed another argument illustrating the close relation between kinematic behavior and wear patterns. They wrote a computer model to predict wear patterns based on kinematic in vivo analysis and validated this model against a retrieval specimen [17].
The kinematic patterns we observed for this TKA prosthesis were consistent for subject-to-subject comparison, reflected by the low SDs in the data. Although the overall motion patterns were similar, we observed intrasurgeon differences in the in vivo kinematics: the relative axial rotation between 0° and 90° flexion was greater for the patients of Surgeon 2 compared with the two other surgeon groups (Table 4). This might be the result of a more externally rotated orientation of the tibial component. Looking at AP condylar position, the medial condyle is on average more posterior and the lateral condyle more anterior in the patients of Surgeon 3. This also may be a phenomenon of the small sample size, but the differences in midflexion were statistically different between Surgeon 3 and the other two surgeon groups. On the basis of the reported differences in kinematic patterns among the surgeon groups, the null hypothesis that the implant would act as a constraint mechanical device is refuted.
We found consistent kinematic patterns from patient to patient. Surgeon-to-surgeon comparison revealed some dissimilarities, showing surgical technique and soft tissue handling do play a role when using this particular implant. Although we did not observe normal kinematics in all patients, all patients achieved femoral rollback during flexion and the axial rotation pattern was normal in pattern for 95% of the patients.
References
Andriacchi TP. Functional analysis of pre and post-knee surgery: total knee arthroplasty and ACL reconstruction. J Biomech Eng. 1993;115:575–581.
Banks SA, Markovich GD, Hodge WA. In vivo kinematics of cruciate-retaining and -substituting knee arthroplasties. J Arthroplasty. 1997;12:297–304.
Bellemans J, Banks S, Victor J, Vandenneucker H, Moemans A. Fluoroscopic analysis of the kinematics of deep flexion in total knee arthroplasty: influence of posterior condylar offset. J Bone Joint Surg Br. 2002;84:50–53.
Bertin KC, Komistek RD, Dennis DA, Hoff WA, Anderson DT, Langer T. In vivo determination of posterior femoral rollback for subjects having a NexGen posterior cruciate-retaining total knee arthroplasty. J Arthroplasty. 2002;17:1040–1048.
Blunn GW, Walker PS, Joshi A, Hardinge K. The dominance of cyclic sliding in producing wear in total knee replacements. Clin Orthop Relat Res. 1991;273:253–260.
Bull AM, Kessler O, Alam M, Amis AA. Changes in knee kinematics reflect the articular geometry after arthroplasty. Clin Orthop Relat Res. 2008;466:2491–2499.
D’Lima DD, Steklov N, Fergly BJ, Banks SA, Colwell CW Jr. In vivo contact stresses during activities of daily living after knee arthroplasty. J Orthop Res. 2008;26:1549–1555.
Dennis DA, Komistek RD, Colwell CE Jr, Ranawat CS, Scott RD, Thornhill TS, Lapp MA. In vivo anteroposterior femorotibial translation of total knee arthroplasty: a multicenter analysis. Clin Orthop Relat Res. 1998;356:47–57.
Dennis DA, Komistek RD, Mahfouz MR, Haas BD, Stiehl JB. Multicenter determination of in vivo kinematics after total knee arthroplasty. Clin Orthop Relat Res. 2003;416:37–57.
Dennis DA, Komistek RD, Mahfouz MR, Walker SA, Tucker A. A multicenter analysis of axial femorotibial rotation after total knee arthroplasty. Clin Orthop Relat Res. 2004;428:180–189.
Dennis DA, Komistek RD, Scuderi GR, Zingde SM. Factors affecting flexion after total knee arthroplasty. Clin Orthop Relat Res. 2007;464:53–60.
Dennis DA, Komistek RD, Stiehl JB, Walker SA, Dennis KN. Range of motion after total knee arthroplasty: the effect of implant design and weight-bearing conditions. J Arthroplasty. 1998;13:748–752.
Dennis DA, Komistek RD, Walker SA, Cheal EJ, Stiehl JB. Femoral condylar lift-off in vivo in total knee arthroplasty. J Bone Joint Surg Br. 2001;83:33–39.
Dorr LD. Contrary view: wear is not an issue. Clin Orthop Relat Res. 2002;404:96–99.
Draganich LF, Andriacchi TP, Andersson GB. Interaction between intrinsic knee mechanics and the knee extensor mechanism. J Orthop Res. 1987;5:539–547.
Feng EL, Stulberg SD, Wixson RL. Progressive subluxation and polyethylene wear in total knee replacements with flat articular surfaces. Clin Orthop Relat Res. 1994;299:60–71.
Fregly BJ, Sawyer WG, Harman MK, Banks SA. Computational wear prediction of a total knee replacement from in vivo kinematics. J Biomech. 2005;38:305–314.
Hill PF, Vedi V, Williams A, Iwaki H, Pinskerova V, Freeman MA. Tibiofemoral movement 2: the loaded and unloaded living knee studied by MRI. J Bone Joint Surg Br. 2000;82:1196–1198.
Hsieh HH, Walker PS. Stabilizing mechanisms of the loaded and unloaded knee joint. J Bone Joint Surg Am. 1976;58:87–93.
Insall JN, Hood RW, Flawn LB, Sullivan DJ. The total condylar knee prosthesis in gonarthrosis: a five to nine-year follow-up of the first one hundred consecutive replacements. J Bone Joint Surg Am. 1983;65:619–628.
Insall JN, Scuderi GR, Komistek RD, Math K, Dennis DA, Anderson DT. Correlation between condylar lift-off and femoral component alignment. Clin Orthop Relat Res. 2002;403:143–152.
Iwaki H, Pinskerova V, Freeman MA. Tibiofemoral movement 1: the shapes and relative movements of the femur and tibia in the unloaded cadaver knee. J Bone Joint Surg Br. 2000;82:1189–1195.
Johal P, Williams A, Wragg P, Hunt D, Gedroyc W. Tibio-femoral movement in the living knee: a study of weight bearing and non-weight bearing knee kinematics using `interventional’ MRI. J Biomech. 2005;38:269–276.
Komistek RD, Dennis DA. Fluoroscopic Analysis of Total Knee Replacement. Surgery of the Knee. Vol 2, Ed 3. New York, NY: Churchill Livingstone; 2001:1695.
Komistek RD, Dennis DA, Mahfouz M. In vivo fluoroscopic analysis of the normal human knee. Clin Orthop Relat Res. 2003;410:69–81.
Komistek RD, Mahfouz MR, Bertin KC, Rosenberg A, Kennedy W. In vivo determination of total knee arthroplasty kinematics: a multicenter analysis of an asymmetrical posterior cruciate retaining total knee arthroplasty. J Arthroplasty. 2008;23:41–50.
Kurosawa H, Walker PS, Abe S, Garg A, Hunter T. Geometry and motion of the knee for implant and orthotic design. J Biomech. 1985;18:487–499.
Lewis P, Rorabeck CH, Bourne RB, Devane P. Posteromedial tibial polyethylene failure in total knee replacements. Clin Orthop Relat Res. 1994;299:11–17.
Moro-oka T, Hamai S, Miura H, Shimoto T, Higaki H, Fregly BJ, Iwamoto Y, Banks SA. Dynamic activity dependence of in vivo normal knee kinematics. J Orthop Res. 2008;26:428–434.
Mahfouz MR, Hoff WA, Komistek RD, Dennis DA. A robust method for registration of three-dimensional knee implant models to two-dimensional fluoroscopy images. IEEE Trans Med Imaging. 2003;22:1561–1574.
Markolf KL, Finerman GM, Amstutz HC. In vitro measurements of knee stability after bicondylar replacement. J Bone Joint Surg Am. 1979;61:547–557.
Massin P, Gournay A. Optimization of the posterior condylar offset, tibial slope, and condylar roll-back in total knee arthroplasty. J Arthroplasty. 2006;21:889–896.
Noble PC, Gordon MJ, Weiss JM, Reddix RN, Conditt MA, Mathis KB. Does total knee replacement restore normal knee function? Clin Orthop Relat Res. 2005;431:157–165.
Stiehl JB, Dennis DA, Komistek RD, Crane HS. In vivo determination of condylar lift-off and screw-home in a mobile-bearing total knee arthroplasty. J Arthroplasty. 1999;14:293–299.
Stiehl JB, Dennis DA, Komistek RD, Keblish PA. In vivo kinematic analysis of a mobile bearing total knee prosthesis. Clin Orthop Relat Res. 1997;345:60–66.
Stiehl JB, Komistek RD, Dennis DA. Detrimental kinematics of a flat on flat total condylar knee arthroplasty. Clin Orthop Relat Res. 1999;365:139–148.
Stiehl JB, Komistek RD, Dennis DA, Paxson RD, Hoff WA. Fluoroscopic analysis of kinematics after posterior-cruciate-retaining knee arthroplasty. J Bone Joint Surg Br. 1995;77:884–889.
Victor J, Banks S, Bellemans J. Kinematics of posterior cruciate ligament-retaining and -substituting total knee arthroplasty: a prospective randomised outcome study. J Bone Joint Surg Br. 2005;87:646–655.
Victor J, Bellemans J. Physiologic kinematics as a concept for better flexion in TKA. Clin Orthop Relat Res. 2006;452:53–58.
Author information
Authors and Affiliations
Corresponding author
Additional information
One or more of the authors (JV, JB, RDK) have received funding of Smith and Nephew, Memphis, TN.
Each author certifies that his or her institution has approved the reporting of these cases, that all investigations were conducted in conformity with ethical principles of research, and that informed consent for participating in the study was obtained.
This work was performed at AZ Sint-Lucas, Brugge, Belgium; UZ Leuven, Leuven, Belgium; and Knoxville Orthopaedic Clinic, Knoxville, TN, USA.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Victor, J., Mueller, J.K.P., Komistek, R.D. et al. In Vivo Kinematics after a Cruciate-substituting TKA. Clin Orthop Relat Res 468, 807–814 (2010). https://doi.org/10.1007/s11999-009-1072-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11999-009-1072-7