Abstract
The aim of this work was to study the composition of aroma volatile compounds (AVCs) of blood orange cv. Moro treated with γ-aminobutyric acid (GABA), methyl jasmonate (MeJA) or methyl salicylate (MeSA) by headspace gas chromatography-mass spectrometry (HS-GC-MS). GABA at 20 and 40 mM was vacuum infiltrated (at 30 kPa for 8 min), and MeJA or MeSA at 50 and 100 μM (vapour treatments) was applied separately at 20 °C for 18 h. AVCs were measured at harvest and at the end of the experiment (150 days at 3 °C + 2 days at 20 °C, shelf life) for all control and treated fruits. The most abundant compound was limonene, followed by β-myrcene and α-pinene. α-Thujene, camphene, β-pinene, n-decane, α-phellandrene, α-terpinene, p-cymene, (E)-β-ocimene, citronellyl acetate and γ-terpinene were affected by treatments after long-term cold storage. The highest and the lowest percentages of limonene were measured in control samples (94.31 ± 1) and fruits treated with 100 μM MeSA (86.68 ± 1), respectively. (E)-Caryophyllene and (E)-ethyl cinnamate were not detected in all samples after 150 days of cold storage. The multivariate statistical analysis revealed that β-myrcene, n-decane, linalool, limonene, trans-limonene oxide, ethyl hexanoate and sabinene were positively correlated with chilling injury. Overall, it can be concluded that these compounds can be used as biological markers related to CI based on the multivariate statistical analyses.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Blood oranges (Citrus sinensis L. Osbeck) are among the commercial citrus species having anthocyanin pigment that can increase antioxidant activity. They are produced not only for fresh fruit consumption but also for juice and beverages due to being a rich source of bioactive compounds (Habibi et al. 2020).
Taste, aroma, colour and texture are the most important quality attributes of blood oranges. Aroma volatile compounds (AVCs) are secondary metabolites that play an important role in citrus fruit flavour. Citrus fruit comprises numerous volatile compounds, each one with a distinct flavour and aroma (González-Mas et al. 2011). Citrus fruit volatiles are divided into lipid- and water-soluble compounds commonly identified in the peel and in the juice, respectively. It was revealed that juice aroma is a result of the complex combination of 200 distinct volatile compounds, although only 20–40 of those are important contributors of the aroma of orange fruit (Bai et al. 2016). On the other hand, combinations of several odour compounds have interdependent quantitative relationships for orange juice aroma (Selli and Kelebek 2011).
Citrus AVCs have been thoroughly investigated and the research on citrus AVCs has resulted in considerable advancement. Different methods such as headspace gas chromatography-mass spectrometry (HS-GC-MS), aroma extract dilution analysis (AEDA) and headspace solid phase micro-extraction (HS-SPME) have been applied in order to identify AVCs in citrus fruit (Zhang et al. 2017). The headspace (HS) method is a direct sampling technique for AVC analysis and divided into static (vapour-phase extraction) and dynamic (purge and trap). HS is a simple, rapid, solvent-free and sensitive method, which does not change the chemical profile of volatile compounds (Chaudhary et al. 2014).
Volatile compounds of the citrus fruit juice have been identified in mandarins (Tietel et al. 2010; Tietel et al. 2012; Obenland et al. 2013), grapefruit (Biolatto et al. 2005), lemon (Allegrone et al. 2006) and sweet orange (Obenland et al. 2003; Bai et al. 2016). Some dominant compounds include monoterpenes, sesquiterpenes, aldehydes, hydrocarbons, esters, alcohols and ketones (Rambla et al. 2014). Among them, monoterpenes and sesquiterpenes are the major volatile compounds reported in different citrus fruits (Bai et al. 2016). Furthermore, AVCs of citrus fruits are affected by some postharvest treatments such as degreening (Sdiri et al. 2017), wax coating (Tietel et al. 2010) and storage temperatures (Biolatto et al. 2005; Tietel et al. 2012; Obenland et al. 2013; Lado et al. 2019). In addition, rootstock, growing conditions and ripening stage are other factors that can affect the AVC profile (Lado et al. 2019). Moreover, some physiological disorders, such as oleocellosis in the citrus fruit peel, can change the volatile compounds and expression of genes involved in terpenoid metabolism (Xie et al. 2018). Additionally, increased volatile emission of Navel oranges could be used as a predictor of freeze injury (Obenland et al. 2003). On the other hand, storage temperature can also affect the emission of different volatiles. Thus, red grapefruit cv. ‘Star Ruby’ (a cultivar sensitive to chilling injury (CI)) stored at 2 °C emitted not only mainly limonene, but also linalool and α-terpineol, while cyclic sesquiterpenes and aliphatic esters were enhanced at 12 °C (Lado et al. 2019).
Cold storage is a common approach for preserving and extending postharvest life of blood orange fruit. It has been reported that blood orange fruits undergo CI symptoms and changes in bioactive compounds during cold temperatures, as previously reported (Habibi and Ramezanian 2017; Habibi et al. 2019; Habibi et al. 2020). γ-Aminobutyric acid (GABA), methyl jasmonate (MeJA) and methyl salicylate (MeSA) on postharvest application are recognized as elicitor compounds which were found effective on alleviating CI and increasing storability of blood oranges (Habibi et al. 2019). In fact, these compounds were also effective on reducing CI in lemon by enhancing antioxidant enzymes (Siboza et al. 2017). On the other hand, artificially decayed oranges by Penicillium digitatum demonstrated that several primary metabolites and volatile organic compounds (VOCs) were significantly regulated in response to P. digitatum infection (Tang et al. 2018). Postharvest application of MeJA and SA extended cold storage life of ‘Kinnow’ mandarin fruit up to 75 days by reducing weight loss, spoilage and firmness loss, and retarding the activities of fruit-softening enzymes (Baswal et al. 2020).
There is no information available about the effect of postharvest treatment with elicitors and AVCs in blood oranges at chilling temperatures after long-term storage. Up to now, only two research works have been focused on flavour components (Maccarone et al. 1998) and aromatic profile (Selli and Kelebek 2011) in the juice of blood oranges at harvest time. However, this is the first report on AVCs of blood oranges treated with GABA, MeJA and MeSA after long-term cold storage. Therefore, the purpose of this work was to analyse the AVCs in blood orange cv. Moro treated by aforementioned elicitors at chilling temperatures after long-term storage. In addition, this information probably will make evidence about which AVCs can affect by treatments after chilling stress and may be used as marker related to CI based on the multivariate statistical analyses.
Materials and Methods
Fruit Treatments and Storage Conditions
Blood orange cv. ‘Moro’ fruits were harvested from 7-year-old trees according to total soluble solids and total acidity (TSS/TA) ratio from a commercial citrus orchard in Jahrom (28° 30′ N, 87 53° 31′ E), Fars Province, Iran, and were transported to the postharvest laboratory at Shiraz University. Harvested fruits were checked for no defects or peel injuries and selected in uniformity of size. Fruits were disinfected by 2% NaOCl solution and then washed with distilled water. After disinfection, fruits were treated with GABA, MeJA and MeSA. Vacuum infiltration was done by putting the 7 fruits in 2 L of GABA solutions (at 20 and 40 mM) at 30 kPa for 8 min. MeJA and MeSA vapour treatments were applied separately at 50 and 100 μM. These concentrations and duration of treatments were selected according to previous results (Habibi et al. 2019). MeJA and MeSA vapour treatments were performed by placing the fruits in a 20-L plastic container, containing the appropriate volume of solutions to reach the desirable concentration (167 and 233 μL, for 50 and 100 μM, respectively). These volumes were deposited on filter paper at the bottom of the container and then immediately hermetically sealed for 18 h. No treatment was performed on control fruit. Following treatment, control and treated fruits were divided into sets of three replicates of 5 fruits, placed in polyethylene bags and stored for 150 days at 3 °C and 90% relative humidity (RH). At harvest, three lots of five uniform fruits were selected for aroma volatile compound (AVC) analysis.
Chilling Injury (CI) Index
CI symptoms were evaluated by the incidence and severity of pitting and necrotic area on the peel surface. CI index was scored on a 0–3 scale, in which 0 means no injury, 1 slight injury, 2 medium injury and 3 severe injury. CI index was reported using the following formula (Palma et al. 2013):
Aroma Volatile Compound Analysis by HS-GC-MS
Blood orange fruits were sampled at harvest and from cold storage after 150 days and 2 days at 20 °C in order to simulate the shelf life. The fruits were peeled with a knife carefully in order to avoid probable contamination of peel oil into the flesh of fruit. The flesh of five fruits of each replicate of treatment was mixed for identifying and analysing AVCs. The AVCs of blood orange fruits were conducted using static headspace (SHS) analysis on the Combi-PAL system technique (CH-4222 Zwingen, Switzerland) which was equipped with an auto-sampler, heater and agitator. Three grammes of sample was weighed quickly and placed in 20-mL headspace vials including 1 μL of n-heptadecane containing 200 mg L−1 n-hexadecane as internal standard. Vials are immediately wrapped with silicone rubber septa and aluminium caps and then placed to the headspace tray. Volatile compounds were analysed through gas chromatography-mass spectrometry (GC-MS) (Agilent, Model 5975C, USA). GC-MS equipped with a split/splitless injector, an auto-sampler and an Agilent HP5-MS fused silica column (5% phenylmethylpolysiloxane, 30 m × 0.25 mm i.d., film thickness 0.25 μm). Oven temperature program was 60–210 °C at the 121 rate of 4 °C/min, which was then programmed to 240 °C at the rate of 20 °C min−1, and finally 122 held isothermally for 8.5 min. Injector and detector temperatures were 250 and 280 °C, respectively, and nitrogen was used as carrier gas at 1.0 mL min−1. Injections were made in splitting ratio 1:50. The syringe needle was inserted into the headspace vials containing samples. One millilitre of the headspace sample was injected into the GC-MS injector for the analysis of AVCs. Run time was 60 min. The response was calculated as analyte peak area in the GC-MS chromatogram relative to the internal standard (Bahmanzadegan et al. 2017).
Statistical Analysis
The experiment was done according to a completely randomized design (CRD) with three replicates. Data were analysed using one-way analysis of variance (ANOVA) using SAS version 9.4 software for Windows. Least significant difference (LSD) test (P < 0.05) was used for mean comparisons and represented with standard deviation (SD). Principal component analysis (PCA) was done by Minitab version 16 software. Hierarchical cluster analysis (HCA) was conducted using a Ward linkage method with Euclidean distance, and the data was depicted in a dendrogram plot (Almeida et al. 2007). The HCA was implemented using Minitab version 16 software. Data from the correlation matrix was obtained using SAS version 9.4 software and was represented as a heatmap by pheatmap R package.
Results
AVCs
A list of AVCs, retention indices (RI) and chemical class is represented in Table 1. In total, 28 and 26 AVCs were detected by HS-GC-MS in the flesh of blood orange fruit at harvest time and after 150 days of cold storage, respectively (Table 2). AVCs belong to different chemical classes, including aldehyde, alkane hydrocarbon, bicyclic monoterpenes, cyclic monoterpenes, cyclic sesquiterpenes, ester, linear alkane hydrocarbon, monoterpene alcohol aldehydes and monoterpene hydrocarbons (Table 1). The most abundant compound was limonene (85–90%), followed by β-myrcene and α-pinene at harvest time and also after 150 days of cold storage (Table 2).
Volatiles were affected by both treatments and storage (Table 2). Some AVCs including α-thujene, camphene, β-pinene, n-decane, α-phellandrene, α-terpinene, p-cymene, (E)-β-ocimene, citronellyl acetate and γ-terpinene were not affected by treatments after long-term cold storage. In addition, (E)-caryophyllene and (E)-ethyl cinnamate were not detected in control or treated fruit after 150 days of cold storage, although they were identified at harvest time. Some differences were found in AVCs in fruits treated with GABA, MeJA and MeSA after long-term cold storage (Table 2). Limonene was the main AVC in blood orange fruit and increased after 150 days of cold storage. The highest limonene was detected in control samples and the lowest amount was measured in fruits treated with 100 μM MeSA and 50 μM MeJA. However, there was no significant difference between them. In addition, β-myrcene, sabinene, linalool and trans-limonene oxide increased after 150 days in all treatments, and the rate of increase was higher in control than in treated fruit. Valencene, γ-terpinene, α-terpinene and p-mentha-1(7),8-diene decreased in all treated and untreated samples after 150 days of cold storage in comparison with harvest time. On contrary, some AVCs remained constant after 150 days of cold storage including α-thujene, camphene and p-cymene (Table 2).
CI Index
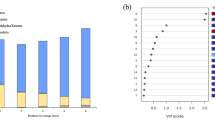
In this study, CI index of blood orange fruit was evaluated in each treatment after 150 days of cold storage (Fig. 1). There was a significant difference among treatments. Control fruit had the highest CI. Generally, fruit treated with 100 μM MeSA had the lowest CI after long-term cold storage (Fig. 1). This reduction was 55% compared to control samples.
Effect of treatments on chilling injury of ‘Moro’ blood orange fruit after 150 days of storage at 3 °C and subsequent 2-day storage at 20 °C. Vertical bars on columns represent ± standard errors (SE) of means. Different letters above the bars on columns indicate a significant difference at P < 0.05 according to LSD test
Hierarchical Clustering Analysis (HCA)
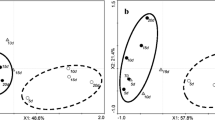
HCA of the AVCs and treatments was shown in Fig. 2. HCA for treatments revealed four main clusters (Fig. 2a). In the first cluster, only the control treatment was included, while the second cluster corresponded to treated fruit with GABA 20 and 40 mM. The third cluster included MeJA 50 μM and MeSA 100 μM treatments, and finally the fourth cluster consisted of MeJA 100 μM and MeSA 50 μM treatments; and therefore, it can be concluded that both treatments are close together (Fig. 2a).
HCA also revealed three main clusters for AVCs (Fig. 2b). Compounds in the first cluster represented the limonene, linalool, sabinene, β-myrcene, trans-limonene oxide, ethyl hexanoate and n-decane (Fig. 2b). Compounds in the second cluster were n-nonane, n-undecane, p-cymene, n-dodecane, n-tridecane, n-tetradecane, citronellyl acetate and γ-terpinene. The third cluster included other identified compounds such as (E)-2-hexenal, valencene, α-phellandrene, p-mentha-1(7),8-diene, terpinolene, α-terpinene, α-thujene, α-pinene, β-pinene and camphene (E)-β-ocimene (Fig. 2b). HCA analysis illustrated that some AVCs increased after long-term storage and some treatments effected these compounds. In addition, the compounds in the first cluster were close to CI index (Fig. 2b).
Heatmap of the Correlation Matrix
Correlation analysis of the AVCs was performed, in order to assess how these compounds were related to CI and also each other. A heatmap of the correlation matrix for AVCs of ‘Moro’ blood orange fruit after 150 days is shown in Fig. 3. Positive and negative correlations are represented in red and blue colours, respectively. CI had a positive correlation with limonene, trans-limonene oxide, linalool, citronellyl acetate, β-myrcene, ethyl hexanoate, n-decane and sabinene. On the contrary, CI had a negative correlation with valencene, α-phellandrene and (E)-2-hexenal (Fig. 3). In this study, some AVCs had a correlation with each other. For example, β-myrcene, ethyl hexanoate and linalool had a negative correlation with (E)-2-hexenal. Limonene and linalool had a positive correlation with sabinene, β-myrcene, ethyl hexanoate and n-decane. Valencene had a negative correlation with limonene, linalool, trans-limonene oxide, β-myrcene and n-decane (Fig. 3).
Principal Component Analysis (PCA)
The multivariate statistical analysis of the measured attributes is presented as a PCA (Fig. 4). PCA was achieved to compare the AVCs listed in Table 1 and CI of blood orange fruit treated with GABA, MeJA and MeSA (Fig. 4). The score plot revealed that the projection on the main first two principal components (PC) namely PC1 and PC2 covered 50.4% and 17.8% of the total variance, respectively (Fig. 4). The score plot demonstrated a clear separation of treatments by PC1 and PC2 and grouped to four quadrants of matrix. Control was separated with other treatments according to PC1 same as GABA 20 mM. GABA 40 mM and MeJA 100 μM were pooled on the left of the plot matrix (Fig. 4). There was a clear and pronounced difference between the AVCs and CI after long-term storage for 150 days. Compounds pointing in the same direction were highly correlated and are mainly described by the first two PCs. Samples with lower CI index representing a clear variance with some AVCs were separated from the control fruit along PC1 (Fig. 4). On the other hand, biplot illustrated that control treatment completely correlated with CI and displayed a significant difference with other treatments. The biplot also showed that β-myrcene, n-decane, linalool, limonene, trans-limonene oxide, ethyl hexanoate and sabinene correlated with CI in control samples (Fig. 4). In the upper right part of the matrix, γ-terpinene, p-cymene, n-tetradecane, n-dodecane, n-tridecane, citronellyl acetate, n-nonane and n-undecane were correlated with GABA 20 mM. Compounds including α-thujene, β-pinene, α-pinene, p-mentha-1(7),8-diene, α-phellandrene, terpinolene, (E)-β-ocimene and camphene correlated with GABA 40 mM and MeJA 100 μM. In addition, MeSA 50 μM and MeSA 100 μM correlated with valencene, (E)-2-hexenal and α-terpinene (Fig. 4).
Discussion
Information about postharvest treatments using elicitors on volatile compounds of citrus fruit at chilling temperature is rare. Our results revealed that AVC content of ‘Moro’ blood orange was affected by treatments at chilling temperature. In this case, some compounds increased, while others decreased or remained constant after 150 days of cold storage. Compounds including α-thujene, camphene and p-cymene remained constant in all treatments after long-term cold storage in comparison with levels found at harvest. On the contrary, limonene, trans-limonene oxide, β-myrcene and α-pinene increased. Degradation and modification of volatile compound precursors are the main mechanisms for their changes during cold storage (Obenland et al. 2003). In our study, (E)-caryophyllene and (E)-ethyl cinnamate were degraded in all samples after 150 days of cold storage. Furthermore, degradation of specific carotenoids probably can drive norisoprenoids and some monoterpenes (Lado et al. 2019).
In citrus fruit, terpenes are the most abundant compounds, with some volatile compounds being common among citrus fruit (Dharmawan et al. 2007). In our study, monoterpenes were the main volatile compounds of blood orange fruit. In ‘Moro’ blood oranges, treatments had a significant effect on limonene content in treated fruit during cold storage. Thus, the lowest and the highest limonene after 150 days of storage was obtained in MeSA 100 μM treated and in control samples, respectively. Limonene also was detected in other sweet orange cultivars such as in ‘Washington navel’ and ‘Valencia late’ (Arena et al. 2006). Previous studies have reported the changes in limonene in different citrus fruits such as grapefruit (Biolatto et al. 2005; Lado et al. 2019) and sweet orange (Obenland et al. 2003) during cold storage.
HS-GC-MS analysis showed that limonene was the major aroma compound which had the highest percentage at harvest time and after cold storage for all control and treated fruits. It has been reported that, among aroma compounds in citrus fruits, γ-terpinene, β-myrcene and α-pinene are comparatively abundant after limonene (Arena et al. 2006). The specific compounds of aroma volatiles can significantly influence the citrus flavour, as has been reported for mandarin (Obenland et al. 2013), grapefruit (Lado et al. 2019), lemon (Allegrone et al. 2006) and sweet orange (Obenland et al. 2003).
Storage of blood orange fruit at low temperature (3 °C) could induce chilling stress (Habibi et al. 2019), which can affect the volatile compounds of fruit (Obenland et al. 2013). In our study, the highest CI was observed in control samples at the end of cold storage. Treatments alleviated CI by some involved mechanisms such as increasing the antioxidant enzyme activities of catalase (CAT), ascorbate peroxidase (APX) and superoxide dismutase (SOD), higher phenylalanine ammonia lyase (PAL) activity, enhancing the proline content and suppressing peroxidase (POD) and polyphenol oxidase (PPO) activities which prevented the accumulation of hydrogen peroxide (H2O2), lipid peroxidation and subsequently increased maintenance of membrane integrity as previously reported (Habibi et al. 2019, 2020).
PCA presented in Fig. 4 showed that the control samples were completely correlated with CI, and also with limonene, linalool, trans-limonene oxide, β-myrcene, n-decane and ethyl hexanoate, citronellyl acetate and sabinene. In addition, the heatmap revealed that CI had a positive correlation with these compounds. For example, emission of volatiles was different (both quantitative and qualitative) in intact red grapefruit when storage temperature changed from 2 to 12 °C, the 2 °C being responsible of the increased content of limonene and associated to CI (Lado et al. 2019). The volatile compounds presented in citrus fruit consist of oil-soluble and water-soluble compounds. Oil-soluble components, such as lipophilic terpenes and terpenoids, were present in the flavedo of citrus fruit (Maccarone et al. 1998). Furthermore, limonene is the major volatile compound in the oil glands of citrus fruit flavedo (Selli and Kelebek 2011). Citrus fruits are sensitive to CI, and the possible mechanism for the increase of limonene could be the occurrence of disruption of oil glands by chilling damage and subsequently limonene leakage to the flesh (Lado et al. 2019). Our previous study demonstrated that electrolyte leakage (EL) increased during cold storage due to CI, and treatments with GABA, MeJA and MeSA alleviated this physiological disorder (Habibi et al. 2019). Therefore, these treatments could prevent limonene leakage due to preserving peel structure. In addition, trans-limonene oxide increased after cold storage with the highest amount in control samples, probably associated with the limonene content under cold stress. Reactions between organic acids and alcohols can help in the formation of ester compounds (Tietel et al. 2010). In our study, this reaction probably is the possible mechanism for increasing ethyl hexanoate and citronellyl acetate as ester compounds after long-term storage at chilling temperature, as has been observed in tomato. In addition, chilling temperature induced changes in aroma volatile profiles of tomato fruit (Farneti et al. 2015, Wang et al. 2016). The concentration of alcoholic compounds can increase as a result of wax coating and long-term cold storage (Tietel et al. 2010). In addition, cold conditions can produce ethanol as a product of fermentation resulting from disruption of aerobic respiration by chilling stress (Obenland et al. 2003). Reactions between organic acids and alcohols can help in the formation of ester compounds (Tietel et al. 2010). In our study, this reaction probably is the possible mechanism for increasing ethyl hexanoate and citronellyl acetate as ester compounds after long-term storage at chilling temperature. In this case, freezing injury increased ethanol content of ‘Washington navel’ orange fruit and ethanol was a precursor for formation of ethyl butanoate and ethyl octanoate as ester compounds (Obenland et al. 2003). In our study, linalool as a monoterpene alcohol aldehyde increased in blood oranges subjected to chilling temperature after long-term storage, since treated fruits resulted in a lower linalool in comparison to control samples, which was in accordance with the PCA and HCA results. In other fruits, icariin maintained postharvest quality of banana by the delayed transformation of starch to α-D-glucose and β-D-glucose, the accelerated accumulation of bioactive compound GABA and the increased production of glutamine and alanine (Yang et al. 2019), while hot air combined with MeJA alleviated chilling injury in loquat fruit, by enhancing the antioxidant enzymes (Jin et al. 2014). In peach, treatment of nitric oxide alleviated CI symptoms by a reduction of malondialdehyde (MDA) content and electrolyte leakage by the increase of the enzyme alternative oxidase (Jiao and Duan 2019).
Conclusion
This is the first report on AVCs of blood orange fruits treated with GABA, MeJA and MeSA exposed to chilling temperature after long-term storage. Monoterpenes were the main compounds of blood orange fruit. These results provided evidence that AVCs could be affected by treatments under chilling temperature. The multivariate statistical analyses revealed that limonene, linalool, β-myrcene, n-decane, trans-limonene oxide, ethyl hexanoate and sabinene correlated with CI. It can be concluded that these compounds can be used as markers related to CI based on the multivariate statistical analyses. However, treated fruits had lower CI, especially in those treated with MeSA 100 μM and MeJA 50 μM. Overall, chilling stress can change some AVCs and elicitors by preventing CI can affect the percentage of AVCs. These vapour treatments had more positive effect than vacuum infiltration of GABA for maintaining reducing CI symptoms which could be related to a delay of the fruit senescence process. Overall, it can be concluded that these compounds (especially limonene) can be used as biological markers related to CI based on the multivariate statistical analyses. In the future, the roles of these treatments in other citrus species and storage in different temperatures deserve further investigations.
References
Allegrone, G., Belliardo, F., & Cabella, P. (2006). Comparison of volatile concentrations in hand-squeezed juices of four different lemon varieties. Journal of Agricultural and Food Chemistry, 54(5), 1844–1848.
Almeida, J. A. S., Barbosa, L. M. S., Pais, A. A. C. C., & Formosinho, S. J. (2007). Improving hierarchical cluster analysis: a new method with outlier detection and automatic clustering. Chemometrics and Intelligent Laboratory Systems, 87(2), 208–217.
Arena, E., Guarrera, N., Campisi, S., & Asmundo, C. N. (2006). Comparison of odour active compounds detected by gas-chromatography–olfactometry between hand-squeezed juices from different orange varieties. Food Chemistry, 98(1), 59–63.
Bahmanzadegan, A., Rowshan, V., & Zareian, F. (2017). Chemical constituents of the essential oil, static headspace analysis of volatile compounds, polyphenolic content and antioxidative capacity of Trigonella elliptica Boiss. grown in Iran. Analytical Chemistry Letters, 7(2), 261–270.
Bai, J., Baldwin, E. A., McCollum, G., Plotto, A., Manthey, J. A., Widmer, W., Luzio, G., & Cameron, R. (2016). Changes in volatile and non-volatile flavor chemicals of “Valencia” orange juice over the harvest seasons. Foods, 5(1), 4.
Baswal, A. K., Dhaliwal, H. S., Singh, Z., Mahajan, B. V. C., & Gill, K. S. (2020). Postharvest application of methyl jasmonate, 1-methylcyclopropene and salicylic acid extends the cold storage life and maintain the quality of ‘Kinnow’ mandarin (Citrus nobilis L. X C. deliciosa L.) fruit. Postharvest Biology and Technology, 161, 111064.
Biolatto, A., Vazquez, D. E., Sancho, A. M., Carduza, F. J., & Pensel, N. A. (2005). Effect of commercial conditioning and cold quarantine storage treatments on fruit quality of “Rouge La Toma” grapefruit (Citrus paradisi Macf.). Postharvest Biology and Technology, 35(2), 167–176.
Chaudhary, P. R., Jayaprakasha, G. K., & Patil, B. S. (2014). Headspace and solid-phase microextraction methods for the identification of volatile flavor compounds in citrus fruits. In Instrumental methods for the analysis and identification of bioactive molecules (pp. 243-256). American Chemical Society.
Dharmawan, J., Kasapis, S., Curran, P., & Johnson, J. R. (2007). Characterization of volatile compounds in selected citrus fruits from Asia. Part I: freshly-squeezed juice. Flavour and Fragrance Journal, 22(3), 228–232.
Farneti, B., Alarcón, A. A., Papasotiriou, F. G., Samudrala, D., Cristescu, S. M., Costa, G., Harren, F. J., & Woltering, E. J. (2015). Chilling-induced changes in aroma volatile profiles in tomato. Food and Bioprocess Technology, 8(7), 1442–1454.
González-Mas, M. C., Rambla, J. L., Alamar, M. C., Gutiérrez, A., & Granell, A. (2011). Comparative analysis of the volatile fraction of fruit juice from different Citrus species. PLoS One, 6(7), e22016.
Habibi, F., & Ramezanian, A. (2017). Vacuum infiltration of putrescine enhances bioactive compounds and maintains quality of blood orange during cold storage. Food Chemistry, 227, 1–8.
Habibi, F., Ramezanian, A., Rahemi, M., Eshghi, S., Guillén, F., Serrano, M., & Valero, D. (2019). Postharvest treatments with γ-aminobutyric acid, methyl jasmonate, or methyl salicylate enhance chilling tolerance of blood orange fruit at prolonged cold storage. Journal of the Science of Food and Agriculture, 99(14), 6408–6417.
Habibi, F., Ramezanian, A., Guillén, F., Serrano, M., & Valero, D. (2020). Blood oranges maintain bioactive compounds and nutritional quality by postharvest treatments with γ-aminobutyric acid, methyl jasmonate or methyl salicylate during cold storage. Food Chemistry, 306, 125634.
Jiao, C., & Duan, Y. (2019). The mediation of NO-enhanced chilling tolerance by GSK-3 in postharvest peach fruit. Food and Bioprocess Technology, 12(12), 2028–2035.
Jin, P., Duan, Y., Wang, L., Wang, J., & Zheng, Y. (2014). Reducing chilling injury of loquat fruit by combined treatment with hot air and methyl jasmonate. Food and Bioprocess Technology, 7(8), 2259–2266.
Lado, J., Gurrea, A., Zacarías, L., & Rodrigo, M. J. (2019). Influence of the storage temperature on volatile emission, carotenoid content and chilling injury development in Star Ruby red grapefruit. Food Chemistry, 295, 72–81.
Maccarone, E., Campisi, S., Fallico, B., Rapisarda, P., & Sgarlata, R. (1998). Flavor components of Italian orange juices. Journal of Agricultural and Food Chemistry, 46(6), 2293–2298.
Obenland, D. M., Aung, L. H., Bridges, D. L., & Mackey, B. E. (2003). Volatile emissions of navel oranges as predictors of freeze damage. Journal of Agricultural and Food Chemistry, 51(11), 3367–3371.
Obenland, D., Collin, S., Sievert, J., & Arpaia, M. L. (2013). Mandarin flavor and aroma volatile composition are strongly influenced by holding temperature. Postharvest Biology and Technology, 82, 6–14.
Palma, A., D’Aquino, S., Vanadia, S., Angioni, A., & Schirra, M. (2013). Cold quarantine responses of ‘Tarocco’ oranges to short hot water and thiabendazole postharvest dip treatments. Postharvest Biology and Technology, 78, 24–33.
Rambla, J. L., Gonzalez-Mas, M. C., Pons, C., Bernet, G. P., Asins, M. J., & Granell, A. (2014). Fruit volatile profiles of two citrus hybrids are dramatically different from those of their parents. Journal of Agricultural and Food Chemistry, 62(46), 11312–11322.
Sdiri, S., Rambla, J. L., Besada, C., Granell, A., & Salvador, A. (2017). Changes in the volatile profile of citrus fruit submitted to postharvest degreening treatment. Postharvest Biology and Technology, 133, 48–56.
Selli, S., & Kelebek, H. (2011). Aromatic profile and odour-activity value of blood orange juices obtained from Moro and Sanguinello (Citrus sinensis L. Osbeck). Industrial Crops and Products, 33(3), 727–733.
Siboza, X. I., Bertling, I., & Odindo, A. O. (2017). Enzymatic antioxidants in response to methyl jasmonate and salicylic acid and their effect on chilling tolerance in lemon fruit [Citrus limon (L.) Burm. F.]. Scientia Horticulturae, 225, 659–667.
Tang, N., Chen, N., Hu, N., Deng, W., Chen, Z., & Li, Z. (2018). Comparative metabolomics and transcriptomic profiling reveal the mechanism of fruit quality deterioration and the resistance of citrus fruit against Penicillium digitatum. Postharvest Biology and Technology, 145, 61–73.
Tietel, Z., Bar, E., Lewinsohn, E., Feldmesser, E., Fallik, E., & Porat, R. (2010). Effects of wax coatings and postharvest storage on sensory quality and aroma volatile composition of ‘Mor’ mandarins. Journal of the Science of Food and Agriculture, 90(6), 995–1007.
Tietel, Z., Lewinsohn, E., Fallik, E., & Porat, R. (2012). Importance of storage temperatures in maintaining flavor and quality of mandarins. Postharvest Biology and Technology, 64(1), 175–182.
Wang, L., Baldwin, E. A., & Bai, J. (2016). Recent advance in aromatic volatile research in tomato fruit: the metabolisms and regulations. Food and Bioprocess Technology, 9(2), 203–216.
Xie, J., Deng, L., Zhou, Y., Yao, S., & Zeng, K. (2018). Analysis of changes in volatile constituents and expression of genes involved in terpenoid metabolism in oleocellosis peel. Food Chemistry, 243, 269–276.
Yang, J., Zhu, H., Tu, J., Jiang, Y., Zeng, J., & Yang, B. (2019). Icariin as a preservative to maintain the fruit quality of banana during postharvest storage. Food and Bioprocess Technology, 12(10), 1766–1775.
Zhang, H., Xie, Y., Liu, C., Chen, S., Hu, S., Xie, Z., Deng, X., & Xu, J. (2017). Comprehensive comparative analysis of volatile compounds in citrus fruits of different species. Food Chemistry, 230, 316–326.
Acknowledgements
We would like to express our sincere thanks to Shiraz University and University Miguel Hernández for research visit opportunity. Also, we thank Fars Agricultural and Natural Resources Research and Education Center, AREEO, Shiraz, Iran, for analysis of aroma volatile compounds of samples.
Funding
The authors received financial support from Shiraz University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Habibi, F., Ramezanian, A., Guillén, F. et al. Effect of Various Postharvest Treatment on Aroma Volatile Compounds of Blood Orange Fruit Exposed to Chilling Temperature After Long-Term Storage. Food Bioprocess Technol 13, 2054–2064 (2020). https://doi.org/10.1007/s11947-020-02547-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-020-02547-1