Abstract
The effect of edible cassava starch–chitosan coatings incorporated with rosemary pepper (Lippia sidoides Cham.) essential oil and pomegranate peel extract on the shelf-life of tomatoes during storage at 25 °C for 12 days was investigated. Sixteen formulations, containing 10 g L−1 cassava starch and various concentrations of chitosan (5, 10, 20, 30 g L−1), essential oil (0, 2.5, 5, 10 mL L−1) and pomegranate peel extract (0, 5, 10, 20 mL L−1) were prepared and applied to tomatoes. Physical–chemical and microbiological analyses were performed on days 1, 4, 8 and 12. Most of the coatings delayed the ripening of tomatoes, lowering the total soluble solids (38−44 g sucrose kg−1) and weight loss (93−128 g kg−1) and maintaining constant firmness compared to the uncoated tomatoes (45 g sucrose kg−1, 175 g kg−1) at 12 days of storage. Conversely, except red intensity (a*), which was higher for the uncoated samples, the colour parameters (L*, b*) of the coated and control tomatoes were similar at the end of storage. Uncoated and coated tomatoes showed no contamination during storage. The coatings showed potential to maintain the quality of tomatoes during storage at 25 °C for 12 days. In this context, tomatoes coated with the formulation comprising 10 g L−1 cassava starch, 10 g L−1 chitosan, 10 mL L−1 essential oil and 20 mL L−1 pomegranate peel extract showed the lowest weight loss and reduced total soluble solids content compared with uncoated ones.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Edible coatings have proven to be highly useful in food preservation, acting as a barrier to gases and water vapour, improving food appearance and controlling microbial contamination (Aider 2010). Chitosan (CH) is of interest as an edible film component, as it is a natural, non-toxic, biocompatible and biodegradable polymer, with antimicrobial activity and excellent oxygen barrier properties (Elsabee and Abdou 2013). One of the most important polysaccharides used in the formulation of biodegradable films and edible coatings is starch. Cassava starch (CS) is particularly useful in coatings as it is tasteless, odourless and transparent, thereby retaining the original taste, aroma and appearance of the product (Garcia et al. 2010; Chiumarelli and Hubinger 2012; Chiumarelli and Hubinger 2014). Besides, this product is cheap and abundant in Brazil, for this reason, it was considered the use of cassava starch as one of components of edible coatings in this work. In this context, several researchers have developed coatings or films based on cassava starch and chitosan blends (Azevedo et al. 2014; Aquino et al. 2015; Frazão et al. 2017; Valencia-Sullca et al. 2018; Zhao et al. 2018).
The tomato (Solanum lycopersicon L.) is a climacteric fruit, consumed mainly in a natural or processed form (in sauces, purees, ketchup and others). However, this fruit has a relatively short postharvest life due to several factors, such as transpiration, postharvest diseases and increased ripening and senescence, which accelerate the deterioration during storage and transport, causing a serious problem for commercialisation (Ali et al. 2010; Nawab et al. 2017). Moreover, in tropical regions, where the temperature is high, tomatoes show rapid ripening and deterioration, due to the increased respiration of the fruit (Bailén et al. 2006). Consequently, numerous researchers have developed various edible coating formulations to extend the shelf-life of tomatoes (Mehyar et al. 2011; Ramos-García et al. 2012; Das et al. 2013; Fagundes et al. 2014; Fagundes et al. 2015; Guerra et al. 2015; Rodriguez-Garcia et al. 2016; Nawab et al. 2017).
Bioactive compounds, such as plant extracts, are increasingly being incorporated in edible coatings to further extend the shelf-life and facilitate the processing and consumption of food (Sung et al. 2013; Azevedo et al. 2014; He et al. 2014; Aquino et al. 2015; Frazão et al. 2017; Wang et al. 2017). Among compounds with potential use in edible coatings, the essential oil (EO) from rosemary peeper (Lippia sidoides Cham.) has shown high pharmacological and commercial value, attributed to the presence of thymol and carvacrol monoterpenes, as major constituents in the leaves. These compounds are responsible for insecticidal, fungicidal, leishmanicidal, larvicidal, acaricidal and anti-inflammatory activities (Cavalcanti et al. 2010; Veras et al. 2012; Lima et al. 2013). Thymol and carvacrol have been accepted as flavouring agents in Europe, as they are not considered to present any health risk to consumers at the estimated intake level (Hyldgaard et al. 2012). However, the incorporation of EOs in active food packaging requires additional research to establish the effective and safe concentrations of these ingredients (Seow et al. 2014).
Another useful source of plant bioactives is pomegranate peel extracts (PPEs), which have been highlighted as potent, natural inhibitors of several bacteria (e.g. Bacillus subtilis, Bacillus coagulans, Bacillus cereus, Escherichia coli, Klebsiella pneumoniae) and fungi (e.g. Aspergillus niger, Mucor indicus, Penicillium citrinum, Rhizopus oryzae, Trichoderma reesei, Rhizopus stolonifer, Botrytis cinerea) (Dahham et al. 2010; Tehranifar et al. 2011; Akhtar et al. 2015; Nicosia et al. 2016). Pomegranate extracts have been applied to meat surfaces, chicken breasts and fish, and have also been implicated as antibacterial therapeutics to ameliorate diseases and topical infections, such as those in the oral cavity or on the skin surface (Howell and D’Souza 2013; Tanveer et al. 2015). The extracts of pomegranate peels have demonstrated wound healing properties, immunomodulatory activity, antibacterial activity and antiatherosclerotic and antioxidative capacities. Although PPE has been used in CH- and alginate-based coatings, to extend the postharvest quality of guava (Nair et al. 2017), there are currently no reports about the effects of PPE incorporated in active packagings on consumer health (Goula and Lazarides 2015).
In this context, the current work evaluated the effect of edible CS-CH coatings enriched with L. sidoides EO and PPE on the physical–chemical and microbiological quality of tomatoes stored at 25 °C for 12 days.
Materials and Methods
Materials
L. sidoides leaves, collected at Mossoró county, Rio Grande do Norte, Brazil, were cultivated in the Research Farm of the Federal University of Sergipe, Department of Agronomical Engineering, São Cristóvão, Brazil. For extraction of EO, the leaves were pre-dried at 40 °C in a forced air oven (Marconi MA 037, Piracicaba, São Paulo, Brazil) for 48 h and pulverised using a mill (Grindomix GM 200, Haan, Germany) (Cavalcanti et al. 2010). The EO (major components, thymol (0.7 mL L−1) and p-cymene (0.1 mL L−1)) was obtained by hydrodistillation using Clevenger equipment and donated by the Department of Agronomical Engineering of Federal University of Sergipe, São Cristóvão, Brazil. Powdered CH (80 mesh particle size, 859 g kg−1 deacetylation degree, pH 8.2) was obtained from Polymar (Fortaleza, Brazil), and CS (Dinha Bá brand) (850 g kg−1 carbohydrates, 0.45 g kg−1 calcium and 0.03 g kg−1 iron) was purchased from “Cachoeira Alimentos e Bebidas Ltda” company (São Cristóvão, Sergipe, Brazil). Italian tomatoes, without peduncles and sepals (tomatoes designated as the Italian group cultivated in São Paulo and Minas Gerais, Brazil, characterised by elongated fruits of 7 to 10 cm, with a transverse diameter of 3 to 5 cm, intense red colour, bilocular, thick wall and sweet taste), were purchased from Antônio Franco market of Aracaju (Brazil) and transferred to the laboratory in sterile boxes at room temperature within 1 h. The number of tomatoes used for each formulation was purchased separately, and the experiments were started on the same day. The mature fruits (with green traces) were selected for size uniformity, without visible physical damage and microbial deterioration.
PPE Preparation
The fruit was sanitised by immersion in 0.2 g L−1 chlorinated solution for 15 min. The peels were then manually separated from the pulp and dried at 50 °C for 24 ± 2 h before they were triturated and stored in sterile glass bottles, until use. To obtain the PPE, the peels (5 g) were mixed with 10 mL methanol (600 mL L−1) under agitation (Solab SL222 orbital shaker, Brazil) at 250 rpm/45 °C for 35 min, based on Gullon et al. (2016), with modifications.
Preparation of Edible Coatings
Sixteen formulations of edible coatings were prepared, as described by Azevedo et al. (2014), adapted from Vasconez et al. (2009), using CS (10 g L−1) and various concentrations of CH (5, 10, 20, 30 g L−1), L. sidoides EO (0, 2.5, 5, 10 mL L−1) and PPE (0, 5, 10, 20 mL L−1) (Table 1). CH was dissolved (w/v) in acetic acid solution (0.26 mol L−1) at 25 °C, and glycerol (12.8 g L−1) was then added, to complete the volume to 100 mL. A CS solution (100 mL) was prepared in glycerol (6.4 g L−1). The starch solution was stirred in a heated water bath not exceeding 70 °C. After cooling to 25 °C, the solution was added to the CH solution and stirred until it was completely homogeneous. Then, the PPE and L. sidoides EO were added to the mixture. All experiments were performed in triplicate.
Application of Edible Coatings to Tomatoes
The tomatoes were sanitised by immersion in sodium hypochlorite solution (0.2 g L−1) for 15 min, rinsed with water and dried at room temperature on plastic trays. Then, groups of 10 tomatoes were immersed in each formulation, dried at room temperature for 30 min (all tomatoes showed visually uniform texture) and stored on aluminium trays in biochemical oxygen demand chambers (SP Labor, Brazil) with internal forced air circulation by micro-ventilators, at 25 °C and 86−89% relative humidity for 1, 4, 8 and 12 days (i.e. 10 tomatoes per day of storage for each formulation, totalling 40 tomatoes per formulation). A total of 640 tomatoes for 16 formulations, plus 40 uncoated tomatoes, were used. The physical–chemical and microbiological analyses were performed at 1, 4, 8 and 12 days of storage, for each applied formulation. Uncoated tomatoes were used as the control.
pH, Titratable Acidity and Total Soluble Solids Analyses
Five grams of each fruit was homogenised with 50 mL distilled water and the pH value recorded (HI 9219 pH meter, Hanna Instruments, Woonsocket, RI, USA). The titratable acidity (TA) was measured using 5 g of each tomato macerated and homogenised with 50 mL distilled water, with 0.1 N NaOH as the titrant, and the results expressed as g citric acid kg−1, as described by Bassetto et al. (2005), with modifications. The total soluble solids (TSS) was determined on 5 g of macerated fruit sample, using a bench refractometer and the results were expressed as g sucrose kg−1 (Guerra et al. 2015). The measurements were taken from two tomatoes at 1, 4, 8 and 12 days of storage at 25 °C.
Weight Loss
For determining the weight loss, the fruit was weighed at 0, 1, 4, 8 and 12 days of storage (10 fruit per interval) on a balance with 0.001 g accuracy (Radwag brand, Poland). The total weight loss was calculated as a percentage of the initial weight (Ali et al. 2010).
Tomato Firmness
The firmness of uncoated and coated tomatoes was analysed using a Brookfield CT3 Texture Analyser (Middleboro, USA), equipped with a 2-mm diameter probe, and maximum load cell of 25 kg. The central region (three points) of each tomato (three fruit per interval) was compressed at a rate of 1 mm s−1, using a 4.0 to 8.6 N load cell. Firmness (N) was defined as the maximum force required to penetrate to a depth of 10 mm. The analysis was performed at 1, 4, 8 and 12 days of storage (Aquino et al. 2015).
Colour Parameters
Superficial colour alterations were monitored using a CR-400 Minolta colourimeter (Osaka, Japan) on three tomatoes per interval of storage. The International Commission on Illumination (CIE 1986) colour parameters, a* (redness), b* (yellowness) and L* (luminosity), were directly recorded on the surface of the tomatoes from three regions, at 1, 4, 8 and 12 days of storage at 25 °C.
Microbiological Analyses
For microbiological analyses, tomatoes stored at 1, 4, 8 and 12 days were placed in a sterile plastic bag (five fruit per interval) with saline solution (8.5 g L−1) (corresponding to a 10−1 dilution) and homogenised. Then, successive dilutions (10−2 and 10−3) were obtained. The yeasts and moulds were enumerated, by the surface-seeding technique with potato dextrose agar and incubation at 35 °C for 5 days. The results were expressed as colony-forming units (CFU) per gram, as detailed by Rojas-Grau et al. (2007), with modifications. The most probable number (MPN) of thermotolerant coliforms was determined as described in the Bacteriological Analytical Manual (FDA 2010). First, tubes containing lauryl sulphate tryptose (LST) broth were incubated at 35 °C for 48 h in a bacteriological incubator, for a presumptive test. Then, tubes that exhibited gas production in the LST broth were transferred to tubes containing E. coli broth and incubated at 44.5 °C for 48 h (confirmatory test). The presence of thermotolerant coliforms was verified in tubes that exhibited growth with gas production in E. coli broth. The MPN of thermotolerant coliforms was calculated with the use of the Hoskins table (Vanderzant and Splittstoesser 2001).
Statistical Analysis
The composition of edible coatings varied, according to a completely randomised design. All data were analysed using Assistat 7.7 beta software. The analyses were performed using descriptive statistics (mean and standard deviation). The differences among means were determined by Duncan’s range test at a 5% significance level.
Results and Discussion
Effect of Edible Coatings on pH, TA, TSS and Weight Loss of Tomatoes
The pH, weight loss, TA and TSS content of uncoated and coated tomatoes were determined during the storage at 25 °C for 1, 4, 8 and 12 days. Citric acid is the most abundant acid in tomatoes and the largest contributor to the TA (90 g citric acid kg−1). During maturation, there is an initial increase in the citric acid content, followed by a decrease over time until full maturity (Ali et al. 2010). Thus, an increased pH and decreased TA in tomatoes can be attributed to the decline in, primarily, citric acid concentration, as the fruit ripens (Das et al. 2013). The majority of the coated tomatoes (pH 4.62−5.77) showed a slight pH increase, with a significant difference (p < 0.05) and no difference (p > 0.05) in TA on days 1 and 12 of storage, respectively. Only tomatoes coated with formulations 6 (10 g L−1 CS, 10 g L−1 CH, 2.5 mL L−1 EO, 5 mL L−1 PPE), 7 (10 g L−1 CS, 10 g L−1 CH, 5 mL L−1 EO, 10 mL L−1 PPE), 10 (10 g L−1 CS, 20 g L−1 CH, 2.5 mL L−1 EO, 5 mL L−1 PPE), 11 (10 g L−1 CS, 20 g L−1 CH, 5 mL L−1 EO, 10 mL L−1 PPE) and 16 (10 g L−1 CS, 30 g L−1 CH, 10 mL L−1 EO, 20 mL L−1 PPE), as well as the uncoated tomatoes, showed a significant difference (p < 0.05) in TA from 1 to 12 days of storage, with reductions of 47, 33, 32, 55, 15 and 29 g citric acid kg−1, respectively, at the end of storage, meaning they exhibited faster ripening than the others. However, comparing coated with uncoated tomatoes, there was no significant difference (p > 0.05) in TA on day 12 of storage, with the values maintained at around 30 g citric acid kg−1 (Table 1), except tomatoes coated with formulations 1, 6, 7, 10 and 11.
The TSS content of various tomato varieties commonly ranges from 40 to 80 g sucrose kg−1 (Cramer et al. 2001). According to Tigist et al. (2013), an initial increase of TSS content during maturation of fresh market tomato varieties, followed by a decrease over time, can be attributed to a gradual decline in the rate of hydrolysis of carbohydrates at the end of storage. In the present work, the TSS content ranged from 31 to 42 g sucrose kg−1, and the coated tomatoes tended to show a slight but significant difference (p < 0.05) in TSS, throughout storage (Table 2). The TSS of coated and uncoated tomatoes differed statistically (p < 0.05) at 12 days of storage, with the uncoated fruit displaying the higher TSS content (45 g sucrose kg−1), implying a relatively faster ripening.
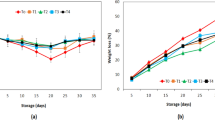
The weight loss primarily results from transpiration and the loss of carbon atoms from fruit, in each cycle of respiration (Das et al. 2013). Most of the coated tomatoes showed a weight loss between 105 and 128 g kg−1 on day 12 of storage, without significant differences (p > 0.05) among them (Table 2). The highest weight loss (175 g kg−1) was recorded for the uncoated tomatoes, and the lowest (93 and 94 g kg−1) corresponded to those coated with formulation 8 (10 g L−1 CS, 10 g L−1 CH, 5 mL L−1 EO, 10 mL L−1 PPE) and 11 (20 g L−1 CH, 5 mL L−1 EO, 10 mL L−1 PPE), at 12 days of storage. The weight losses were 4.2 to 7.7% less for coated tomatoes than those for uncoated ones (at 12 days of storage), demonstrating the efficacy of the coatings as moisture barriers. When stored at 24 °C for 20 days, Das et al. (2013) obtained a weight loss of 3.53% less for tomatoes coated with rice starch-based edible coating containing coconut oil and tea leaf extract than the control. For the same duration at 20 °C, Ali et al. (2010) detected 2% less weight loss for tomatoes coated with 10% gum arabic compared to the control. In comparison, the results in the present work show greater efficiency in reducing the weight loss of tomatoes during storage.
Most of the coated tomatoes maintained a constant firmness (p > 0.05) between 1 and 12 days of storage (Table 2). However, a higher firmness loss (77%) was noted in tomatoes coated with 10 g L−1 CS, 30 g L−1 CH, 2.5 mL L−1 EO and 5 mL L−1 PPE (formulation 14) than the others, while uncoated tomatoes showed a 22% loss of firmness. Probably, the high percentage of CH in the edible coatings contributed to the softening of the tomatoes. Ali et al. (2010) mentioned the softening of fruit during maturation reflects changes in the structure/composition of the cell wall and intracellular components, involving the enzymatic hydrolysis of pectin and starch.
In this work, the edible coatings contributed to a lower weight loss and firmness but maintained the pH and TSS of the tomatoes compared to the uncoated fruit. In contrast, Guerra et al. (2015) obtained no differences in TA, TSS and firmness among cherry tomato fruit treated with coatings comprising CH and Mentha piperita L. or Mentha × villosa than the uncoated fruit, at 12 or 25 °C during 12 days of storage. Fagundes et al. (2014) also verified that a coating application (edible composite coatings based on hydroxypropyl methylcellulose (HPMC), beeswax and food preservatives) did not significantly affect the TA, pH and firmness of cherry tomatoes stored at 5 °C for 15 days, followed by 20 °C for 5 days. Dávila-Aviña et al. (2011) noticed tomatoes coated with carnauba and mineral oil edible coatings had a relatively constant TA and TSS content, but reduced weight loss and firmness. Ali et al. (2010) coated tomatoes with various concentrations of aqueous arabic gum and documented a delay in changes of weight, firmness, TA and TSS compared to the uncoated control fruit. Das et al. (2013) detected a reduction in weight loss and TA, but an increase in TSS and a slight increase in pH of tomatoes coated with a rice starch-based edible film containing coconut oil and tea leaf extract. The same authors concluded that the coated fruit exhibited delayed ripening effects compared to the uncoated controls.
Most of the coated tomatoes maintained a constant pH and TA and lower TSS content, weight loss and firmness loss than uncoated ones at day 12 of storage, characterising a delay in fruit ripening.
Effect of Edible Coatings on Colour Parameters (L*, a*, b*) of Tomatoes
The colour is an important quality parameter of tomato fruit, with redness predominant, being indicative of lycopene content, followed by carotenes (yellow to orange) and xanthophylls (yellow) (López et al. 2001; Ali et al. 2010). In contrast to the tomatoes coated with formulations 13−16, which contained 30 g L−1 CH and showed a constant luminosity, a reduced brightness was observed in the uncoated fruit and remaining coated fruit, which exhibited a significant difference between 1 and 12 days of storage (p < 0.05) (Table 3). Thus, the tomatoes coated with formulations 13−16 displayed higher luminosity values than the control samples. Probably, the higher percentage of CH contributed to these results because a higher luminosity has previously been described for CH-coated fruits, which could be associated with the luminosity and high transparency of CH films (Oliveira et al. 2014).
Coated and uncoated tomatoes recorded increased a* values during storage, with a significant difference (p < 0.05) between days 1 and 12 while the b* values remained predominantly constant, with no significant differences among the samples (p > 0.05) (Table 3). The uncoated tomatoes had higher a* values than those coated with formulations 1 (10 g L−1 CS, 5 g L−1 CH), 2 (10 g L−1 CS, 5 g L−1 CH, 2.5 mL L−1 EO, 5 mL L−1 PPE), 5 (10 g L−1 CS, 10 g L−1 CH), 7 (10 g L−1 CS, 10 g L−1 CH, 5 mL L−1 EO, 10 mL L−1 PPE) and 16 (10 g L−1 CS, 30 g L−1 CH, 10 mL L−1 EO, 20 mL L−1 PPE), which showed no significant difference (p > 0.05) on the last day of storage. The results meant that these coatings were more efficacious at delaying the ripening of tomatoes. Overall, the colour parameters indicated that the applied coatings delayed ripening of the fruit relative to the control samples. Dávila-Aviña et al. (2011) also verified that mineral oil coatings delayed colour changes in tomatoes at the end of storage. Fagundes et al. (2014) stated that HPMC-beeswax edible coatings did not alter the colour parameters of cold-stored cherry tomatoes. Guerra et al. (2015) stated that the application of coatings comprising CH and M. piperita L. or Mentha × villosa Huds. EO presented no adverse effect on lycopene formation in red cherry tomatoes. Das et al. (2013) delayed the colour (L*, a* b*) change in tomatoes during 20-day storage, by coating the fruit with a film containing starch, glycerol, coconut oil and tea leaf extract. Ali et al. (2010) observed that uncoated tomatoes turned from green to red within 4–8 days of storage while fruits coated with 50 and 100 g L−1 gum arabic were red at 12–16 days, and those coated with 150 and 200 g L−1 gum arabic remained green, even at 20 days of storage.
The colour parameters of the majority of the coated tomatoes showed similar behaviour to the uncoated fruit, revealing decreased L*, constant b* and increased a* values during storage.
Effect of Edible Coatings on Microbiological Quality of Tomatoes
The microbiological quality of uncoated and coated tomatoes was evaluated during storage at 25 °C for 12 days. The mould and yeast counts were typically maintained at < 10 CFU g−1 for the coated and uncoated tomatoes until day 12 of storage, except for formulations 1, 3 and 9 (Table 4). Overall, all samples remained within the mould and yeast count limit of 1.0 × 106 CFU g−1, considered acceptable for fruit-based products (Bierhals et al. 2011). On the first and last day of storage, the thermotolerant coliforms were present at 3.0 MPN g−1 for uncoated and coated tomatoes. These results comply with the Brazilian legislation (RDC Resolution No. 12; Brasil 2001), where fresh fruit prepared ‘in nature’ (peeled, selected or fractionated), sanitised, refrigerated or frozen for direct consumption should contain a maximum of 5.0 × 102 MPN g−1.
Few authors have studied the microbiological quality of tomatoes. Among them, Das et al. (2013) affirmed that a film containing starch, glycerol, coconut oil and tea leaf extract acted as an antimicrobial barrier, reducing the number of microorganisms in tomatoes during storage for 20 days. Fagundes et al. (2015) established that edible coatings based on HPMC, beeswax and sodium benzoate did not prevent the appearance of the spores of Alternaria alternate in the artificially inoculated fruit but reduced the incidence and severity of the fungus on cherry tomatoes stored at 5 °C for 21 days, followed by 20 °C for 4 days. Finally, Guerra et al. (2015) demonstrated that coatings comprising CH and M. piperita L. or Mentha × villosa Huds. delayed the growth of decay-causing A. niger, B. cinerea, Penicillium expansum and R. stolonifer in artificially contaminated tomato fruit during storage at either room temperature or low temperature.
The coatings did not influence the microbiological quality, given that both uncoated and coated tomatoes maintained the mould and yeast counts below the limit recommended by Brazilian legislation, until day 12 of storage.
Conclusions
In this study, edible coatings based on CS and CH were enriched with L. sidoides EO and PPE, for the first time. The majority of the analysed physical–chemical parameters were not significantly different between the coated and uncoated tomatoes at day 12 of storage. However, the coatings lowered the weight loss and TSS content, demonstrating their effectiveness in delaying fruit ripening. In this context, the tomatoes coated with 10 g L−1 CS, 10 g L−1 CH, 10 mL L−1 EO and 20 mL L−1 PPE (formulation 8) showed the best results (lowest weight loss and reduced TSS content). Considering the perishability of Italian tomatoes, the findings proved that these coatings effectively delayed ripening and maintained the physical–chemical quality of the fruit during 12 days of storage at 25 °C. The coatings have a potential to be used in future applications as bioactive packaging. However, further studies are needed to evaluate the sensorial parameters of coated tomatoes and the safety of L. sidoides EO and PPE for human consumption.
References
Aider, M. (2010). Chitosan application for active bio-based films production and potential in the food industry: review. LWT-Food Science and Technology, 43(6), 837–842.
Akhtar, S., Ismail, T., Fraternale, D., & Sestili, P. (2015). Pomegranate peel and peel extracts: chemistry and food features. Food Chemistry, 174, 417–425.
Ali, A., Maqbool, M., Ramachandran, S., & Alderson, P. G. (2010). Gum Arabic as a novel edible coating for enhancing shelf-life and improving postharvest quality of tomato (Solanum lycopersicum L.) fruit. Postharvest Biology and Technology, 58(1), 42–47.
Aquino, A. B., Blank, A. F., & Santana, & L.C.L.A. (2015). Impact of edible chitosan-cassava starch coatings enriched with Lippia gracilis Schauer genotype mixtures on the shelf life of guavas (Psidium guajava L.) during storage at room temperature. Food Chemistry, 171, 108–116.
Azevedo, A. N., Buarque, P. R., Cruz, E. M. O., Blank, A. F., Alves, P. B., Nunes, M. L., & Santana, L. C. L. A. (2014). Response surface methodology for optimisation of edible chitosan coating formulations incorporating essential oil against several foodborne pathogenic bacteria. Food Control, 43, 1–9.
Bailén, G., Guillén, F., Castillo, S., Serrano, M., Valero, D., & Martínez-Remero, D. (2006). Use of activated carbon inside modified atmosphere packages to maintain tomato fruit quality during cold storage. Journal of Agricultural and Food Chemistry, 54(6), 2229–2235.
Bassetto, E., Jacomino, A. P., Pinheiro, A. L., & Kluge, R. A. (2005). Delay of ripening of ‘Pedro Sato’ guava with 1-methylcyclopropene. Postharvest Biology and Technology, 35(3), 303–308.
Bierhals, V. S., Chiumarelli, M., & Hubinger, M. D. (2011). Effect of cassava starch coating on quality and shelf life of fresh-cut pineapple (Ananas Comosus L. Merril cv “Perola”). Journal of Food Science, 76, 62–72.
Brasil, Anvisa. Resolução RDC no. 12. (de 02 de Janeiro de 2001) Estabelece Padrões Microbiológicos para Alimentos. Diário Oficial da Republica Federativa do Brasil.
Cavalcanti, S. C. H., Niculau, E. S., Blank, A. F., Câmara, C. A. G., Araújo, I. N., & Alves, P. B. (2010). Composition and acaricidal activity of Lippia sidoides essential oil against two-spotted spider mite (Tetranychus urticae Koch). Bioresource Technology, 101(2), 829–832.
Chiumarelli, M., & Hubinger, M. D. (2012). Stability, solubility, mechanical and barrier properties of cassava starch-Carnauba wax edible coatings to preserve fresh-cut apples. Food Hydrocolloids, 28(1), 59–67.
Chiumarelli, M., & Hubinger, M. D. (2014). Evaluation of edible films and coatings formulated with cassava starch, glycerol, carnauba wax and stearic acid. Food Hydrocolloids, 38, 20–27.
CIE (1986). Colorimetrie, 2nd ed. Comission Internationale de L’Éclairage, Viena, Publication CIE 15.2.
Cramer, M. D., Oberholzer, J. A., & Combrink, N. J. (2001). The effect of supplementation of root zone dissolved inorganic carbon on fruit yield and quality of tomatoes (cv ‘Daniela’) grown with salinity. Scientia Horticulturae, 89(4), 269–289.
Dahham, S. S., Ali, M. N., Tabassum, H., & Khan, M. (2010). Studies on antibacterial and antifungal activity of pomegranate (Punica granatum L.). American-Eurasian Journal Agricultural & Environmental Sciences, 9(3), 273–281.
Das, D. K., Dutta, H., & Mahanta, C. L. (2013). Development of a rice starch-based coating with antioxidant and microbe-barrier properties and study of its effect on tomatoes stored at room temperature. LWT – Food Science and Technology, 50(1), 272–278.
Dávila-Aviña, J. E. J., Villa-Rodriguez, J., Cruz-Valenzuela, R., Rodriguez-Armenta, M., Espino-Díaz, M., Ayala-Zavala, J. F., Olivas-Orozco, G. I., Heredia, B., & González- Aguilar, G. (2011). Effect of edible coatings, storage time and maturity stage on overall quality of tomato fruits. American Journal of Agricultural and Biological Sciences, 6(1), 162–171.
Elsabee, M. Z., & Abdou, E. S. (2013). Chitosan based edible films and coatings: a review. Materials Science and Engineering C, 33(4), 1819–1841.
Fagundes, C., Palou, L., Monteiro, A. R., & Pérez-Gago, M. B. (2014). Effect of antifungal hydroxy methylcellulose-beeswax edible coatings on gray mold development and quality attributes of cold-stored cherry tomato fruit. Postharvest Biology and Technology, 92, 1–8.
Fagundes, C., Palou, L., Monteiro, A. R., & Pérez-Gago, M. B. (2015). Hydroxypropyl methylcellulose-beeswax edible coatings formulated with antifungal food additives to reduce Alternaria black spot and maintain postharvest quality of cold-stored cherry tomatoes. Scientia Horticulturae, 193, 249–257.
FDA (U.S. Food and Drug Administration) (2010) Bacteriological analytical manual online. Available from: https://www.fda.gov/food/foodscienceresearch/laboratorymethods/ucm2006949.htm Accessed 20.01.18.
Frazão, G. G. S., Blank, A. F., & Santana, L. C. L. A. (2017). Optimisation of edible chitosan coatings formulations incorporating Myrcia ovata Cambessedes essential oil with antimicrobial potential against foodborne bactéria and natural microflora of mangaba fruits. LWT – Food Science and Technology, 79, 1–10.
Garcia, L. C., Pereira, L. M. R., Sarantópoulos, C. I. G. L., & Hubinger, M. D. (2010). Selection of an edible starch coating for minimally processed strawberry. Food Bioprocess & Technology, 3(6), 834–842.
Goula, A. M., & Lazarides, H. N. (2015). Integrated processes can turn industrial food waste into valuable food by-products and/or ingredients: the cases of olive mill and pomegranate wastes. Journal of Food Engineering, 167, 45–50.
Guerra, I. C. D., Oliveira, P. D. L., Pontes, A. L. S., Lúcio, A. S. S. C., Tavares, J. F., Barbosa-Filho, J. M., Madruga, M. S., & Souza, E. L. (2015). Coatings comprising chitosan and Mentha piperita L. or Mentha × villosa Huds essential oils to prevent common postharvest mold infections and maintain the quality of cherry tomato fruit. International Journal of Food Microbiology, 214, 168–178.
Gullon, B., Pintado, M. E., Perez-Alvarez, J. A., & Viuda-Martos, M. (2016). Assessment of polyphenolic profile and antibacterial activity of pomegranate peel (Punica granatum) flour obtained from co-product of juice extraction. Food Control, 59, 94–98.
He, S., Yang, Q., Ren, X., Zi, J., Lu, S., Wang, S., Zang, Y., & Wang, Y. (2014). Antimicrobial efficiency of chitosan solutions and coatings incorporated with clove oil and/or ethylenediaminetetraacetate. Journal of Food Safety, 34(4), 345–352.
Howell, A.B. & D'Souza, D.H. (2013) The Pomegranate: Effects on Bacteria and Viruses That Influence Human Health. Evidence-Based Complementary and Alternative Medicine, 2013, 1–11.
Hyldgaard, M., Mygind, T., & Meyer, R. L. (2012). Essential oils in food preservation: mode of action, synergies, and interaction with food matrix components. Frontiers in Microbiology, 3, 1–24.
Lima, J. K. A., Albuquerque, E. L. D., Santos, A. C. C., Oliveira, A. P., Araújo, A. P. A., Blank, A. F., Arrigoni-Blank, M. F., Alves, P. B., Santos, D. A., & Bacci, L. (2013). Biotoxicity of some plant essential oils against the termite Nasutitermes corniger (Isoptera:Termitidae). Industrial Crops and Products, 47, 246–251.
López, J., Ruiz, R. B., Ballesteros, R., Ciruelos, A., & Ortiz, R. (2001). Effect of edible coatings: storage time and maturity stage on overall quality of tomato fruits color and lycopene content of several commercial tomato varieties at different harvesting dates. Acta Horticulture, 542, 243–245.
Mehyar, G. F., Al-Qadiri, H. M., Abu-Blan, H. A., & Swanson, B. G. (2011). Antifungal effectiveness of potassium sorbate incorporated in edible coatings against spoilage molds of apples, cucumbers, and tomatoes during refrigerated storage. Journal of Food Science, 76(3), 210–217.
Nair, M. S., Saxena, A., & Kaur, C. (2017). Effect of chitosan and alginate based coatings enriched with pomegranate peel extract to extend the postharvest quality of guava (Psidium guajava L.). Food Chemistry, 240, 245–252.
Nawab, A., Alam, F., & Hasnain, A. (2017). Mango kernel starch as a novel edible coating for enhancing shelf- life of tomato (Solanum lycopersicum) fruit. International Journal of Biological Macromolecules, 103, 581–586.
Nicosia, M. G. L. D., Pangallo, S., Raphael, G., Romeo, F. V., Strano, M. C., Rapisarda, P., Droby, S., & Schena, L. (2016). Control of postharvest fungal rots on citrus fruit and sweet cherries using a pomegranate peel extract. Postharvest Biology and Technology, 114, 54–61.
Oliveira, C. E. V., Magnani, M., Sales, C. V., Pontes, A. L. S., Campos-Takaki, G. M., Stamford, T. C. M., & Souza, E. L. (2014). Effects of chitosan from Cunninghamella elegans on virulence of post-harvest pathogenic fungi in table grapes (Vitis labrusca L.). International Journal of Food Microbiology, 171, 54–61.
Ramos-García, M., Bosquez-Molina, E., Hernández-Romano, J., Zavala-Padilla, G., Terrés-Rojas, E., Alia-Tejacal, I., Barrera-Necha, L., Hernández-López, M., & Bautista-Baños, S. (2012). Use of chitosan-based edible coatings in combination with other natural compounds, to control Rhizopus stolonifer and Escherichia coli DH5a in fresh tomatoes. Crop Protection, 38, 1–6.
Rodriguez-Garcia, I., Cruz-Valenzuela, M. R., Silva-Espinoza, B. A., Gonzalez-Aguilar, G. A., Moctezuma, E., Gutierrez-Pacheco, M. M., Tapia-Rodriguez, M. R., Ortega-Ramirez, L. A., & Ayala-Zavala, J. F. (2016). Oregano (Lippia graveolens) essential oil added within pectin edible coatings prevents fungal decay and increases the antioxidant capacity of treated tomatoes. Journal of the Science of Food and Agriculture, 96(11), 3772–3778.
Rojas-Grau, M.A., Raybaudi-Massilia, R.M., Soliva-Fortuny, R.C., Avena-Bustillos, R.J.; McHugh, T.H., & Mart’ın-Belloso, O. (2007). Apple puree-alginate edible coating as carrier of antimicrobial agents to prolong shelf-life of fresh-cut apples. Postharvest Biology and Technology, 45, 254–264, 2.
Seow, Y. X., Yeo, C. R., Chung, H. L., & Yuk, H. G. (2014). Plant essential oils as active antimicrobial agents. Critical Review in Food Science and Nutrition, 54(5), 625–644.
Sung, S.-Y., Sina, L. T., Tee, T.-T., Bee, S. T., Rahmat, A. R., Rahman, W. A. W. A., Tan, A.-C., & Vikhraman, M. (2013). Antimicrobial agents for food packaging applications. Trends in Food Science & Technology, 33(2), 110–123.
Tanveer, A., Farooq, U., Akram, K., Hayat, Z., Shafi, A., Nazar, H., & Ahmad, Z. (2015). Pomegranate extracts: a natural preventive measure against spoilage and pathogenic microorganisms. Food Reviews International, 31(1), 29–51.
Tehranifar, A., Selahvarzi, Y., Kharrazi, M., & Bakhsh, V. J. (2011). High potential of agro-industrial by-products of pomegranate (Punica granatum L.) as the powerful antifungal and antioxidant substances. Industrial Crops and Products, 34(3), 1523–1527.
Tigist, M., Workneh, T., & Woldetsadik, K. (2013). Effects of variety on the quality of tomato stored under ambient conditions. Journal of Food Science and Technology, 50(3), 477–486.
Vanderzant C, & Splittstoesser DF (2001) Enterobacteriaceae, coliforms, and Escherichia coli as quality and safety indicators. In APHA/TCMMF. Compendium of methods for the microbiological examination of foods (4th ed.). Washington.
Valencia-Sullca, C., Vargas, M., Atarés, L., & Chiralt, A. (2018). Thermoplastic cassava starch-chitosan bilayer films containing essential oils. Food Hydrocolloids, 75, 107–115.
Vásconez, M. B., Flores, S. K., Campos, C. A., Alvarado, J., & Gerschenson, L. N. (2009). Antimicrobial activity and physical properties of chitosan-tapioca starch based edible films and coatings. Food Research International, 42(7), 762–769.
Veras, H. N. H., Araruna, M. K. A., Costa, J. G. M., Coutinho, H. D. M., Kerntopf, M. R., Botelho, M. A., & Menezes, I. R. A. (2012). Topical anti inflammatory activity of essential oil of Lippia sidoides Cham.: possible mechanism of action. Phytotherapy Research, 27, 179–185.
Wang, Y., Xia, Y., Zhang, P., Ye, L., Wu, L., & He, S. (2017). Physical characterization and pork packaging application of chitosan films incorporated with combined essential oils of cinnamon and ginger. Food and Bioprocess Technology, 10(3), 503–511.
Zhao, Y., Teixeira, J. S., Gänzle, M. M., & Saldaña, M. D. A. (2018). Development of antimicrobial films based on cassava starch, chitosan and gallic acid using subcritical water technology. The Journal of Supercritical Fluids, 137, 101–110.
Acknowledgments
All of the authors thank the Coordination for the Improvement of Higher Education Personnel (CAPES, Brazil) for providing a scholarship for the first author.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Araújo, J.M.S., de Siqueira, A.C.P., Blank, A.F. et al. A Cassava Starch–Chitosan Edible Coating Enriched with Lippia sidoides Cham. Essential Oil and Pomegranate Peel Extract for Preservation of Italian Tomatoes (Lycopersicon esculentum Mill.) Stored at Room Temperature. Food Bioprocess Technol 11, 1750–1760 (2018). https://doi.org/10.1007/s11947-018-2139-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-018-2139-9