Abstract
A protein and astaxanthin-concentrated fraction (R f) can be recovered from shrimp cooking wastewater by ultrafiltration at 300 kDa, indicating that astaxanthin is somehow associated to membrane-retained proteins. Response surface analysis showed that astaxanthin can be extracted from R f using sunflower oil (3:1 v/v) under milder conditions (T < 40 °C) than directly from shrimp exoskeleton. Modeling astaxanthin extraction kinetics at 30 °C revealed that the process is a consequence of both mass transfer and hydrogen bonding between astaxanthin and oil. The freeze-dried concentrate (FR f ) showed two-phase extraction profiles with a much faster pigment recovery observed at 30 °C compared to the liquid form (R f). The best yields of astaxanthin extraction were not further improved after hydrolysis with alcalase at 45 °C for 30 min (HR f ), although higher yields were obtained when both R f and LR f were extracted in the presence of 200 mg/L butylated hydroxyanisole or ethoxyquin. Astaxanthin from this shrimp by-product has low thermal stability in oil at high temperatures (60 and 70 °C), suggesting the carotenoid is mainly free as a result of the cooking process and not bounded to proteins or lipids as it occurs in its natural form.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The fish processing industry generates several wastewater effluents (washing, thawing, rinsing, and cooking), which involve serious problems of pollution and environmental health. Among these effluents, cooking juice (more than 40 % of the total) contains a high saline content and organic load (Cros et al. 2006). To reduce their pollutant content, cooking wastewaters need to be treated, increasing the cost of the manufacturing process. So, an alternative to reduce wastewater processing costs would be the recovery of products with high added value such as proteins, aromas, and flavors (Vandajon et al. 2002).
During the last years, the application of membrane technology as the main method of separation, concentration, and purification of these valuable compounds from fish processing residual materials has been highly developed (Afonso et al. 2004; Murado et al. 2009, 2010). But also, given the carotenoprotein character of the pigmented by-product from crustacean process wastewaters (Cano-López et al. 1987; Simpson and Haard 1985), these effluents can be a possible source of carotenoids.
Astaxanthin (3,3-dihydroxy-β,β-carotene-4,4 dione) is a ketocarotene widely used in aquaculture as feed additive for the pigmentation of salmonid meat and shrimp and lobster shells. The majority of commercial astaxanthin for aquaculture is industrially produced by chemical synthesis (Rodríguez-Sáiz et al. 2010), although its increasing interest, due to novel applications as nutraceutical in the food, pharmaceutical, and cosmetic industries (Del Campo et al. 2007), has led to the search for new natural sources of astaxanthin. In this way, many studies describe the recovery of astaxanthin from shrimp by-products such as head and body skeleton (Armenta-López et al. 2002; Bi et al. 2010; Sachindra and Mahendrakar 2005). De Holanda and Netto (2006) also reported obtaining astaxanthin as a valuable subproduct of the chitin production from shrimp processing waste. Different extraction methods are applied for the recovery of astaxanthin from shrimp solid by-products such as enzymatic hydrolysis (De Holanda and Netto 2006), fermentative process (Sachindra and Bhaskar 2008), organic solvents (Sachindra et al. 2006), and vegetable oils (Chen and Meyers 1982; Handayani et al. 2008; Sachindra and Mahendrakar 2005).
The optimization of these extraction processes is usually conducted using one-factor-at-a-time approach; however, the use of response surface methodology (RSM) allows predicting optimal extraction conditions and interactions between variables. In fact, using this methodology to optimize alkali (Liu et al. 2012), solid-phase (Zhu and Row 2013), and supercritical CO2 (Yang et al. 2013), astaxanthin extraction from shrimp by-products has been recently reported. Also, factorial design methodologies have been successfully applied to the optimization of astaxanthin extraction using vegetable oils (Sachindra and Mahendrakar 2005) and organic solvents (Sachindra et al. 2006).
Astaxanthin in crustacean shells is mainly esterified or complexed with proteins (Matsuno 2001), increasing pigment stability. However, when extracted from natural sources, astaxanthin is unstable due to its sensitivity to various environmental factors such as light, oxygen, acidity, and heat (Mezzomo et al. 2011), causing the loss of its bioactivity. Lactic acid fermentation (Armenta-López et al. 2002) and the addition of antioxidants (Armenta-López et al. 2002; Chen and Meyers 1982) have improved the stability of astaxanthin, thereby leading to increase astaxanthin yield after extraction with different solvents.
In the present study, we describe a feasible process using membrane technology for the recovery of astaxanthin from shrimp cooking wastewater. This methodology allows obtaining a protein and astaxanthin-concentrated fraction that can be used as additive in the animal feed industry, while reducing the costs of wastewater treatment. This study also reports the optimized conditions (temperature, time, oil/waste ratio, and use of antioxidants) for carotenoid extraction using sunflower oil and proposes kinetic models that would be helpful for the further scale-up of the process.
Materials and Methods
Materials
The company Bajamar Séptima, Pescanova Group (A Coruña, Galicia, Spain) kindly provided the cooking wastewater from the industrial manufacturing of shrimp (Penaeus vannamei). Shrimp cooking juice was sampled and immediately stored at −18 °C until further use.
Analytical Determinations
Protein, total nitrogen, total sugar, and reducing sugar contents were determined from the samples taken before storage. Total nitrogen was determined by the method of Havilah et al. (1977). Soluble proteins were determined using the method of Lowry et al. (1951); total sugar content by the phenol-sulfuric acid method (Dubois et al. 1956), according to Strickland and Parsons (1968); and reducing sugars were quantified by means of a 3,5-dinitrosalicylic reaction (Bernfeld 1951).
Recovery of Astaxanthin by Ultrafiltration of Shrimp Wastewater
The concentration of astaxanthin from shrimp cooking juice consisted of ultrafiltration-diafiltration using a spiral polyethersulfone membrane (Millipore Prepscale) of 0.56 m2 with molecular weight cut-off (MWCO) of 300 kDa. The operation mode was as follows: an initial phase of ultrafiltration (UF) with total recycling of retentate, immediately followed by diafiltration (DF). During UF, the inlet pressure remained constant to determine the drops of flow rate due to the increased concentration of the retentate and to possible membrane adhesions. The final retentate (after DF) was divided into two batches: one was directly stored at −18 °C (R f) and the other freeze-dried (FR f ) and stored at 4 °C for further analysis. Both permeates in the UF and DF phase were discarded after analysis. The kinetics of UF and DF of the effluent were defined by the protein levels as determined by two procedures: the method of Lowry and total nitrogen multiplied by 6.25.
Enzymatic Hydrolysis Process
The enzymatic hydrolysis of the concentrated fraction was performed using a commercial protease, alcalase 2.4 L from Novo Co. (Novozyme Nordisk, Bagsvaerd, Denmark) at a ratio of 0.01:1 (Anson units per milliliter, AU/mL) enzyme/substrate. One AU is the amount of enzyme that, under standard conditions, digests hemoglobin at an initial rate that produces an amount of trichloroacetic acid-soluble product that gives the same color with the Folin reagent as one milliequivalent of tyrosine released per minute. The pH of the retentate was adjusted to pH 9.0 using 5 mM Britton-Robinson buffer, and proteolysis was carried out in a water bath with soft agitation at 45 °C for 30 min. The hydrolysate (HR f ) was stored at −18 °C until further use.
Combined Effect of Temperature, Heating Time, and Oil/Waste Ratio on Astaxanthin Extraction
A second-order rotatable design, based on three variables at five levels (Akhnazarova and Kafarov 1982; Box et al. 2005), was used to study the combined effect of temperature (T), time (t), and oil/waste ratio (R) on the yield of recovered astaxanthin from shrimp process wastewater. The joint effect of the three variables was studied in the R f fraction.
The experimental domains of each variable were 40–100 °C for T, 30–300 min for t, and 1.0–3.0 (v/v) for R. The design consisted of 20 experiments with four (22) factorial points, four axial points to form a central composite design with α = 1.682, and six center points for replication. The experimental domain and codification of the variables are shown in Table 1. Experimental data were fitted to the following empirical model with the yield of astaxanthin as dependent variable:
Statistical significance of the coefficients was evaluated by Student’s t test (α = 0.05). Consistency of the model was tested by Fisher’s F test (α = 0.05), using the following mean squares ratios:
The model is acceptable if | |
F 1 = Model / Total error | F 1 ≥ F numden |
F 2 = (Model + Lack of fitting) / Model | F 2 ≤ F numden |
F 3 = Total error / Experimental error | F 3 ≤ F numden |
F 4 = Lack of fitting / Experimental error | F 4 ≤ F numden |
Data fitting, parametric estimation performed by minimization of the sum of quadratic differences between experimental and model-predicted values, and significance tests both for parameters and model were performed with the Microsoft Excel spreadsheet.
Extraction of Astaxanthin Using Sunflower Oil
The extraction of astaxanthin in sunflower oil was carried out from the final retentate (R f), but also two other pretreated samples were studied as astaxanthin sources. For this purpose, R f was hydrolyzed using alcalase (HR f ) or freeze-dried (FR f ) in order to test if the carotenoid was more available to sunflower oil in any of these forms.
Extraction from both R f and HR f was performed using the optimized conditions defined by a second-order rotatable design, as previously described. In case of FR f , the oil/waste ratio was increased to 100:1, an adequate relation due to the increased concentration of the carotenoid as a consequence of the freeze-drying process. In the latter fraction, the extraction was studied at different temperatures: 30, 40, 50, and 60 °C. Extractions were carried out in stirred 250 mL flasks and appropriate R f or HR f volumes or FR f masses were added to sunflower oil preheated at the appropriate temperature. Duplicate samples were removed after different incubation times. Then samples were filtered through washed glass wool and centrifuged at 5,000g for 15 min, and the pigmented oil layer from the supernatant was recovered. The astaxanthin concentration was measured spectrophotometrically at λ max (487 nm: A 487), and the carotenoid yield as astaxanthin, for liquid (μg/mL) or solid (μg/g) samples, was determined using the following equation (Sachindra and Mahendrakar 2005):
where Y is the astaxanthin yield per volume of bulk liquid (μg/mL) or per shrimp waste mass (μg/g), V oil is the volume of recovered pigmented oil, V w is the volume of waste (for R f and HR f samples) or the weight of freeze-dried powder (for FR f samples), and E is the specific extinction coefficient.
Finally, the effect of the addition of butylated hydroxyanisole (BHA) or ethoxyquin (ETQ) at 200 mg/L on the astaxanthin extraction was also studied in both R f and FR f .
Mathematical Modeling of Extraction Kinetics
The dynamics of astaxanthin extraction were modeled using a mass transfer kinetic model (Handayani et al. 2008):
where Y and Y e are the astaxanthin yield in bulk liquid and at equilibrium per volume (μg/mL) or per mass of shrimp waste (μg/g), respectively; t is the time of the extraction process (min); and k L a is a volumetric mass transfer coefficient (min−1).
We also used a pseudo-second-order model that accounts for the esterification between hydroxyl groups in free astaxanthin and fatty acids in sunflower oil (Handayani et al. 2008). Considering that the concentration of astaxanthin at the beginning of the extraction process is zero and rewriting the equation in terms of yield:
where Y and Y e are the astaxanthin yield in bulk liquid and at equilibrium per volume (μg/mL) or per mass of shrimp waste (μg/g), respectively; t is the time of the extraction process (min); and k A is a reaction constant (min−1).
Finally, when a two-phase extraction profile was observed, data were modeled using the sum of two mass transfer kinetic models (biphasic model) with different volumetric mass transfer coefficients and yields at equilibrium (Y e1 and Y e2):
where Y e1 and Y e2 are the astaxanthin yields per mass of shrimp waste (μg/g) of the first and second phase, respectively; t is the time of the extraction process (min); k L1 a and k L2 a are the volumetric mass transfer coefficients of the first and second phase, respectively (min−1).
Considering that the sum of both Y e1 and Y e2 is the maximum yield of extraction achieved (Y m), and rewriting Eq. 5 in terms of a global process with a single yield at equilibrium (Y e), we have:
For comparative purposes, data were normalized by dividing astaxanthin yields (μg/mL or μg/g) by the highest yield (μg/mL or μg/g) of astaxanthin extracted from each fraction (R f, HR f , or FR f ) regardless of the experimental conditions assayed in each case.
Numerical and Statistical Methods
Fitting procedures and parametric estimates from the experimental results were performed by minimization of the sum of quadratic differences between observed and model-predicted values, using the nonlinear least-squares (quasi-Newton) method provided by the macro Solver of Microsoft Excel XP spreadsheet. Then, confidence intervals from the parametric estimates (Student’s t test) and consistence of mathematical models (Fisher’s F test), both with α = 0.05, were determined using SolverAid macro, which is freely available from de Levie’s Excellaneous website: http://www.bowdoin.edu/~rdelevie/excellaneous/.
Results and Discussion
Ultrafiltration of Shrimp Wastewater
The shrimp cooking wastewater utilized in this work had a pH of 6.07 ± 0.04, a protein content of 1.92 ± 0.08 g/L, and a total soluble sugar concentration of 0.21 ± 0.02 g/L. In the diafiltration with constant volume (filtration flow = water intake flow), the concentration (or the total amount) of a permeable solute in the retentate follows a first-order kinetics (Amado et al. 2013; Murado et al. 2010):
where C is the concentration of the permeable solute in the retentate, with C 0 as the initial value. C f is the final asymptotic value if only a part of a polydisperse solute is permeable. When using normalized values (%): C 0 + C f = 100, with C f = 0 when the solute is completely permeable. The parameter s is the solute-specific retention, which varies between 0 (the solute is filtered as the solvent) and 1 (the solute is totally retained). D r is the relative diavolume or the ratio volume of added water/constant retentate volume.
This equation satisfactorily described the kinetics of protein diafiltration process with a molecular cut-off at 300 kDa (Fig. 1). The values of the coefficients were C f = 75.9 % and s = 0.381, which means a rather high retention of the protein and also a specific retention that would demand a relative diavolume of 5.7 to eliminate 99 % of permeable protein. In a common diafiltration, with an initial volume of 2 L of concentrated shrimp wastewater and working with a relative diavolume of 5, at 50–55 °C and 2 atm (~30 psi), the protein concentration in the retentate can be maintained around 15–20 g/L, with a filtrate flow that decays 40–45 % during the process and maintains an average value of 325 mL min−1 m−2 (data not shown). Under these experimental conditions, the values of protein calculated by the Lowry method or total nitrogen × 6.25 were almost indistinguishable (Fig. 1). These results indicate a high retention of peptidic material after ultrafiltration of shrimp cooking wastewater despite the heat treatment during shrimp processing.
Ultrafiltration-diafiltration kinetics of shrimp (Penaeus vannamei) cooking wastewater using a polyethersulfone membrane with MWCO at 300 kDa. Left: concentration of retained protein in linear relation with the factor of volumetric concentration (fc) showing experimental data (points) and theoretical profiles (discontinuous line). Right: progress of protein (empty circle) and nitrogen (filled circle) retention with the increase of diavolume from DF process (D). For clarity, confidence intervals (in all cases, less than 5 % of the experimental mean value; α = 0.05; n = 2) were omitted
The ultrafiltration-diafiltration process with a molecular cut-off at 300 kDa showed a high retention of astaxanthin despite the low molecular weight (597 Da) of this pigment. During the ultrafiltration phase, the initial permeates showed slight yellowish coloration, completely disappearing after diafiltration and leading to an intense colored retentate. In fact, a concentration factor of 13 is achieved after 300 kDa UF-DF of shrimp cooking wastewater, reaching average astaxanthin concentrations between 10 and 13 μg mL−1 in the retentate extracted with sunflower oil.
These results suggest that aggregation phenomena are occurring due to the hydrophobic properties of astaxanthin. It is known that astaxanthin from the shell matrix of crustaceans is mainly found esterified or complexed with proteins (Matsuno 2001). Therefore, astaxanthin in the retentate must be forming polymeric aggregates (Velu et al. 2003) and/or bound to macromolecules, mainly proteins, that are retained during ultrafiltration using the reported cut-off membrane.
Accordingly, the 300 kDa concentrated fraction could be used as a supplement for animal diets due to its high astaxanthin and protein content. In the same way, Pérez-Santín et al. (2013) obtained a concentrate rich in lipids and proteins with crustacean aroma, attractive orange coloring, and antioxidant and ACE-inhibitory capacities making it attractive for the formulation of feeds or functional foods.
Enzymatic Hydrolysis
The use of proteolytic enzymes has been widely reported to disrupt the protein-carotenoid complex and increase astaxanthin extraction from solid shrimp by-products (De Holanda and Netto 2006; Sowmya et al. 2011). With this purpose, in a preliminary experiment, the hydrolysis conditions using alcalase were optimized to maximize the astaxanthin recovery without compromising its stability. Different temperatures (35, 45, and 55 °C) and times (30, 60, 90, and 120 min) of hydrolysis were assayed, maintaining a constant ratio of 0.01:1 (AU/mL) enzyme/substrate. After each incubation time, samples were withdrawn and quickly cooled down in an ice-water bath for 5 min to inactivate the protease. Then astaxanthin was extracted in sunflower oil at 70 °C, according to the optimal conditions reported in the literature for the extraction of carotenoids from shrimp waste using vegetable oils (Sachindra and Mahendrakar 2005), for 30 min.
Results are shown in Fig. 2 where the yields of astaxanthin recovery were calculated according to Eq. (2). The highest recovery of astaxanthin under these conditions (70 °C, 30 min) is obtained at 45 °C, falling a 70 % on average when the temperature of hydrolysis is 35 or 55 °C. Moreover, astaxanthin yields decreased correlatively with the incubation time at all temperatures tested. At the optimum temperature, the recovery of astaxanthin decreased about 36 % when the reaction time increased from 30 to 120 min. Taking into account these results, the hydrolysis conditions selected were as follows: 30 min at 45 °C using a ratio of 0.01:1 (AU/mL) alcalase/substrate.
Combined Effect of Temperature, Heating Time, and Oil/Waste Ratio on the Extraction of Astaxanthin
The effects of temperature, heating time, and oil/waste ratio on the yield of astaxanthin recovery are important factors that must be considered for a further scale-up of the process. Although the combined effect of these variables can be studied using a one-factor-at-a-time approach, this methodology cannot predict the optimal reaction conditions, ignores interactions, and may lead to misleading conclusions. In this regard, experimental design methodologies (Box et al. 2005) are more efficient than the one-factor-at-a-time approach. Response surface methodology uses statistical and mathematical techniques to evaluate the combined effect of factors instead of single factors at different times.
In this work, a second-order rotatable design, based on three variables at five levels (Akhnazarova and Kafarov 1982; Box et al. 2005), was used to study the combined effect of temperature (T), time (t), and ratio oil/waste ration (R) on the yield of astaxanthin recovery. The experimental domain is shown in Table 1, with temperatures and oil/waste ratios selected according to previous reported conditions for the extraction of astaxanthin using vegetable oils (Sachindra and Mahendrakar 2005). Applying the significance criteria specified in the “Materials and Methods” section, the empirical model obtained for the theoretical yield of extracted astaxanthin (A e) as a function of the three processing variables was:
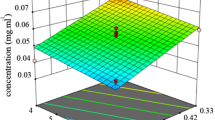
The response surfaces obtained varying two independent variables, when the third variable is kept at a constant value, are depicted in Fig. 3, and the complete statistical analysis is shown in Table 2. The analysis of variance indicates that the model is significant (α = 0.05) and the adjusted R 2 value shows a good correlation with the experimental data. Besides, according to the statistical analysis, all the parameters in Eq. (8) were significant.
Response surfaces of the combined effect of temperature (T) and ratio oil/waste (R) (left) and temperature (T) and time of extraction (t) (right) on the predicted yield of extracted astaxanthin (Y) according to Eq. (8)
The response surface for astaxanthin yield as a function of temperature and oil/waste ratio (Fig. 3, left) indicates that the extraction yield increases linearly with the oil/waste ratio. At high temperatures, the response increases notably (96 % within the experimental domain) with the proportion of extracting agent. By contrast, at low extraction temperatures, the differences on the astaxanthin yield by varying the phase relationship are much lower (20 %). It should also be noted that at high temperatures and low phase relationships, a degradation of the pigment is observed resulting in practically null values of recovered astaxanthin. Figure 3 (right) further confirms these results since it shows that the increase in the extraction time has an effect on astaxanthin recovery only at low temperatures. This result also agrees with that reported by Pu et al. (2010), who found that shrimp astaxanthin degradation in flaxseed oil was significantly influenced by temperature, with increased degradation rates at 50 and 60 °C compared to 30 and 40 °C using a 1:1 (w/v) phase relationship.
Although an absolute maximum response was not achieved within the experimental domain, maximal yields can be obtained at low temperature (<40 °C), high oil/waste ratio (3:1 v/v), and high incubation time (>4 h). Our results also suggest that the extraction could be performed at lower temperatures (25–30 °C) without appreciable loss in astaxanthin yield and even improving pigment recovery.
Interestingly, our results reveal that astaxanthin can be recovered from shrimp cooking wastewaters using milder conditions than the usual high temperatures (Sachindra and Mahendrakar 2005) and organic solvents (Sachindra et al. 2006) utilized for the extraction of astaxanthin from crustacean shells. The fact that astaxanthin is more easily extracted from the liquid effluent than from solid by-products is likely to be due to the cooking process. In fact, several authors suggest that cooking can break the carotenoid-protein complex, releasing the carotenoid compounds and facilitating its extraction (Hornero-Méndez and Mínguez-Mosquera 2007; Mezzomo et al. 2011).
Mathematical Modeling of Astaxanthin Extraction Kinetics
Optimal values from the factorial design were applied to further improve astaxanthin yield, and so extraction kinetics were performed at low temperature (30 °C) and increasing extraction times. Extraction kinetics from R f and HR f at different oil/waste ratios and their predicted profiles using Eqs. (3) and (4) are shown in Fig. 4. All parameters were statistically significant (Student’s t test, α = 0.05), and the predictive ability of both equations was high with a goodness of fit of not less than 0.970 (Table 3).
Nevertheless, the pseudo-second-order model (Eq. (4)) showed better correlations (R 2) than the mass transfer kinetic model (Eq. (3)) in all oil/waste ratios. Handayani et al. (2008) also observed better adjustment of Eq. (4) to the extraction kinetics of shrimp waste in palm oil, which they attributed to the reaction between the hydroxyl groups in astaxanthin with fatty acids. According to these authors, the extraction process is a consequence of both mass transfer and hydrogen bonding between astaxanthin and oil.
Higher astaxanthin yield at equilibrium (Y e) was found for R f fraction with the increase of oil/waste ratio, whereas identical Y e values were obtained for HR f at the three assayed oil/waste ratios (Table 3). According to these results, the lowest oil/waste ratio might be insufficient to allocate globular proteins in R f which would tend to be more retained in the oil-water interphase. Astaxanthin can then be partially partitioned between the oil and water interphase. Owing to the excluded volume interactions (Mazzola et al. 2008) between the carotenoid and these proteins, lower astaxanthin concentrations can be recovered in the oily phase. On the contrary, peptides in HR f are easier to order and tend to go into the aqueous phase and so astaxanthin could be more easily separated after filtration and centrifugation.
On the other hand, kinetic constants from Eqs. (3) and (4) (k L a, min−1 and k A, min−1) were greater when a 2:1 (v/v) ratio was used for oil extraction (Table 3), suggesting that this phase relationship is optimal for mass transfer and reaction between astaxanthin and fatty acids in both R f and HR f .
The effect of increasing temperatures on the pigment extraction kinetics from FR f was also studied. Experimental trends showed the existence of two phases along extraction time, that is, two mass transfer phenomena with different rates (Fig. 5). Such behavior could be due to astaxanthin existing in different forms dependent on the affinity, degree or strength of pigment-protein interactions, and also to the presence of free astaxanthin (Pérez-Santín et al. 2013). These profiles made it necessary to use a biphasic equation as Eq. (6) to more adequately adjust the experimental data than Eq. (4), as can be seen according to determination coefficients (Table 5). The normalized maximum yield of extraction (Y m) or the asymptotic value on the plateau phase (Fig. 5, continuous line) was calculated by parametric estimation, minimizing the sum of quadratic differences between the observed and model (Eq. (6)) predicted values. Y m was dependent with temperature, and so lower Y m values were obtained with temperature increase. This result is in concordance with those using the response surface approach where maximal yields were achieved at low temperatures (<40 °C). According to the literature, astaxanthin in its free form is unstable and extremely sensitive to factors such as light, oxygen, acidity, and heat (Mezzomo et al. 2011), so these results also support the hypothesis that cooking can break the carotenoid-protein complex and that astaxanthin from cooking wastewater could be mainly in its free form. Indeed, a recent study conducted to investigate the compositional properties and bioactive potential of shrimp cooking juice (Pérez-Santín et al. 2013) identified free astaxanthin (cis and trans isomers) and derived monoesters or diesters in the organic phase extracted from the liquid supernatant of this waste material.
Kinetics of astaxanthin extraction from the 300 kDa freeze-dried retentate (FR f ) from shrimp cooking wastewater at different temperatures: 30 (filled circle), 40 (filled square), 50 (filled diamond), and 60 °C (filled triangle). Experimental data (points) and fittings to Eqs. (4) (broken line) and (6) (solid line)
Moreover, extraction at 30 °C was much faster from FR f than from either R f or HR f , as can be seen in view of the kinetic constants from Eq. (4) (Tables 3 and 4). The effect of freeze-drying on the improvement of astaxanthin recovery is in accordance with the results of Mezzomo et al. (2011), who observed that cooking, drying, and milling improved the total carotenoid content recovered from shrimp residues, for instance, the head, carapace, and tail, after extraction using different solvents.
Finally, extraction kinetics in the presence of synthetic antioxidants were performed in order to study the effect these compounds had on astaxanthin recovery. A concentration of 200 mg/L was selected according to previous dosages applied for astaxanthin extraction in oil (Chen and Meyers 1982) and to the maximum concentrations approved by the EU (Regulation 1831/2003) for animal feeds (150 mg/kg). According to our results (Fig. 6 and Table 5), the addition of either BHA or ETQ improved astaxanthin extraction in sunflower oil. Although different behaviors were observed whether astaxanthin was extracted from R f (Fig. 6a) or FR f (Fig. 6b). The addition of BHA or ETQ significantly (p < 0.05) increased astaxanthin extraction compared to the control when the pigment was extracted from R f, although these differences were not significant (p > 0.05) in FR f . However, a slower extraction is also observed when performed in the presence of either two antioxidants (Table 5), suggesting that they have a stabilizing effect on astaxanthin that in turn explains the improved extraction observed in the water-oil system. The stabilizing effect of antioxidants such as ethoxyquin has been reported to increase astaxanthin extraction from crawfish waste using soy oil (Chen and Meyers 1982). Also, the inoculation of shrimp wastes with a Lactobacillus culture isolated from tropical shrimp stabilized astaxanthin structure, increasing its extraction yields using different organic solvents (Armenta-López et al. 2002).
In the present study, the normalized maximum yields of extraction (Y m) for R f, HR f , and FR f correspond, under the best conditions determined for each of these fractions, to astaxanthin concentrations of 12.9, 11.2 μg/mL, and 210 μg/g, respectively. The addition of BHA increased astaxanthin recovery to 17.3 μg/mL in R f and 299 μg/g in FR f , while ETQ led to concentrations of 22.6 μg/mL and 282 μg/g for the liquid and solid shrimp concentrates, respectively.
Conclusions
Our results show that a protein and astaxanthin-concentrated fraction (R f) can easily be recovered from shrimp processing wastewaters by UF at 300 kDa. Response surface methodology was successfully applied to optimize extraction conditions from R f showing an optimal temperature lower (<40 °C) than that utilized for astaxanthin recovery from solid shrimp wastes. Further analysis of extraction kinetics at 30 °C showed that astaxanthin recovery is a consequence of both mass transfer and hydrogen bonding between astaxanthin and oil. The freeze-dried concentrate wastewater (FR f ) showed a two-phase extraction and, at 30 °C, was much faster than from the liquid form. No improvement in astaxanthin yields was observed after hydrolysis with alcalase at 45 °C for 30 min (HR f ), but higher recoveries were obtained when both R f and LR f were extracted in the presence of 200 mg/L BHA or ETQ. Astaxanthin from this shrimp by-product showed low thermal stability in oil at high temperatures (60 and 70 °C), suggesting that the carotenoid is mainly free as a result of the cooking process and not bounded to proteins or lipids as it occurs in its natural form (Fig. 7).
References
Afonso, M. D., Ferrer, J., & Bórquez, R. (2004). An economic assessment of proteins recovery from fish meal effluents by ultrafiltration. Trends in Food Science and Technology, 15(10), 506–512.
Akhnazarova, S. L., & Kafarov, V. V. (1982). Experiment optimization in chemistry and chemical engineering. Moscow: MIR.
Amado, I. R., Vázquez, J. A., González, M. P., & Murado, M. A. (2013). Production of antihypertensive and antioxidant activities by enzymatic hydrolysis of protein concentrates recovered by ultrafiltration from cuttlefish processing wastewaters. Biochemical Engineering Journal, 76, 43–54.
Armenta-López, R., Guerrero, I. L., & Huerta, S. (2002). Astaxanthin extraction from shrimp waste by lactic fermentation and enzymatic hydrolysis of the carotenoprotein complex. Journal of Food Science, 67(3), 1002–1006.
Bernfeld, P. (1951). Enzymes of starch degradation and synthesis. Advances in Enzymology, 12, 379–427.
Bi, W., Tian, M., Zhou, J., & Row, K. H. (2010). Task-specific ionic liquid-assisted extraction and separation of astaxanthin from shrimp waste. Journal of Chromatography B, 878(24), 2243–2248.
Box, G. E. P., Hunter, J. S., & Hunter, W. G. (2005). Statistics for experimenters: design, innovation, and discovery. Hoboken: Wiley.
Cano-López, B. K., Simpson, B. K., & Haard, N. F. (1987). Extraction of carotenoprotein from shrimp process wastes with the aid of trypsin from Atlantic cod. Journal of Food Science, 52(2), 503–504.
Chen, H., & Meyers, S. P. (1982). Extraction of astaxanthin pigment from crawfish waste using a soy oil process. Journal of Food Science, 47(3), 892–896.
Cros, S., Lignot, B., Jaouen, P., & Bourseau, P. (2006). Technical and economical evaluation of an integrated membrane process capable both to produce an aroma concentrate and to reject clean water from shrimp cooking juices. Journal of Food Engineering, 77(3), 379–471.
De Holanda, H. D., & Netto, F. M. (2006). Recovery of components from shrimp (Xiphopenaeus kroyeri) processing waste by enzymatic hydrolysis. Journal of Food Science, 71(5), C298–C303.
Del Campo, J. A., García-González, M., & Guerrero, M. G. (2007). Outdoor cultivation of microalgae for carotenoid production: current state and perspectives. Applied Microbiology and Biotechnology, 74(6), 1163–1174.
Dubois, M., Gilles, K., Hamilton, J., Rebers, P., & Smith, F. (1956). Colorimetric method for determination of sugars and related substances. Analytical Chemistry, 28(3), 350–356.
European Union. Regulation (EC/No 1831/2003) on additives for use in animal nutrition. (2003). Official Journal L 268, 29–43
Handayani, A. D., Sutrisno, Indraswati, N., & Ismadji, S. (2008). Extraction of astaxanthin from giant tiger (Panaeus monodon) shrimp waste using palm oil: studies of extraction kinetics and thermodynamic. Bioresource Technology, 99(10), 4414–4419.
Havilah, E. J., Wallis, D. M., Morris, R., & Woolnough, J. A. (1977). A micro-colorimetric method for determination of ammonia in Kjeldahl digests with a manual spectrophotometer. Laboratory Practice, 26, 545–547.
Hornero-Méndez, D., & Mínguez-Mosquera, M. I. (2007). Bioaccessibility of carotenes from carrots: effect of cooking and addition of oil. Innovative Food Science and Emerging Technologies, 8, 407–412.
Liu, H., Li, P., Wang, G., Yu, H., Zeng, Z., & Yang, D. (2012). Optimization for extraction of astaxanthin from shrimp shell using response surface method. Advanced Materials Research, 396–398, 609–613.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Protein measurement with the Folin phenol reagent. Journal of Biological Chemistry, 193(1), 265–275.
Matsuno, T. (2001). Aquatic animal carotenoids. Fisheries Science, 67, 771–783.
Mazzola, P. G., Lopes, A. M., Hasmann, F. A., Jozala, A. F., Penna, T. C. V., Magalhaes, P. O., et al. (2008). Liquid-liquid extraction of biomolecules: an overview and update of the main techniques. Journal of Chemical Technology & Biotechnology, 83(2), 143–157.
Mezzomo, N., Maestri, B., dos Santos, R. L., Maraschin, M., & Ferreira, S. R. S. (2011). Pink shrimp (P. brasiliensis and P. paulensis) residue: influence of extraction method on carotenoid concentration. Talanta, 85(3), 1383–1391.
Murado, M. A., González, M. P., & Vázquez, J. A. (2009). Recovery of proteolytic and collagenolytic activities from viscera by-products of rayfish (Raja clavata). Marine Drugs, 7(4), 803–815.
Murado, M. A., Fraguas, J., Montemayor, M. I., Vázquez, J. A., & González, P. (2010). Preparation of highly purified chondroitin sulphate from skate (Raja clavata) cartilage by-products. Process optimization including a new procedure of alkaline hydroalcoholic hydrolysis. Biochemical Engineering Journal, 49(1), 126–132.
Pérez-Santín, E., Calvo, M. M., López-Caballero, M. E., Montero, P., & Gómez-Guillén, M. C. (2013). Compositional properties and bioactive potential of waste material from shrimp cooking juice. LWT - Food Science and Technology, 54(1), 87–94.
Pu, J., Bechtel, P. J., & Sathivel, S. (2010). Extraction of shrimp astaxanthin with flaxseed oil: effects on lipid oxidation and astaxanthin degradation rates. Biosystems Engineering, 107(4), 364–371.
Rodríguez-Sáiz, M., de la Fuente, J. L., & Barredo, J. L. (2010). Xanthophyllomyces dendrorhous for the industrial production of astaxanthin. Applied Microbiology and Biotechnology, 88(3), 645–658.
Sachindra, N. M., & Bhaskar, N. (2008). In vitro antioxidant activity of liquor from fermented shrimp biowaste. Bioresource Technology, 99(18), 9013–9016.
Sachindra, N. M., & Mahendrakar, N. S. (2005). Process optimization for extraction of carotenoids from shrimp waste with vegetable oils. Bioresource Technology, 96(10), 1195–1200.
Sachindra, N. M., Bhaskar, N., & Mahendrakar, N. S. (2006). Recovery of carotenoids from shrimp waste in organic solvents. Waste Management, 26(10), 1092–1098.
Simpson, B. K., & Haard, N. F. (1985). The use of proteolytic enzymes to extract carotenoproteins from shrimp processing wastes. Journal of Applied Biochemistry, 7, 212–222.
Sowmya, R., Rathinaraj, K., & Sachindra, N. M. (2011). An autolytic process for recovery of antioxidant activity rich carotenoprotein from shrimp heads. Marine Biotechnology, 13(5), 918–927.
Strickland, J. D. H., & Parsons, T. R. (1968). A practical handbook of seawater analysis. Journal of the Fisheries Research Board of Canada, 167, 57–62.
Vandajon, L., Cros, S., Jaouen, P., Quéméneur, F., & Bourseau, P. (2002). Recovery by nanofiltration and reverse osmosis of marine flavours from seafood cooking waters. Desalination, 144, 379–385.
Velu, C. S., Czeczuga, B., & Munuswamy, N. (2003). Carotenoprotein complexes in entomostracan crustaceans (Streptocephalus dichotomus and Moina micrura). Comparative Biochemistry and Physiology Part B: Biochemistry and Molecular Biology, 135(1), 35–42.
Yang, X., Zhang, Z., Zheng, Q., Zu, T., & Shu, Y. (2013). Optimization of supercritical CO2 extraction of astaxanthin from pacific white shrimp (Litopenaeus vannamei) using response surface methodology. Nongye Gongcheng Xuebao/Transactions of the Chinese Society of Agricultural Engineering, 29(SUPPL1), 294–300.
Zhu, T., & Row, K. H. (2013). Extraction of astaxanthin from shrimp waste using response surface methodology and a new hybrid organic-inorganic monolith. Separation Science and Technology, 48(10), 1510–1517.
Acknowledgments
We thank Dalva Martínez from Bajamar Séptima, Pescanova Group (A Coruña, Galicia, Spain) for providing the fresh shrimp cooking wastewater. Bajamar Séptima (Contract No. 20090910 co-financed by the Centre for Industrial Technological Development (CDTI)) supported this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Amado, I.R., Vázquez, J.A., Murado, M.A. et al. Recovery of Astaxanthin from Shrimp Cooking Wastewater: Optimization of Astaxanthin Extraction by Response Surface Methodology and Kinetic Studies. Food Bioprocess Technol 8, 371–381 (2015). https://doi.org/10.1007/s11947-014-1403-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-014-1403-x