Abstract
Historically, open radical nephrectomy (ORN) represented the standard of care for localized renal cell carcinoma (RCC). While the incidence of T1 RCC is rising, treatment options are developing fast and the standard of care according to European and American guidelines has changed to partial nephrectomy (PN), or laparoscopic radical nephrectomy in patients not suitable for PN. To assess the implementation of guideline recommendations and to profile recent surgical and technical innovations, we reviewed the current literature. We observed that ORN still represents the most commonly used treatment in T1 RCC patients. Utilization of PN increased over time but implementation is still in progress. Whereas PN is frequently used in tertiary care centers, population-based studies suggest discrepancies in the diffusion of standard of care treatments. Alternative minimally invasive approaches for PN are available but their superiority is not yet proven. Further efforts in improving the training of urologic surgeons are required to continue the implementation of guideline recommendations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Surgical management is associated with a reduction in cancer specific mortality in patients with T1 renal cell carcinoma (RCC) compared to observation [1]. Improvements and increased use of imaging techniques lead to a rising incidence of RCC with a downward stage migration and an increased number of T1 RCC patients [2, 3••]. Simultaneously, novel surgical techniques such as partial nephrectomy (PN) and minimally invasive techniques have evolved tremendously and represent an alternative to open radical nephrectomy (ORN). In fact, PN has been shown to provide improved renal function, lower rates of chronic kidney disease [4, 5], fewer cardiovascular events [6] and comparable oncological outcomes, relative to radical nephrectomy (RN) [4, 5, 7–11, 12••, 13–15]. Moreover, laparoscopic radical nephrectomy (LRN) has been shown to provide equal oncological outcomes [16–18] and lower perioperative morbidity [17–19] compared to ORN. As a consequence, according to current guidelines from the European Association of Urology (EAU) [20] and the American Urologic Association (AUA) [21], PN has become the standard of care for T1 RCC patients and should be performed whenever possible. If a PN is not feasible, a LRN should be the alternative.
Despite explicit guideline recommendations [20, 21], and even consideration as malpractice [22], ORN remains the most widely used treatment in the management of T1 RCC. In the United States (US), even in patients harboring tumors of 4 cm or less (T1a), up to 66 % were treated with ORN in 2005. Between 1988 and 2005, a majority of patients (78.3 %) received ORN whereas 4.1 % were treated by LRN, 16.4 % were treated by open partial nephrectomy (OPN), and 1.3 % were treated by laparoscopic partial nephrectomy (LPN). [23••] Knowing this data, we hypothesize that there is an underuse of current guideline conform treatments such as PN or minimally invasive nephrectomies. In this review we focus on the spread of nephron-sparing and minimally invasive nephrectomy among T1 RCC patients and highlight potential reasons for their underuse in the urologic community.
Radical Nephrectomy
At present, RN–preferably performed via the laparoscopic approach–is only recommended in the treatment of T1 RCC when PN is not feasible [20]. However, ORN represented the predominant treatment even among North American RCC patients treated for tumors sized 4 cm or less between 1988 and 2005. For those patients with the smallest renal masses, the authors observed a decrease in the utilization of ORN during this time period, from 89 % in 1988 to 66 % in 2005. At the same time, utilization of LRN increased to a peak of 6 % in 2000 and decreased moderately to 4 % in 2005 [23••]. Intuitively, in patients harboring T1b RCC, rates of RN are even higher and range from 89.6 % to 96.6 % in population-based studies (Table 1) [24, 26, 31•]. In general, for patients harboring T1 RCC, the rate of RN in population-based studies decreased over time [24–26, 31•, 33], but remains high with still 63.4 % in 2008 [31•]. Unfortunately, the authors did not differentiate between LRN and ORN, so that currently, evidence on the nationwide use of LRN relative to ORN and PN for T1 RCC patients after 2005 is sparse. The American Board of Urology (ABU) records all renal surgeries, not considering the underlying treatment indication, (e.g. tumor nephrectomy for any stage, renal transplant). According to this dataset, the rate of ORN decreased from 54 % (2002) to 29 % (2010) [35]. Regarding a population-based study on T1a RCC patients (1988–2005), 5 % of all RN were performed laparoscopically [23••]. In contrast, within T1 RCC patients undergoing nephrectomy at Memorial Sloan-Kettering Cancer Center between 2000 and 2007, 15.4 % were performed via the laparoscopic approach [12••].
Reasons for the excessive use of ORN are severalfold. First, as shown for other uro-oncologic procedures, laparoscopic surgery requires special equipment, which is more costly, compared to open surgery [36]. Second, laparoscopic or nephron-sparing surgery requires special training and surgical skills, which might not be universally available. Third, laparoscopic access to the kidney is not possible or reasonable in every patient, due to medical conditions such as polycystic kidneys, history of extensive abdominal surgery, non-tolerance to pneumoperitoneum, difficult retraction, bleeding, failure to progress, and difficult access [37].
However, these arguments cannot fully explain the excessive use of ORN in T1 RCC patients, which does not conform to the widely accepted guideline recommendations. Accordingly, there still is an overuse of ORN in the urologic community, relative to LRN and-even more important-relative to PN.
Partial Nephrectomy
PN currently represents the standard of care for T1 RCC patients [20]. Following population-based studies, the rates of PN in the US for T1 RCC patients increased from 9.6 % [24] (1988–2001), over 15.9 % [26] (1989–2004), and 27.1 % [31•] (1998–2008), to 31.2 % [33] (2000–2008). In 2008, 36.6 % of all T1 RCC patients who underwent nephrectomy were treated with PN [31•]. Intuitively, the likelihood of receiving PN compared to RN increases with decreasing tumor size and tumor complexity. Accordingly, for T1a RCC patients, the rates of PN in population based studies (Table 1) varied from 15.2 % to 37.9 %, with an increase from 4.8 % (1988) to 49.4 % (2008). In contrast, only 3.4 % to 10.4 % of all patients with T1b RCC patients were treated with PN in the community setting [24, 26, 31•],
Kutikov et al. [38] showed that the probability to receive PN was significantly associated with tumor complexity, as quantified by the R.E.N.A.L. (radius, exophytic/endophytic properties, nearness of tumor to the collecting system or sinus in millimeters, anterior/posterior and location relative to polar lines) score. Specifically, patients with a respectively low, moderate or high R.E.N.A.L. score, indicating low, moderate or high tumor complexity, received PN in 88.9 %, 84.2 % and 31.9 % of all cases, respectively [38]. These results were confirmed by Canter et al. [39••] who observed that patients with a low R.E.N.A.L. score were more likely to receive PN (94 %) compared to their high R.E.N.A.L. score counterparts (34 %).
In general, the diffusion of PN in daily practice advanced more rapidly in high volume, teaching and tertiary care centers. Regarding data from tertiary care centers the rate of PN among T1 RCC patients increased from 37.8 % (1987–2007) [28] to 56 % (2000–2007) [12••].
Stratified for tumor size, up to 51.4 % and 20.5 % of all patients with T1a and T1b tumors treated in six European tertiary centers between 1987–2007 [28] were treated with PN. According to data from North American centers, between 2000 and 2007, the rates of PN increased from 69 % up to 89 % in patients with T1a RCC, and from 20 % to 60 % in patients harboring T1b RCC [12••].
Only few studies separately report rates of OPN vs. LPN. For example, Bianchi et al. reported that within T1a RCC patients treated with PN, 92.7 % underwent OPN (1988–2005). In this population based study, the usage of OPN within all T1a RCC patients increased from 7 % to 29 % [23••]. Analyzing case logs from certified urologists, not considering the underlying treatment indication, Poon et al. reported a 41.2 % OPN rate in 2011 in patients treated with PN [35].
Regarding T1 RCC patients treated with PN, the rate of an open approach ranged between 57.2 % [30] in a multi-institutional series and 88.1 % [12••] in a single center experience from Memorial Sloan-Kettering Cancer Center.
LPN is technically more challenging compared to OPN [20] but has shown excellent perioperative outcomes in experienced hands, providing comparable oncologic and survival outcomes [30, 40–42]. Although, compared to OPN, renal damage is potentially more pronounced, due to warm ischaemia, a longer ischaemia time and longer OP time [41, 42], renal function after LPN seems to be comparable after a follow-up of 3.6 years [42]. More prospective long-term-and ideally randomized-data are warranted to evaluate the impact of LPN on renal function and survival compared to OPN.
According to a population-based study (1988–2005), the LPN rate within T1a RCC patients treated with PN was 7.3 %, whereas the proportion of LPN usage overall T1a RCC patients was 1.3 % and remained low throughout the study period with 1.9 % in 2005 [23••]. The usage of LPN was more frequent in tertiary care centers. Here, the rate of LPN within all T1 RCC patients was 6.6 %, whereas, within patients treated with PN, the ratio ranged between 11.9 % and 42.8 % [30].
In an observational study including patients harboring benign and malignant kidney tumors of any size and stage, an increase in the use of LPN from 2 % (2002) to 17 % (2010) has been observed. Here, the rate of LPN within the patients treated with PN was 40.2 % [35]. To summarize, in the urological community PN is underutilized, especially in non-academic, non-teaching hospitals [23••, 24, 25, 43]. Recent data suggest that at present, LPN remains a treatment alternative in the management of selected patients with easy accessible, preferably exophytic T1 RCC, treated at high volume laparoscopic centers and academic or teaching institutions.
Reasons for the slow implementation of PN are most likely multifactorial. First, PN represents a technically more challenging procedure compared to RN and might, therefore, be avoided by surgeons less well trained during their medical education or with a lower annual case load. A second hypothesis is that, in non-academic and non-teaching hospitals, the beneficial impact of PN on renal outcomes and OS compared to RN might be less appreciated [43]. Third, it has been shown that in the US, certain sociodemographic and socioeconomic factors determine the access to the standard of care in T1 RCC patients. For example, younger age, male gender, Caucasian, married status, more recent year of diagnosis [3••, 23••, 34•, 44], as well as higher income and private insurance [33], predispose patients for PN. In contrast, is has been shown that black females were 47 % less likely to receive PN compared to their male and white counterparts [45].
New Techniques
Minimally invasive treatment options in the context of nephrectomy include traditional laparoscopic nephrectomy, as well as more recent approaches such as laparoendoscopic single-site surgery (LESS), robot-assisted surgery and natural orifice transluminal endoscopic surgery (NOTES).
Laparoendoscopic Single-Site Surgery
LESS is performed by one single skin incision for the introduction of camera and instruments [46]. The first laparoendoscopic single-site surgery radical nephrectomy (LESS-RN) was performed in 2008 [47] and feasibility of LESS-RN has been confirmed [48] with a main advantage in cosmetic outcomes.
Based on the novelty of that technique, no observational data are available but several smaller studies exist, comparing LESS-RN and LRN, suggesting both approaches to be equal in terms of operative complications, estimated blood loss and warm ischaemia time (WIT). LESS-RN patients might be associated with reduced postoperative pain, shorter length of stay (LOS), shorter recovery time and better cosmetic results but also with longer operative time and higher rates of conversion to open nephrectomy compared to LRN [49–59].
Laparoendoscopic single-site surgery partial nephrectomy (LESS-PN) is a challenging technique which is used only in few centers and published experience is sparse. According to the EAU guidelines, LESS-PN can provide an alternative approach in experienced hands but currently it is recommended only as part of clinical studies [60].
Robot-Assisted Nephrectomy
Robot-assisted radical nephrectomy (RARN) was introduced in 2000 [61]; the limited benefit of that approach compared to LRN [62–65] slowed its diffusion into clinical routine and robotic assistance is considered a technical overtreatment for RNs by some authors [60]. However, Patel et al. reported that RARN represented 21 % of all minimally invasive RN performed in the state of Maryland in 2011 [34•].
Robot-assisted partial nephrectomy (RAPN), introduced in 2004 [66], represents a feasible [60] alternative to OPN or LPN [20]. According to recent data from the Nationwide Inpatient Sample (NIS), within patients treated with minimally invasive PN, RAPN is up to supplant LPN with a utilization rate of 72.5 % vs. 27.5 %, respectively (2008 and 2009) [67]. Furthermore Patel et al. reported that RAPN represented the predominant approach (76.4 %) within RCC patients treated with minimally invasive PN in the state of Maryland 2011 [34•].
However, when comparing RAPN vs. LPN, perioperative outcome seems to be equal for estimated glomerular filtration rate, estimated blood loss, transfusion rate, LOS, OP time and intra- and postoperative complications [67–84]. Whereas four studies reported about an equal warm ischaemia time (WIT) [69, 73, 75, 81], nine studies pointed out an advantage for RAPN in terms of WIT [71, 72, 74, 76, 78–80, 82, 83]. Whereas outcomes of RAPN are not clearly superior relative to LPN, the robotic approach is definitely associated with higher costs [85]. Accordingly, economic considerations, including direct and indirect costs, need to be opposed to long term oncologic and functional outcomes in order to clarify the role of RAPN in the management of T1 RCC patients.
Combining the robotic technique with LESS, the R-LESS approach was first described for urologic surgery in 2009 [86]. In a worldwide multi-institutional series of 1,076 LESS cases, R-LESS was used in 13 % of all LESS procedures for urologic surgery [59]. Although R-LESS has the potential to play a major role in LESS surgery [60], the importance for kidney surgery is currently unclear.
Natural Orifice Transluminal Endoscopic Surgery
Feasibility of NOTES was first demonstrated in 2002 by transvaginal LRN in a porcine model [87]. Whereas the initial NOTES LRN was performed in a hybrid technique with an additional umbilical trocar, the first pure NOTES LRN was reported in 2010 [88]. Although an improvement of patient outcomes was anticipated, the implementation of NOTES struggled with a lack of specific instruments and NOTES remains with limited application these days [89].
Conclusions
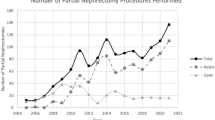
In contrast to current treatment recommendations, ORN remains the predominant treatment approach for T1 RCC and the use of LRN remains low. Despite an encouraging increase of PN rates among T1 RCC patients, we observed an overuse of RN (Figure 1), unnecessarily exposing many patients to an increased risk of renal failure. Higher rates of PN in a current series of T1 RCC patients in tertiary care centers reflect disparities in the practice pattern across the urologic community. Several sociodemographic and socioeconomic factors preclude some patients from access to standard of care treatments.
The rates of PN vs. RN (modified after [32])
Whereas alternative approaches like LPN, RAPN and LESS-PN are evolving, potential benefits relative to the current standard of care OPN need to be confirmed in well-designed prospective trials. Utilization of novel minimally invasive techniques should not undermine considerations of nephron-sparing surgery. Recognizing, that observational data are always delayed and, therefore, do not reflect current treatment patterns, the described trends need to be confirmed in future studies. Raising the awareness of the benefits of minimally invasive and nephron-sparing approaches in renal surgery as well as systematic training of urologic surgeons might represent important efforts in order to expedite the implementation of guideline recommendations.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Sun M, Becker A, Tian Z, Roghmann F, Abdollah F, Larouche A, et al. Management of Localized Kidney Cancer: Calculating Cancer-specific Mortality and Competing Risks of Death for Surgery and Nonsurgical Management. Eur Urol. 2013. doi:10.1016/j.eururo.2013.03.034.
Ng CS, Wood CG, Silverman PM, Tannir NM, Tamboli P, Sandler CM. Renal cell carcinoma: diagnosis, staging, and surveillance. AJR Am J Roentgenol. 2008;191(4):1220–32. doi:10.2214/AJR.07.3568.
Sun M, Abdollah F, Bianchi M, Trinh QD, Jeldres C, Thuret R, et al. Treatment management of small renal masses in the 21st century: a paradigm shift. Ann Surg Oncol. 2012;19(7):2380–7. doi:10.1245/s10434-012-2247-0. This pupulation based study provides an comprehensive overview on current treatment trends in patients with T1a renal cell carcinoma including non-surgical treatment.
Huang WC, Levey AS, Serio AM, Snyder M, Vickers AJ, Raj GV, et al. Chronic kidney disease after nephrectomy in patients with renal cortical tumours: a retrospective cohort study. Lancet Oncol. 2006;7(9):735–40. doi:10.1016/S1470-2045(06)70803-8.
Simmons MN, Weight CJ, Gill IS. Laparoscopic radical versus partial nephrectomy for tumors >4 cm: intermediate-term oncologic and functional outcomes. Urology. 2009;73(5):1077–82. doi:10.1016/j.urology.2008.11.059.
Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004;351(13):1296–305. doi:10.1056/NEJMoa041031.
Van Poppel H, Da Pozzo L, Albrecht W, Matveev V, Bono A, Borkowski A, et al. A prospective, randomised EORTC intergroup phase 3 study comparing the oncologic outcome of elective nephron-sparing surgery and radical nephrectomy for low-stage renal cell carcinoma. Eur Urol. 2011;59(4):543–52. doi:10.1016/j.eururo.2010.12.013.
Huang WC, Elkin EB, Levey AS, Jang TL, Russo P. Partial nephrectomy versus radical nephrectomy in patients with small renal tumors–is there a difference in mortality and cardiovascular outcomes? J Urol. 2009;181(1):55–61. doi:10.1016/j.juro.2008.09.017. discussion 61–52.
Miller DC, Schonlau M, Litwin MS, Lai J, Saigal CS, Urologic Diseases in America P. Renal and cardiovascular morbidity after partial or radical nephrectomy. Cancer. 2008;112(3):511–20. doi:10.1002/cncr.23218.
Thompson RH, Siddiqui S, Lohse CM, Leibovich BC, Russo P, Blute ML. Partial versus radical nephrectomy for 4 to 7 cm renal cortical tumors. J Urol. 2009;182(6):2601–6. doi:10.1016/j.juro.2009.08.087.
Gratzke C, Seitz M, Bayrle F, Schlenker B, Bastian PJ, Haseke N, et al. Quality of life and perioperative outcomes after retroperitoneoscopic radical nephrectomy (RN), open RN and nephron-sparing surgery in patients with renal cell carcinoma. BJU Int. 2009;104(4):470–5. doi:10.1111/j.1464-410X.2009.08439.x.
Thompson RH, Kaag M, Vickers A, Kundu S, Bernstein M, Lowrance W, et al. Contemporary use of partial nephrectomy at a tertiary care center in the United States. J Urol. 2009;181(3):993–7. doi:10.1016/j.juro.2008.11.017. This article highligths surgical treatment patterns in the management of T1 renal cell carcinoma in a North American center of excelence.
Dash A, Vickers AJ, Schachter LR, Bach AM, Snyder ME, Russo P. Comparison of outcomes in elective partial vs radical nephrectomy for clear cell renal cell carcinoma of 4–7 cm. BJU Int. 2006;97(5):939–45. doi:10.1111/j.1464-410X.2006.06060.x.
Weight CJ, Larson BT, Fergany AF, Gao T, Lane BR, Campbell SC, et al. Nephrectomy induced chronic renal insufficiency is associated with increased risk of cardiovascular death and death from any cause in patients with localized cT1b renal masses. J Urol. 2010;183(4):1317–23. doi:10.1016/j.juro.2009.12.030.
Crepel M, Jeldres C, Perrotte P, Capitanio U, Isbarn H, Shariat SF, et al. Nephron-sparing surgery is equally effective to radical nephrectomy for T1BN0M0 renal cell carcinoma: a population-based assessment. Urology. 2010;75(2):271–5. doi:10.1016/j.urology.2009.04.098.
Cadeddu JA, Ono Y, Clayman RV, Barrett PH, Janetschek G, Fentie DD, et al. Laparoscopic nephrectomy for renal cell cancer: evaluation of efficacy and safety: a multicenter experience. Urology. 1998;52(5):773–7.
Ono Y, Kinukawa T, Hattori R, Yamada S, Nishiyama N, Mizutani K, et al. Laparoscopic radical nephrectomy for renal cell carcinoma: a five-year experience. Urology. 1999;53(2):280–6.
Chan DY, Cadeddu JA, Jarrett TW, Marshall FF, Kavoussi LR. Laparoscopic radical nephrectomy: cancer control for renal cell carcinoma. J Urol. 2001;166(6):2095–9. discussion 2099–2100.
McDougall E, Clayman RV, Elashry OM. Laparoscopic radical nephrectomy for renal tumor: the Washington University experience. J Urol. 1996;155(4):1180–5.
Ljungberg B CS, Hora M, et al. (Updated 2013) EAU Guidelines on renal cell cancer. European Association of Urology Web site http://www.uroweb.org/guidelines/online-guidelines/. Updated 2013.
Campbell SC, Novick AC, Belldegrun A, Blute ML, Chow GK, Derweesh IH, et al. Guideline for management of the clinical T1 renal mass. J Urol. 2009;182(4):1271–9. doi:10.1016/j.juro.2009.07.004.
Stief CG. If a partial nephrectomy could be done safely for a renal tumor, would radical nephrectomy be considered malpractice? Eur Urol. 2011;60(3):465–6. doi:10.1016/j.eururo.2011.05.040. discussion 466–467.
Bianchi M, Becker A, Abdollah F, Trinh QD, Hansen J, Tian Z, et al. Rates of open versus laparoscopic and partial versus radical nephrectomy for T1a renal cell carcinoma: A population-based evaluation. Int J Urol. 2013. doi:10.1111/iju.12110. This article provides an overview about current treatment patterns of small renal masse in the urologic community, including all major surgical tretament options.
Miller DC, Hollingsworth JM, Hafez KS, Daignault S, Hollenbeck BK. Partial nephrectomy for small renal masses: an emerging quality of care concern? J Urol. 2006;175(3 Pt 1):853–7. doi:10.1016/S0022-5347(05)00422-2. discussion 858.
Hollenbeck BK, Taub DA, Miller DC, Dunn RL, Wei JT. National utilization trends of partial nephrectomy for renal cell carcinoma: a case of underutilization? Urology. 2006;67(2):254–9. doi:10.1016/j.urology.2005.08.050.
Baillargeon-Gagne S, Jeldres C, Lughezzani G, Sun M, Isbarn H, Capitanio U, et al. A comparative population-based analysis of the rate of partial vs radical nephrectomy for clinically localized renal cell carcinoma. BJU Int. 2010;105(3):359–64. doi:10.1111/j.1464-410X.2009.08745.x.
Sun M, Trinh QD, Bianchi M, Hansen J, Hanna N, Abdollah F, et al. A non-cancer-related survival benefit is associated with partial nephrectomy. Eur Urol. 2012;61(4):725–31. doi:10.1016/j.eururo.2011.11.047.
Zini L, Patard JJ, Capitanio U, Mejean A, Villers A, de La Taille A, et al. The use of partial nephrectomy in European tertiary care centers. Eur J Surg Oncol. 2009;35(6):636–42. doi:10.1016/j.ejso.2008.07.008.
Smaldone MC, Egleston B, Uzzo RG, Kutikov A. Does partial nephrectomy result in a durable overall survival benefit in the Medicare population? J Urol. 2012;188(6):2089–94. doi:10.1016/j.juro.2012.07.099.
Gill IS, Kavoussi LR, Lane BR, Blute ML, Babineau D, Colombo Jr JR, et al. Comparison of 1,800 laparoscopic and open partial nephrectomies for single renal tumors. J Urol. 2007;178(1):41–6. doi:10.1016/j.juro.2007.03.038.
Yang G, Villalta JD, Meng MV, Whitson JM. Evolving practice patterns for the management of small renal masses in the USA. BJU Int. 2012;110(8):1156–61. doi:10.1111/j.1464-410X.2012.10969.x. This manuscript highlights implememtation and rising importance of partial nephrectomy, tumor ablation and surveilance in T1a and T1b renal cell carcinoma.
Dulabon LM, Lowrance WT, Russo P, Huang WC. Trends in renal tumor surgery delivery within the United States. Cancer. 2010;116(10):2316–21. doi:10.1002/cncr.24965.
Small AC, Tsao CK, Moshier EL, Gartrell BA, Wisnivesky JP, Godbold J, et al. Trends and variations in utilization of nephron-sparing procedures for stage I kidney cancer in the United States. World J Urol. 2012. doi:10.1007/s00345-012-0873-6.
Patel HD, Mullins JK, Pierorazio PM, Jayram G, Cohen JE, Matlaga BR, et al. Trends in renal surgery: robotic technology is associated with increased use of partial nephrectomy. J Urol. 2013;189(4):1229–35. doi:10.1016/j.juro.2012.10.024. This well written manuscript convincingly demonstrates the benefits of robotic surgery on utilization of nephron sparing nephrectomy.
Poon SA, Silberstein JL, Chen LY, Ehdaie B, Kim PH, Russo P. Trends in partial and radical nephrectomy: an analysis of case logs from certifying urologists. J Urol. 2013;190(2):464–9. doi:10.1016/j.juro.2013.02.094.
Bolenz C, Freedland SJ, Hollenbeck BK, Lotan Y, Lowrance WT, Nelson JB, et al. Costs of Radical Prostatectomy for Prostate Cancer: A Systematic Review. Eur Urol. 2012. doi:10.1016/j.eururo.2012.08.059.
Fan X, Lin T, Xu K, Yin Z, Huang H, Dong W, et al. Laparoendoscopic single-site nephrectomy compared with conventional laparoscopic nephrectomy: a systematic review and meta-analysis of comparative studies. Eur Urol. 2012;62(4):601–12. doi:10.1016/j.eururo.2012.05.055.
Kutikov A, Uzzo RG. The R.E.N.A.L. nephrometry score: a comprehensive standardized system for quantitating renal tumor size, location and depth. J Urol. 2009;182(3):844–53. doi:10.1016/j.juro.2009.05.035.
Kutikov A, Smaldone MC, Egleston BL, Manley BJ, Canter DJ, Simhan J, et al. Anatomic features of enhancing renal masses predict malignant and high-grade pathology: a preoperative nomogram using the RENAL Nephrometry score. Eur Urol. 2011;60(2):241–8. doi:10.1016/j.eururo.2011.03.029. This article is of major importance for the standardization of reporting on renal masses and analyzes decision making process for nephron sparing surgery.
Lane BR, Gill IS. 7-year oncological outcomes after laparoscopic and open partial nephrectomy. J Urol. 2010;183(2):473–9. doi:10.1016/j.juro.2009.10.023.
Gong EM, Orvieto MA, Zorn KC, Lucioni A, Steinberg GD, Shalhav AL. Comparison of laparoscopic and open partial nephrectomy in clinical T1a renal tumors. J Endourol. 2008;22(5):953–7. doi:10.1089/end.2007.0300.
Marszalek M, Meixl H, Polajnar M, Rauchenwald M, Jeschke K, Madersbacher S. Laparoscopic and open partial nephrectomy: a matched-pair comparison of 200 patients. Eur Urol. 2009;55(5):1171–8. doi:10.1016/j.eururo.2009.01.042.
Huang WC, Donin NM. Partial nephrectomy is the standard of care for T1a kidney tumors. Urol Oncol. 2013;31(2):140–3. doi:10.1016/j.urolonc.2012.07.002.
Becker A, Roghmann F, Trinh QD, Hansen J, Tian Z, Shariat SF, et al. Sociodemographic disparities in the treatment of small renal masses. BJU Int. 2013;111(8):E274–82. doi:10.1111/bju.12111.
Kates M, Whalen MJ, Badalato GM, McKiernan JM. The effect of race and gender on the surgical management of the small renal mass. Urol Oncol. 2012. doi:10.1016/j.urolonc.2012.05.005.
Box G, Averch T, Cadeddu J, Cherullo E, Clayman R, Desai M, et al. Nomenclature of natural orifice translumenal endoscopic surgery (NOTES) and laparoendoscopic single-site surgery (LESS) procedures in urology. J Endourol. 2008;22(11):2575–81. doi:10.1089/end.2008.0471.
Desai MM, Rao PP, Aron M, Pascal-Haber G, Desai MR, Mishra S, et al. Scarless single port transumbilical nephrectomy and pyeloplasty: first clinical report. BJU Int. 2008;101(1):83–8. doi:10.1111/j.1464-410X.2007.07359.x.
White WM, Haber GP, Goel RK, Crouzet S, Stein RJ, Kaouk JH. Single-port urological surgery: single-center experience with the first 100 cases. Urology. 2009;74(4):801–4. doi:10.1016/j.urology.2009.04.030.
Raman JD, Bagrodia A, Cadeddu JA. Single-incision, umbilical laparoscopic versus conventional laparoscopic nephrectomy: a comparison of perioperative outcomes and short-term measures of convalescence. Eur Urol. 2009;55(5):1198–204. doi:10.1016/j.eururo.2008.08.019.
White MA, Autorino R, Spana G, Laydner H, Hillyer SP, Khanna R, et al. Robotic laparoendoscopic single-site radical nephrectomy: surgical technique and comparative outcomes. Eur Urol. 2011;59(5):815–22. doi:10.1016/j.eururo.2011.02.020.
Greco F, Hoda MR, Mohammed N, Springer C, Fischer K, Fornara P. Laparoendoscopic single-site and conventional laparoscopic radical nephrectomy result in equivalent surgical trauma: preliminary results of a single-centre retrospective controlled study. Eur Urol. 2012;61(5):1048–53. doi:10.1016/j.eururo.2012.01.043.
Zhang SD, Ma LL, Xiao B, Xiao CL, Huang Y, Wang GL. Efficacy and safety of transumbilical single-port laparoscopic radical nephrectomy. Beijing Da Xue Xue Bao. 2011;43(4):535–9.
Wang L, Liu B, Wu Z, Yang Q, Chen W, Xu Z, et al. A matched-pair comparison of laparoendoscopic single-site surgery and standard laparoscopic radical nephrectomy by a single urologist. J Endourol. 2012;26(6):676–81. doi:10.1089/end.2011.0161.
Seo IY, Lee JW, Rim JS. Laparoendoscopic single-site radical nephrectomy: a comparison with conventional laparoscopy. J Endourol. 2011;25(3):465–9. doi:10.1089/end.2010.0465.
Tugcu V, Ilbey YO, Mutlu B, Tasci AI. Laparoendoscopic single-site surgery versus standard laparoscopic simple nephrectomy: a prospective randomized study. J Endourol. 2010;24(8):1315–20. doi:10.1089/end.2010.0048.
Raybourn 3rd JH, Rane A, Sundaram CP. Laparoendoscopic single-site surgery for nephrectomy as a feasible alternative to traditional laparoscopy. Urology. 2010;75(1):100–3. doi:10.1016/j.urology.2009.05.028.
Park YH, Park JH, Jeong CW, Kim HH. Comparison of laparoendoscopic single-site radical nephrectomy with conventional laparoscopic radical nephrectomy for localized renal-cell carcinoma. J Endourol. 2010;24(6):997–1003. doi:10.1089/end.2009.0554.
Mir SA, Best SL, Donnally 3rd CJ, Gurbuz C, Tracy CR, Raman JD, et al. Minimally invasive nephrectomy: the influence of laparoendoscopic single-site surgery on patient selection, outcomes, and morbidity. Urology. 2011;77(3):631–4. doi:10.1016/j.urology.2010.06.063.
Kaouk JH, Autorino R, Kim FJ, Han DH, Lee SW, Yinghao S, et al. Laparoendoscopic single-site surgery in urology: worldwide multi-institutional analysis of 1076 cases. Eur Urol. 2011;60(5):998–1005. doi:10.1016/j.eururo.2011.06.002.
Merseburger AS, Herrmann TR, Shariat SF, Kyriazis I, Nagele U, Traxer O, et al. EAU guidelines on robotic and single-site surgery in urology. Eur Urol. 2013;64(2):277–91. doi:10.1016/j.eururo.2013.05.034.
Klingler DW, Hemstreet GP, Balaji KC. Feasibility of robotic radical nephrectomy–initial results of single-institution pilot study. Urology. 2005;65(6):1086–9. doi:10.1016/j.urology.2004.12.020.
Boger M, Lucas SM, Popp SC, Gardner TA, Sundaram CP. Comparison of robot-assisted nephrectomy with laparoscopic and hand-assisted laparoscopic nephrectomy. Jsls. 2010;14(3):374–80. doi:10.4293/108680810X12924466007124.
Hemal AK, Kumar A. A prospective comparison of laparoscopic and robotic radical nephrectomy for T1-2N0M0 renal cell carcinoma. World J Urol. 2009;27(1):89–94. doi:10.1007/s00345-008-0321-9.
Nazemi T, Galich A, Sterrett S, Klingler D, Smith L, Balaji KC. Radical nephrectomy performed by open, laparoscopy with or without hand-assistance or robotic methods by the same surgeon produces comparable perioperative results. Int Braz J Urol. 2006;32(1):15–22.
Rogers C, Laungani R, Krane LS, Bhandari A, Bhandari M, Menon M. Robotic nephrectomy for the treatment of benign and malignant disease. BJU Int. 2008;102(11):1660–5. doi:10.1111/j.1464-410X.2008.07895.x.
Gettman MT, Blute ML, Chow GK, Neururer R, Bartsch G, Peschel R. Robotic-assisted laparoscopic partial nephrectomy: technique and initial clinical experience with DaVinci robotic system. Urology. 2004;64(5):914–8. doi:10.1016/j.urology.2004.06.049.
Sammon JD, Karakiewicz PI, Sun M, Ravi P, Ghani KR, Jeong W, et al. Robot-assisted vs. Laparoscopic Partial Nephrectomy: utilization rates and perioperative outcomes. Int Braz J Urol. 2013;39(3):377–86. doi:10.1590/S1677-5538.IBJU.2013.03.11.
Haber GP, White WM, Crouzet S, White MA, Forest S, Autorino R, et al. Robotic versus laparoscopic partial nephrectomy: single-surgeon matched cohort study of 150 patients. Urology. 2010;76(3):754–8. doi:10.1016/j.urology.2010.03.058.
Long JA, Yakoubi R, Lee B, Guillotreau J, Autorino R, Laydner H, et al. Robotic versus laparoscopic partial nephrectomy for complex tumors: comparison of perioperative outcomes. Eur Urol. 2012;61(6):1257–62. doi:10.1016/j.eururo.2012.03.012.
Aboumarzouk OM, Stein RJ, Eyraud R, Haber GP, Chlosta PL, Somani BK, et al. Robotic versus laparoscopic partial nephrectomy: a systematic review and meta-analysis. Eur Urol. 2012;62(6):1023–33. doi:10.1016/j.eururo.2012.06.038.
Williams SB, Kacker R, Alemozaffar M, Francisco IS, Mechaber J, Wagner AA. Robotic partial nephrectomy versus laparoscopic partial nephrectomy: a single laparoscopic trained surgeon’s experience in the development of a robotic partial nephrectomy program. World J Urol. 2013;31(4):793–8. doi:10.1007/s00345-011-0648-5.
Ellison JS, Montgomery JS, Wolf Jr JS, Hafez KS, Miller DC, Weizer AZ. A matched comparison of perioperative outcomes of a single laparoscopic surgeon versus a multisurgeon robot-assisted cohort for partial nephrectomy. J Urol. 2012;188(1):45–50. doi:10.1016/j.juro.2012.02.2570.
Aron M, Koenig P, Kaouk JH, Nguyen MM, Desai MM, Gill IS. Robotic and laparoscopic partial nephrectomy: a matched-pair comparison from a high-volume centre. BJU Int. 2008;102(1):86–92. doi:10.1111/j.1464-410X.2008.07580.x.
Benway BM, Bhayani SB, Rogers CG, Dulabon LM, Patel MN, Lipkin M, et al. Robot assisted partial nephrectomy versus laparoscopic partial nephrectomy for renal tumors: a multi-institutional analysis of perioperative outcomes. J Urol. 2009;182(3):866–72. doi:10.1016/j.juro.2009.05.037.
Deane LA, Lee HJ, Box GN, Melamud O, Yee DS, Abraham JB, et al. Robotic versus standard laparoscopic partial/wedge nephrectomy: a comparison of intraoperative and perioperative results from a single institution. J Endourol. 2008;22(5):947–52. doi:10.1089/end.2007.0376.
DeLong JM, Shapiro O, Moinzadeh A. Comparison of laparoscopic versus robotic assisted partial nephrectomy: one surgeon’s initial experience. Can J Urol. 2010;17(3):5207–12.
Jeong W, Park SY, Lorenzo EI, Oh CK, Han WK, Rha KH. Laparoscopic partial nephrectomy versus robot-assisted laparoscopic partial nephrectomy. J Endourol. 2009;23(9):1457–60. doi:10.1089/end.2009.0302.
Kural AR, Atug F, Tufek I, Akpinar H. Robot-assisted partial nephrectomy versus laparoscopic partial nephrectomy: comparison of outcomes. J Endourol. 2009;23(9):1491–7. doi:10.1089/end.2009.0377.
Wang AJ, Bhayani SB. Robotic partial nephrectomy versus laparoscopic partial nephrectomy for renal cell carcinoma: single-surgeon analysis of >100 consecutive procedures. Urology. 2009;73(2):306–10. doi:10.1016/j.urology.2008.09.049.
Pierorazio PM, Patel HD, Feng T, Yohannan J, Hyams ES, Allaf ME. Robotic-assisted versus traditional laparoscopic partial nephrectomy: comparison of outcomes and evaluation of learning curve. Urology. 2011;78(4):813–9. doi:10.1016/j.urology.2011.04.065.
Seo IY, Choi H, Boldbaatr Y, Lee JW, Rim JS. Operative outcomes of robotic partial nephrectomy: a comparison with conventional laparoscopic partial nephrectomy. Korean J Urol. 2011;52(4):279–83. doi:10.4111/kju.2011.52.4.279.
Faria EF, Caputo PA, Wood CG, Karam JA, Nogueras-Gonzalez GM, Matin SF. Robotic partial nephrectomy shortens warm ischemia time, reducing suturing time kinetics even for an experienced laparoscopic surgeon: a comparative analysis. World J Urol. 2013. doi:10.1007/s00345-013-1115-2.
Panumatrassamee K, Autorino R, Laydner H, Hillyer S, Khalifeh A, Kassab A, et al. Robotic versus laparoscopic partial nephrectomy for tumor in a solitary kidney: a single institution comparative analysis. Int J Urol. 2013;20(5):484–91. doi:10.1111/j.1442-2042.2012.03205.x.
Khalifeh A, Autorino R, Hillyer SP, Laydner H, Eyraud R, Panumatrassamee K, et al. Comparative outcomes and assessment of trifecta in 500 robotic and laparoscopic partial nephrectomy cases: a single surgeon experience. J Urol. 2013;189(4):1236–42. doi:10.1016/j.juro.2012.10.021.
Yu HY, Hevelone ND, Lipsitz SR, Kowalczyk KJ, Hu JC. Use, costs and comparative effectiveness of robotic assisted, laparoscopic and open urological surgery. J Urol. 2012;187(4):1392–8. doi:10.1016/j.juro.2011.11.089.
Kaouk JH, Goel RK, Haber GP, Crouzet S, Stein RJ. Robotic single-port transumbilical surgery in humans: initial report. BJU Int. 2009;103(3):366–9. doi:10.1111/j.1464-410X.2008.07949.x.
Gettman MT, Lotan Y, Napper CA, Cadeddu JA. Transvaginal laparoscopic nephrectomy: development and feasibility in the porcine model. Urology. 2002;59(3):446–50.
Kaouk JH, Haber GP, Goel RK, Crouzet S, Brethauer S, Firoozi F, et al. Pure natural orifice translumenal endoscopic surgery (NOTES) transvaginal nephrectomy. Eur Urol. 2010;57(4):723–6. doi:10.1016/j.eururo.2009.10.027.
Kerbl DC, McDougall EM, Clayman RV, Mucksavage P. A history and evolution of laparoscopic nephrectomy: perspectives from the past and future directions in the surgical management of renal tumors. J Urol. 2011;185(3):1150–4. doi:10.1016/j.juro.2010.10.040.
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
Dr. Jonas Schiffmann, Dr. Marco Bianchi, Dr. Maxine Sun, and Dr. Andreas Becker each declare no potential conflicts of interest relevant to this article.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Kidney Diseases
Rights and permissions
About this article
Cite this article
Schiffmann, J., Bianchi, M., Sun, M. et al. Trends in Surgical Management of T1 Renal Cell Carcinoma. Curr Urol Rep 15, 383 (2014). https://doi.org/10.1007/s11934-013-0383-0
Published:
DOI: https://doi.org/10.1007/s11934-013-0383-0
Keywords
- Renal Cell Carcinoma
- Small Renal Mass
- Localized Renal Cell Carcinoma
- T1 Renal Cell Carcinoma
- Nephrectomy
- Partial Nephrectomy
- Radical Nephrectomy
- Laparoscopic Nephrectomy
- Robot-Assisted Nephrectomy
- Laparoendoscopic Single-Site Surgery
- LESS, Open Nephrectomy
- Natural Orifice Transluminal Endoscopic Surgery
- NOTES
- Laparoscopic Radical Nephrectomy
- Laparoscopic Partial Nephrectomy
- Open Radical Nephrectomy
- Open Partial Nephrectomy
- Nephron-Sparing Surgery
- NSS
- Kidney
- Robot-Assisted Single-Site Surgery
- R-LESS
- Renal Cancer