Abstract
Pathological fibrosis is a distinguishing hallmark of systemic sclerosis (SSc) as well as a number of more common conditions. Fibrosis is a complex and dynamic process associated with immune dysregulation, vasculopathy, and uncontrolled extracellular matrix production leading to intractable scar formation in the skin and internal organs. Persistent or recurrent chemical, infectious, mechanical, or autoimmune injury in genetically predisposed individuals causes sustained fibroblasts activation. Innate immune signaling via toll-like receptors (TLRs) is increasingly recognized as a key player driving the persistent fibrotic response in SSc. In particular, expression of TLR4 as well as its endogenous ligands are elevated in lesional tissue from patients with SSc. Ligand-induced TLR4 activation elicits potent stimulatory effects on fibrotic gene expression and myofibroblast differentiation. Furthermore, TLR4 appears to sensitize fibroblasts to the profibrotic stimulatory effect of transforming growth factor-β. This review highlights recent advances and emerging paradigms for understanding the regulation, complex functional roles, and therapeutic potential of TLRs in SSc pathogenesis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fibrosis is a prevalent medical concern and an unmet need. A hallmark of systemic sclerosis (SSc), fibrosis is characterized by excessive synthesis and accumulation of collagen and other extracellular matrix (ECM) molecules in skin, lungs, and other internal organs [1•]. Increased matrix stiffness, chronic oxidative stress, and extracellular accumulation of damage-associated molecular patterns (DAMPs) are recently recognized additional prominent features of fibrosis [1•, 2•, 3••, 4, 5]. Transforming growth factor-β (TGF-β) induces the full repertoire of fibrotic responses and plays a key role in pathogenesis [6]. Although multiple intracellular pathways are implicated in TGF-β-mediated fibrotic responses, cross-talk among these networks and the nature of their persistent deregulation in pathological inflammation and fibrosis remain poorly understood. Recent studies, discussed below, link innate immune signaling and toll-like receptors (TLRs) to TGF-β activity and persistent fibrotic responses.
Toll-like receptors are evolutionary conserved cellular sensors for both microbial (exogenous) pathogen-associated molecular patterns (PAMPs) and endogenous DAMPs [7]. Oxidative damage and tissue injury promote the local generation of DAMPs such as low-molecular-weight hyaluronan degradation products, alternately spliced fibronectin-extra domain A (Fn-EDA), tenascin C, and biglycan; or release of intracellular stress proteins such as high-mobility group protein-B1 (HMGB1) and heat shock protein 60 (Hsp60); and nucleic acids and immune complexes, each of which can induce cell activation via TLRs. Recent studies provide evidence for fundamental and previously unappreciated roles for TLRs and their endogenous ligands in SSc [8, 9]. Levels of both TLR4 and its endogenous ligands are elevated in SSc and elicit potent stimulatory effects on fibrotic gene expression [4, 3••]. Genetic targeting of TLR4 or its endogenous ligands ameliorates experimental fibrosis in mouse models of SSc. Mechanistic studies demonstrate that TLR4 dramatically enhanced the sensitivity of fibroblasts to the profibrotic stimulatory effect of TGF-β. Despite considerable recent progress, the pathological role of TLRs and its ligands in fibrosis and SSc still remains inadequately defined. We provide a brief overview of TLR structure, expression, and function, followed by recent insights and current understanding of the role of innate immune signaling and TLRs in fibrosis and SSc and discuss future research directions and potential therapeutic strategies targeting TLRs.
Toll-Like Receptors, Signaling, and Biological Outcomes
Toll-like receptors are transmembrane glycoproteins composed of an extracellular leucine-rich repeat extracellular domain and transmembrane and intracellular Toll/IL (interleukin)-1 receptor (TIR) domains [10, 11]. Leucine-rich repeats mediate ligand recognition and TIR domains mediate intracellular signal transduction. Toll-like receptor 4 (TLR4), an orthologue of Drosophila Toll, was the first TLR to be identified [12]. To date, 10 human and 12 mouse TLRs have been identified. While TLR1–TLR9 are conserved in both species, TLR10 is not functional in the mouse, whereas TLR11, TLR12, and TLR13 do not exist in the human genome. TLR1, TLR2, TLR4, TLR5, and TLR6 are expressed on the cell surface, whereas TLR3, TLR7/8, TLR9, and TLR11 are located in the endoplasmic reticulum (ER), endosomes, lysosomes, or endolysosomes [11].
Upon binding to exogenous PAMPs or endogenous DAMPs, TLRs dimerize and undergo the conformational changes necessary for downstream signal transduction. Activated TLRs engage two distinct intracellular pathways, myeloid differentiation factor 88 (MyD88)-dependent or the MyD88-independent. In general, all TLRs, except TLR3, recruit MyD88 through interaction with their respective TIR domains, resulting in the activation of IL-1R-associated kinases (IRAKs), IκB (inhibitor of NF-κB) kinase (IKK), and NF-κB, and secretion of inflammatory cytokines. TLR3 signals through TIR-domain-containing adaptor-inducing interferon (TRIF). TLR4 is the only TLR that engages both MyD88 and TRIF for signaling [11].
Ligands of TLRs
Both microbial PAMPs and endogenous DAMPs can serve as TLR ligands [13]. By recognizing the PAMPs (bacterial lipoprotein, lipopolysaccharide (LPS), and nucleic acid), TLRs play an essential role in host defense against microorganisms and viruses. TLR3 responds to double-stranded RNA produced during viral replication, whereas TLR7 and 8 recognize viral single-stranded RNA [11]. TLR13 in mice recognizes a conserved 23S ribosomal RNA sequence [14•]. Gram-negative bacterial LPS is recognized by TLR4. TLR9 acts as a receptor for unmethylated CpG motifs present in bacterial and viral DNA [13, 11]. The relevant ligands for TLR10 and TLR12 still remain unidentified.
The DAMPs are endogenous TLR ligands, released from cells upon necrosis or tissue injury, generated by stressed cells, or resulting from mechanical or biochemical fragmentation of ECM molecules, that serve as essential “danger signals” [7]. DAMPs enable organisms to sense, and mount a response to, tissue damage. Endogenous ligands for TLR4 include the heat shock proteins HSP60, HSP70, HSP22, and HSP72 and GP96 and HMGB1; fragmented or alternately spliced ECM molecules such as low-molecular-weight hyaluronic acid (LMW HA), fibronectin-EDA, tenascin C, and versican; and mitochondrial DNA released from dead or dying cell [3••, 4, 7]. Interestingly, the type III domain of fibronectin can be unfolded in response to mechanical forces or generated via extracellular proteolysis leading to TLR4-dependent inflammatory responses [15].
Negative Regulation of TLR Signaling
In order to prevent aberrant or sustained injurious innate immune responses, a number of “fail-safe” mechanisms evolved to negatively regulate TLR signaling [16•]. Alternately spliced TLR adaptors, ubiquitin ligases, deubiquitinases, transcriptional regulators, and microRNAs (miRNAs) [17, 13, 18] can all serve to dampen TLR signaling. The direct TLR target A20 (also known as TNFAIP3) acts as an E3 ubiquitin ligase to control NF-κB activation and TLR-mediated responses [19, 20]. In an arthritis model, A20 functions as a negative regulator of Nlrp3 inflammasome activation, and A20-KO mice develops Nlrp3-dependent spontaneous arthritis [21•]. Intriguingly, both A20/Tnfaip3 and Nlrp3 are susceptibility risk alleles for rheumatoid arthritis. The radio protective 105 gene product (RP105) negatively regulates MD2-dependent TLR4 signaling [22, 23]. RP105-deficient mice developed accelerated arthritis [24]. In contrast to murine RP105, little is known about the functional role of this molecule in humans. Other negative TLR regulators include single immunoglobulin IL-1R-related molecule (SIGGR/TIR8), ST2 (IL-33 receptor), soluble TLR9 (sTLR9) generated by cleavage of TLR9 [25], IRAK isoform IRAK-M, and suppressor of cytokine signaling-1 (SOCS-1) that suppresses IRAK activity and TLR4 signaling [16•]. The role of these negative TLR regulators in disease remains to be determined.
A Novel Role for TLRs in Organ Fibrosis Emerges
Persistence and progression of fibrosis involves feed-forward amplification loops that promote fibroblast activation and myofibroblast differentiation. As highlighted in the following section, recent studies implicate deregulated TLR expression and signaling as key factors underlying pathological fibrogenesis in liver, kidney, lungs, heart, skin, and SSc.
TLRs in Liver Fibrosis
Liver fibrosis due to hepatitis C virus (HCV) and alcoholic and nonalcoholic steatohepatitis (NASH) is mediated by the interactions between hepatic stellate cells (HSC) and immune cells, as well as by intracellular immune signaling pathways in HSCs. Innate immunity plays a key role in these processes.
In liver injury, HSC transformation into myofibroblasts and production of type 1 collagen underlie fibrogenesis. Activation of HSC can be elicited by LPS or by endogenous TLR4 ligands [26]. Elevated circulating LPS is prominent in chronic liver diseases [27]. Quiescent HSCs resist TGF-β activation due to constitutive overexpression of bone morphogenetic protein and activin membrane-bound inhibitor (BAMBI), a negative regulator of Smad-dependent TGF-β signaling [28, 29]. Upon TLR4 activation of HSC, BAMBI is downregulated, allowing unrestricted TGF-β signaling. TLR4-mutant mice are resistant to experimental hepatic fibrogenesis [29]. Mice with targeted deletion of TLR9 similarly showed reduced HSC activation and liver fibrosis [30]. Conversely, TLR3 appears to serve on anti-fibrotic function, and its deficiency in mice is associated with progression of alcohol-induced liver fibrosis [31]. These findings highlight the divergent functional roles of different TLRs in liver fibrosis.
TLRs in Pulmonary Fibrosis
In contrast to liver fibrosis, the role of TLR signaling in lung fibrosis is not well defined. Key pathological features of lung fibrosis include loss of alveolar epithelial cell integrity and interstitial fibrosis with distortion of normal tissue architecture [32•, 33]. Common etiologies include viral and bacterial infections, radiotherapy and chemotherapy, and graft versus host disease; however, in most cases, the etiology remains unidentified (idiopathic pulmonary fibrosis, IPF) [33]. A variety of recent studies implicate TLRs in the development of lung fibrosis. Functional polymorphisms in the TLR3 gene (L412F) with attenuated TLR3 function are associated with accelerated progression in IPF, consistent with an anti-fibrotic role [34•]. In contrast, elevated TLR9 expression and signaling in lung fibroblasts are associated with rapid progression of IPF [35, 36]. Furthermore, the TLR9 ligand CpG oligonucleotide (ODN) exacerbated lung fibrosis in a humanized mouse model [35]. However, other studies showed that TLR9 limited the lung fibrotic response induced by gamma herpes virus or by bleomycin [37, 38]. TLR4 plays a profibrotic role in the lung, and its downregulation by small hairpin RNA (shRNA) ameliorated LPS-induced pulmonary fibrosis in mice [39]. In contrast, TLR2/TLR4 double knockout mice showed enhanced radiation-induced lung fibrosis and reduced survival after acute lung injury [40, 41]. Another study showed that TLR4 was required for the resolution of lung inflammation and fibrosis [42]. The contributions of specific TLRs to lung fibrosis in mice and in humans merit better understanding.
Toll-Like Receptor 4 Implicated in Kidney Fibrosis
Glomerulosclerosis and tubulointerstitial fibrosis are hallmarks of chronic kidney disease [43]. Activation of TLR4 appears to play a critical role in renal fibrosis by augmenting TGF-β responses in tubular cells and in myofibroblasts via mechanisms involving downregulation of BAMBI [44]. Endogenous TLR4 ligands prominent in kidney injury include HMGB1, ECM components, and HSPs. Mice deficient in these TLR4 ligands, or in TLR4, were protected from renal fibrogenesis induced by a variety of experimental approaches [44–46]. For instance, mice deficient in TLR4, MyD88, or TRIF showed a significant reduction in fibrosis in chronic allograft nephropathy models [47].
TLRs in Myocardial Fibrosis
Cardiac injury, as well as aging, leads to remodeling, fibrosis, and stiffness that underlie ventricular dysfunction and heart failure [48]. Common triggers include ischemia, pressure overload, and viral infection. A role for innate immunity in cardiac fibrosis, particularly TLR2 and TLR4, is emerging. Recurrent exposure to LPS is, by itself, sufficient to induce cardiac fibrosis [49]. Mice with targeted deletion of TLR4 or TLR2 showed improved ventricular function, reduced remodeling, and collagen accumulation following myocardial infarction [50–53] In contrast, synthetic ligands of TLR9 (1668-thioate or 1612-thioate) were shown to attenuate cardiac hypertrophy and fibrosis following transverse aortic constriction [54]. Endogenous damage-associated TLR ligands potentially implicated in myocardial fibrosis include fibronectin-EDA, HSP60, HMGB1, tenascin C, galectin-3, S100A8, S100A9, and mitochondrial DNA [55].
TLRs in Skin Fibrosis
Skin fibrosis is a prominent feature of keloids, nephrogenic systemic fibrosis (NSF), morphea, and hypertrophic scars. Keloids are benign fibroproliferative tumors commonly occurring following trauma to the skin. A recent study indicated increased expression of TLR6, 7, and 8 in keloid lesions [56]. Nephrogenic systemic fibrosis occurs in individuals with advanced kidney failure exposed to gadolinium contrast agents (GdBCA). In human macrophages, GdBCA was shown to activate TLR4 and TLR7, resulting in production of proinflammatory and profibrotic cytokines and growth factors. These findings suggest that the environmental stimuli participate in the pathogenesis of NSF and other cutaneous fibrotic disorders via TLRs signaling [57]. Hypertrophic scars occur following dermal damage by thermal injury or other forms of trauma. A recent study showed that levels of TLR4, MyD88, and proinflammatory cytokines were all elevated in fibroblasts from hypertrophic scars [58•].
Innate Immune Activation and TLRs in SSc
Genetic Polymorphisms in the TLR Signaling Axis in SSc
Genome-wide association studies (GWAS) in SSc reveal genetic associations with several innate immune signaling variants [59, 60•] (Table 1). A rare functional polymorphism in the TLR2 gene (Pro631His) showed robust association with both anti-topoisomerase antibody-positive SSc and pulmonary arterial hypertension [61]. Other studies identified genes for interferon regulatory factors (IRF5, IRF7, and IRF8) as risk factors for limited cutaneous SSc [62]. Of great interest, A20 or TNFAIP3, a negative regulator of TLR signaling, and (TNFAIP3)-interacting protein 1 (TNIP1) both showed strong association with SSc [63, 64]. Furthermore, expression of TNFAIP3 messenger RNA (mRNA) was significantly reduced in carriers of the rs117480515 allele compared to noncarriers of the allele among SSc patients. Levels of TNIP1 mRNA and protein were markedly reduced in lesional skin as well as in explanted fibroblasts from SSc patients. TNIP1 abrogated fibrotic responses in activated healthy and constitutively active SSc dermal fibroblasts [65]. The consistent genetic associations of TLR signaling variants with SSc, and with specific endophenotype, together with emerging functional studies, suggests that the TLR pathways may play a role in pathogenesis.
Emerging Role of TLRs in SSc Pathogenesis
Production of type I interferon (IFNα and β) is closely linked to TLR-mediated innate immune signaling [66, 67]. Dysregulated expression of type I IFN-inducible genes in peripheral blood cells is seen in SSc [68]. Moreover, sera from patients with anti-topo I-positive SSc patients induce IFN-α production in normal peripheral blood mononuclear cells in vitro [69]. Serum containing other SSc autoantibodies failed to induce this response. In response to TLR8 ligands, peripheral blood monocytes from SSc patients showed enhanced production of tissue inhibitor of metalloproteinase 1 (TIMP-1), a profibrotic factor [70]. Stimulation of dendritic cells from SSc patients showed increased sensitivity to TLR ligands compared to that from healthy controls [71].
Evaluation of SSc skin biopsies reveals elevated expression of TLR3 primarily localized to fibroblast-like dermal cells [72, 73]. In normal skin fibroblasts, TLR3 stimulation by poly(IC) induced dose- and time-dependent increase in IFN-β production and expression of IFN-regulated genes [73]. In contrast, the expression of fibrotic genes in these cells was inhibited. Viral infection of nonimmune cells might cause persistent tissue injury and chronic inflammation and fibrosis in SSc. A recent study described striking accumulation of Epstein-Barr virus (EBV) in dermal fibroblasts and endothelial cells of SSc skin biopsies [74•]. Infection of stromal cells with EBV in vitro induced activation of endosomal TLRs that was accompanied by fibroblast-myofibroblast transdifferentiation and increased profibrotic gene expressions [74•]. These findings suggest that chronic EBV infection or reactivation in SSc may trigger sustained fibroblasts activation and fibrosis. Chronic subcutaneous injection of a synthetic TLR3 ligand in mice induced dermal inflammation and expression of IFN-regulated and fibrotic genes [75].
TLR4 Signaling in SSc Fibrosis
Recent studies further elucidate the role of TLR4 signaling in SSc. Expression of TLR4 was elevated in SSc skin and lung biopsies [4]. Strong TLR4 immunostaining was seen in a large percentage of fibroblasts and vascular cells within lesional tissues. Another report showed that both TLR4 along with its co-receptors MD2 and CD14 are overexpressed in lesional skin [76••]. Importantly, TLR4 levels correlate with progressive skin disease. Some patients with SSc were found to have circulating anti-fibroblast antibodies that were functional and could elicit TLR4-mediated inflammatory and profibrotic responses in vitro [77].
Other ligands of TLR4 also have profibrotic effects. In explanted skin fibroblasts, LPS induced TLR4-dependent increase in collagen synthesis and dramatically enhanced sensitivity to the stimulatory effects of TGF-β [4]. These responses involved downregulation of BAMBI, resulting in the augmented intensity of canonical Smad signaling. In addition, LPS also caused TLR4-dependent downregulation of anti-fibrotic miR-29. Therefore, multiple mechanisms in SSc including enhanced Smad signaling with downregulation of BAMBI and suppression of miR-29 might underlie these profibrotic effects of TLR4. In mouse, chronic LPS exposure augmented the production of proinflammatory chemokines and upregulation of TGF-β-regulated genes in the dermis [76••]. On the other hand, skin fibrosis induced by bleomycin was attenuated in mice harboring a nonfunctional mutant TLR4 [4].
Further evidence linking TLR4 with fibrosis in SSc comes from whole genome-wide transcriptional profiling studies. Previous gene expression studies analysis stratified SSc biopsies into four distinct intrinsic subsets (fibroproliferative, inflammatory, limited, and normal-like groups) [78]. An experimentally derived fibroblasts “LPS-regulated gene signature” was generated using skin fibroblasts incubated with LPS. This “LPS-regulated gene signature” is present in SSc biopsies clustering within inflammatory subset (Bhattacharyya S. and Varga J., unpublished data). Therefore, this SSc might be optimal responders to therapeutic strategies to block TLR4.
Alternately Spliced Fibronectins (Fn-EDA): a Damage-Associated Endogenous TLR4 Ligand With Key Roles in SSc
Using an immunohistochemistry-based survey, we found that several endogenous TLR4 ligands are elevated in SSc skin (Table 2). Most significant among these are low-molecular-weight hyaluronic acid (LMW HA), tenascin C, and, of special interest, fibronectin-EDA [4, 3••].
Fibronectins are high-molecular-weight glycoproteins present in soluble form in plasma or accumulating in tissue as insoluble ECM components [79]. Due to alternate splicing of the fibronectin gene, cellular fibronectin contains two extra domains A (EDA and EDB) which are excluded from plasma fibronectin [79]. Although there is little tissue fibronectin-EDA in adults, marked upregulation occurs during injury. Levels of fibronectin-EDA were found to be significantly elevated in both serum and lesional skin from SSc patients [3].
In vitro, Fn-EDA expression could be induced by treatment with TGF-β in normal fibroblasts in culture. In contrast, in SSc fibroblasts, synthesis of Fn-EDA was constitutively upregulated (Fig. 1a). Treatment of normal fibroblasts with Fn-EDA stimulated collagen production, myofibroblast differentiation, and wound healing. All these responses were abrogated by selective disruption of TLR4 signaling. Moreover, mice with targeted deletion of Fn-EDA showed attenuated skin fibrosis when challenged with bleomycin or with TGF-β [3••]. These observations therefore implicate an endogenous Fn-EDA-TLR4 signaling axis in fibroblasts in the pathogenesis of cutaneous fibrosis in SSc.
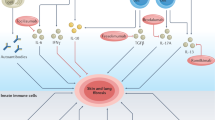
a Fn-EDA-TLR4-mediated fibroblast activation. TGF-β generates extracellular accumulation of Fn-EDA that in turn triggers TLR4-dependent cellular signaling, resulting in profibrotic responses. b Vicious cycle of fibrogenesis. Toll-like receptor signaling switches self-limited repair into sustained fibrogenesis. Chronic injury leads to tissue damage, and generation and accumulation of endogenous DAMPs leading to ongoing activation of TLR4 in fibroblasts and progressive fibrosis
Summary and Perspectives
This review highlights emerging evidence implicating innate immune signaling in persistent fibroblast activation underlying fibrosis in a variety of chronic diseases. Based on these observations, we propose that TLR4 represents a molecular switch that, in response to endogenous damage-associated TLR ligands, convert a self-limited tissue repair process into persistent and progressive pathological fibrogenesis (Fig. 1b).
Although a growing body of evidence supports a key role for DAMPs and TLRs in driving pathological fibrosis, major questions remain. While GWAS provide evidence linking SSc with gene variants implicated in TLRs signaling, functional studies are necessary to elucidate the biological mechanisms by which associated variants contribute to the disease process. Even though multiple endogenous TLR ligands are elevated in SSc skin and lung and their deficiency in mice confers resistance to fibrosis, identification of specific TLR ligands that are most important in SSc, and the relevant target cell populations, is lacking. Moreover, the initial triggers eliciting prolonged injury, the unique and redundant profibrotic functions of the various TLRs and their co-receptors, and the regulation of their function by endogenous inhibitors similarly remain uncertain.
Nevertheless, in light of the central role that TLR4 appears to play in SSc, disrupting sustained TLR4 signaling at the level of ligand or receptor level represent novel potential strategies for therapy [80•]. As TLRs are in the frontline of anti-microbial innate immunity, the major challenge is to dampen overactive TLR-dependent immune signaling without impairing normal host defense. Promising approaches to target TLR4 include selectively blocking intracellular signaling or disrupting ligand-TLR4 receptor or co-receptor complex formation (Bhattacharyya S and Varga J, unpublished). As fibrosis in SSc currently has no effective therapy, intense investigation of the regulation, expression, and function of TLRs in fibroblasts and other cells may yield progress toward development of urgently needed therapies.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Bhattacharyya S, Wei J, Varga J. Understanding fibrosis in systemic sclerosis: shifting paradigms, emerging opportunities. Nat Rev Rheumatol. 2012;8(1):42–54.
Friedman SL, Sheppard D, Duffield JS, Violette S. Therapy for fibrotic diseases: nearing the starting line. Sci Transl Med. 2013;5(167):167sr1. This update highlighted evolving understanding of how tissue injury and repair lead to fibrosis and present promising new approaches for diagnosis and treatment of fibrotic diseases.
Bhattacharyya S, Tamaki Z, Wang W, Hinchcliff M, Hoover P, Getsios S, et al. FibronectinEDA promotes chronic cutaneous fibrosis through toll-like receptor signaling. Sci Transl Med. 2014;6(232):232ra50. This study identifies alternately spliced fibronectin (Fn-EDA) as an endogenous ligand for TLR4 that is markedly elevated in lesional skin in SSc and is responsible for driving persistent myofibroblasts differentiation and progression of fibrosis.
Bhattacharyya S, Kelley K, Melichian DS, Tamaki Z, Fang F, Su Y, et al. Toll-like receptor 4 signaling augments transforming growth factor-beta responses: a novel mechanism for maintaining and amplifying fibrosis in scleroderma. Am J Pathol. 2013;182(1):192–205.
Bhattacharyya S, Wei J, Tourtellotte WG, Hinchcliff M, Gottardi CG, Varga J. Fibrosis in systemic sclerosis: common and unique pathobiology. Fibrogenesis Tissue Repair. 2012;5(Suppl 1 Proceedings of Fibroproliferative disorders: from biochemical analysis to targeted therapiesPetro E Petrides and David Brenner):S18.
Varga J, Pasche B. Transforming growth factor beta as a therapeutic target in systemic sclerosis. Nat Rev Rheumatol. 2009;5(4):200–6.
Piccinini AM, Midwood KS. DAMPening inflammation by modulating TLR signalling. Mediat Inflamm. 2010;2010.
Ciechomska M, Cant R, Finnigan J, van Laar JM, O’Reilly S. Role of toll-like receptors in systemic sclerosis. Expert Rev Mol Med. 2013;15:e9.
Lafyatis R, Farina A. New insights into the mechanisms of innate immune receptor signalling in fibrosis. Open Rheumatol J. 2012;6:72–9.
Medzhitov R. Toll-like receptors and innate immunity. Nat Rev Immunol. 2001;1(2):135–45.
Kawai T, Akira S. Toll-like receptors and their crosstalk with other innate receptors in infection and immunity. Immunity. 2011;34(5):637–50.
Gay NJ, Keith FJ. Drosophila toll and IL-1 receptor. Nature. 1991;351(6325):355–6.
Kawai T, Akira S. The role of pattern-recognition receptors in innate immunity: update on toll-like receptors. Nat Immunol. 2010;11(5):373–84.
Oldenburg M, Kruger A, Ferstl R, Kaufmann A, Nees G, Sigmund A, et al. TLR13 recognizes bacterial 23S rRNA devoid of erythromycin resistance-forming modification. Science. 2012;337(6098):1111–5. This study unraveled an unanticipated link between antibiotic resistance and evasion from TLR13 recognition, due to 23S rRNA modifications by methylation, camouflaging bacteria from TLR13 recognition.
Zheng M, Jones DM, Horzempa C, Prasad A, McKeown-Longo PJ. The first type III domain of fibronectin is associated with the expression of cytokines within the lung tumor microenvironment. J Cancer Educ. 2011;2:478–83.
Kondo T, Kawai T, Akira S. Dissecting negative regulation of toll-like receptor signaling. Trends Immunol. 2012;33(9):449–58. This review highlighted recent insights into the negative regulation of TLR signaling and innate immunity, underscoring how pathogens can negatively target TLR signaling as a strategy to evade the host immune response.
Sheedy FJ, Palsson-McDermott E, Hennessy EJ, Martin C, O’Leary JJ, Ruan Q, et al. Negative regulation of TLR4 via targeting of the proinflammatory tumor suppressor PDCD4 by the microRNA miR-21. Nat Immunol. 2010;11(2):141–7.
Quinn SR, O’Neill LA. A trio of microRNAs that control toll-like receptor signalling. Int Immunol. 2011;23(7):421–5.
Boone DL, Turer EE, Lee EG, Ahmad RC, Wheeler MT, Tsui C, et al. The ubiquitin-modifying enzyme A20 is required for termination of toll-like receptor responses. Nat Immunol. 2004;5(10):1052–60.
Wertz IE, O’Rourke KM, Zhou H, Eby M, Aravind L, Seshagiri S, et al. De-ubiquitination and ubiquitin ligase domains of A20 downregulate NF-kappaB signalling. Nature. 2004;430(7000):694–9.
Vande Walle L, Van Opdenbosch N, Jacques P, Fossoul A, Verheugen E, Vogel P, et al. Negative regulation of the NLRP3 inflammasome by A20 protects against arthritis. Nature. 2014;512(7512):69–73. This study revealed A20 as a novel negative regulator of Nlrp3 inflammasome activation and describe A20myel-KO mice as an experimental model to study the role of inflammasomes in rheumatoid arthritis.
Divanovic S, Trompette A, Atabani SF, Madan R, Golenbock DT, Visintin A, et al. Negative regulation of toll-like receptor 4 signaling by the toll-like receptor homolog RP105. Nat Immunol. 2005;6(6):571–8.
Divanovic S, Trompette A, Petiniot LK, Allen JL, Flick LM, Belkaid Y, et al. Regulation of TLR4 signaling and the host interface with pathogens and danger: the role of RP105. J Leukoc Biol. 2007;82(2):265–71.
Tada Y, Koarada S, Morito F, Mitamura M, Inoue H, Suematsu R, et al. Toll-like receptor homolog RP105 modulates the antigen-presenting cell function and regulates the development of collagen-induced arthritis. Arthritis Res Ther. 2008;10(5):R121.
Chockalingam A, Cameron JL, Brooks JC, Leifer CA. Negative regulation of signaling by a soluble form of toll-like receptor 9. Eur J Immunol. 2011;41(8):2176–84.
Seki E, Schwabe RF. Hepatic inflammation and fibrosis: functional links and key pathways. Hepatology. 2014.
Schwabe RF, Seki E, Brenner DA. Toll-like receptor signaling in the liver. Gastroenterology. 2006;130(6):1886–900.
Liu C, Chen X, Yang L, Kisseleva T, Brenner DA, Seki E. Transcriptional repression of the transforming growth factor beta (TGF-beta) pseudoreceptor BMP and activin membrane-bound inhibitor (BAMBI) by nuclear factor kappaB (NF-kappaB) p50 enhances TGF-beta signaling in hepatic stellate cells. J Biol Chem. 2014;289(10):7082–91.
Seki E, De Minicis S, Osterreicher CH, Kluwe J, Osawa Y, Brenner DA, et al. TLR4 enhances TGF-beta signaling and hepatic fibrosis. Nat Med. 2007;13(11):1324–32.
Duffield JS, Forbes SJ, Constandinou CM, Clay S, Partolina M, Vuthoori S, et al. Selective depletion of macrophages reveals distinct, opposing roles during liver injury and repair. J Clin Invest. 2005;115(1):56–65.
Jeong WI, Park O, Gao B. Abrogation of the antifibrotic effects of natural killer cells/interferon-gamma contributes to alcohol acceleration of liver fibrosis. Gastroenterology. 2008;134(1):248–58.
Herzog EL, Mathur A, Tager AM, Feghali-Bostwick C, Schneider F, Varga J. Review: interstitial lung disease associated with systemic sclerosis and idiopathic pulmonary fibrosis: how similar and distinct? Arthritis Rheumatol. 2014;66(8):1967–78. This review compared and contrasted the features of SSc-associated interstitial lung disease (ILD) and IPF, with significant implications for diagnosis, evaluation, and management.
Selman M, Pardo A. Revealing the pathogenic and aging-related mechanisms of the enigmatic idiopathic pulmonary fibrosis an integral model. Am J Respir Crit Care Med. 2014;189(10):1161–72.
O’Dwyer DN, Armstrong ME, Trujillo G, Cooke G, Keane MP, Fallon PG, et al. The toll-like receptor 3 L412F polymorphism and disease progression in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2013;188(12):1442–50. This study identified the role of a single nucleotide polymorphism in TLR3 that impairs TLR3 function and promotes progression of IPF.
Trujillo G, Meneghin A, Flaherty KR, Sholl LM, Myers JL, Kazerooni EA, et al. TLR9 differentiates rapidly from slowly progressing forms of idiopathic pulmonary fibrosis. Sci Transl Med. 2010;2(57):57ra82.
Hogaboam CM, Trujillo G, Martinez FJ. Aberrant innate immune sensing leads to the rapid progression of idiopathic pulmonary fibrosis. Fibrogenesis Tissue Repair. 2012;5(Suppl 1 Proceedings of Fibroproliferative disorders: from biochemical analysis to targeted therapiesPetro E Petrides and David Brenner):S3.
Luckhardt TR, Coomes SM, Trujillo G, Stoolman JS, Vannella KM, Bhan U, et al. TLR9-induced interferon beta is associated with protection from gammaherpesvirus-induced exacerbation of lung fibrosis. Fibrogenesis Tissue Repair. 2011;4:18.
Vannella KM, Luckhardt TR, Wilke CA, van Dyk LF, Toews GB, Moore BB. Latent herpesvirus infection augments experimental pulmonary fibrosis. Am J Respir Crit Care Med. 2010;181(5):465–77.
He Z, Zhu Y, Jiang H. Inhibiting toll-like receptor 4 signaling ameliorates pulmonary fibrosis during acute lung injury induced by lipopolysaccharide: an experimental study. Respir Res. 2009;10:126.
Paun A, Fox J, Balloy V, Chignard M, Qureshi ST, Haston CK. Combined Tlr2 and Tlr4 deficiency increases radiation-induced pulmonary fibrosis in mice. Int J Radiat Oncol Biol Phys. 2010;77(4):1198–205.
Jiang D, Liang J, Fan J, Yu S, Chen S, Luo Y, et al. Regulation of lung injury and repair by toll-like receptors and hyaluronan. Nat Med. 2005;11(11):1173–9.
Yang HZ, Wang JP, Mi S, Liu HZ, Cui B, Yan HM, et al. TLR4 activity is required in the resolution of pulmonary inflammation and fibrosis after acute and chronic lung injury. Am J Pathol. 2012;180(1):275–92.
Duffield JS. Cellular and molecular mechanisms in kidney fibrosis. J Clin Invest. 2014;124(6):2299–306.
Pulskens WP, Rampanelli E, Teske GJ, Butter LM, Claessen N, Luirink IK, et al. TLR4 promotes fibrosis but attenuates tubular damage in progressive renal injury. J Am Soc Nephrol: JASN. 2010;21(8):1299–308.
Jialal I, Major AM, Devaraj S. Global toll-like receptor 4 knockout results in decreased renal inflammation, fibrosis and podocytopathy. J. Diabetes Complicat. 2014.
Campbell MT, Hile KL, Zhang H, Asanuma H, Vanderbrink BA, Rink RR, et al. Toll-like receptor 4: a novel signaling pathway during renal fibrogenesis. J Surg Res. 2011;168(1):e61–9.
Wang S, Schmaderer C, Kiss E, Schmidt C, Bonrouhi M, Porubsky S, et al. Recipient Toll-like receptors contribute to chronic graft dysfunction by both MyD88- and TRIF-dependent signaling. Dis Model Mech. 2010;3(1–2):92–103.
Vilahur G, Juan-Babot O, Pena E, Onate B, Casani L, Badimon L. Molecular and cellular mechanisms involved in cardiac remodeling after acute myocardial infarction. J Mol Cell Cardiol. 2011;50(3):522–33.
Lew WY, Bayna E, Molle ED, Dalton ND, Lai NC, Bhargava V, et al. Recurrent exposure to subclinical lipopolysaccharide increases mortality and induces cardiac fibrosis in mice. PLoS One. 2013;8(4):e61057.
Riad A, Jager S, Sobirey M, Escher F, Yaulema-Riss A, Westermann D, et al. Toll-like receptor-4 modulates survival by induction of left ventricular remodeling after myocardial infarction in mice. J Immunol. 2008;180(10):6954–61.
Dong RQ, Wang ZF, Zhao C, Gu HR, Hu ZW, Xie J, et al. Toll-like receptor 4 knockout protects against isoproterenol-induced cardiac fibrosis: the role of autophagy. J Cardiovasc Pharmacol Ther. 2014.
Wang L, Li YL, Zhang CC, Cui W, Wang X, Xia Y, et al. Inhibition of Toll-like receptor 2 reduces cardiac fibrosis by attenuating macrophage-mediated inflammation. Cardiovasc Res. 2014;101(3):383–92.
Arslan F, de Kleijn DP, Pasterkamp G. Innate immune signaling in cardiac ischemia. Nat Rev Cardiol. 2011;8(5):292–300.
Velten M, Duerr GD, Pessies T, Schild J, Lohner R, Mersmann J, et al. Priming with synthetic oligonucleotides attenuates pressure overload-induced inflammation and cardiac hypertrophy in mice. Cardiovasc Res. 2012;96(3):422–32.
de Haan JJ, Smeets MB, Pasterkamp G, Arslan F. Danger signals in the initiation of the inflammatory response after myocardial infarction. Mediat Inflamm. 2013;2013:206039.
Bagabir RA, Syed F, Rautemaa R, McGrouther DA, Paus R, Bayat A. Upregulation of toll-like receptors (TLRs) 6, 7, and 8 in keloid scars. J Investig Dermatol. 2011;131(10):2128–30.
Wermuth PJ, Jimenez SA. Gadolinium compounds signaling through TLR4 and TLR7 in normal human macrophages: establishment of a proinflammatory phenotype and implications for the pathogenesis of nephrogenic systemic fibrosis. J Immunol. 2012;189(1):318–27.
Wang J, Hori K, Ding J, Huang Y, Kwan P, Ladak A, et al. Toll-like receptors expressed by dermal fibroblasts contribute to hypertrophic scarring. J Cell Physiol. 2011;226(5):1265–73. This study implicates TLR on dermal fibroblasts as contributory to fibrosis in hypertrophic scarring.
Mayes MD. The genetics of scleroderma: looking into the postgenomic era. Curr Opin Rheumatol. 2012;24(6):677–84.
Assassi S, Radstake TR, Mayes MD, Martin J. Genetics of scleroderma: implications for personalized medicine? BMC Med. 2013;11:9. This review highlighted the implication of recent discoveries in SSc genetics for drug development and identification of predictive biomarkers.
Broen JC, Bossini-Castillo L, van Bon L, Vonk MC, Knaapen H, Beretta L, et al. A rare polymorphism in the gene for toll-like receptor 2 is associated with systemic sclerosis phenotype and increases the production of inflammatory mediators. Arthritis Rheum. 2012;64(1):264–71.
Wu M, Assassi S. The role of type 1 interferon in systemic sclerosis. Front Immunol. 2013;4:266.
Koumakis E, Giraud M, Dieude P, Cohignac V, Cuomo G, Airo P, et al. Brief report: candidate gene study in systemic sclerosis identifies a rare and functional variant of the TNFAIP3 locus as a risk factor for polyautoimmunity. Arthritis Rheum. 2012;64(8):2746–52.
Bossini-Castillo L, Martin JE, Broen J, Simeon CP, Beretta L, Gorlova OY, et al. Confirmation of TNIP1 but not RHOB and PSORS1C1 as systemic sclerosis risk factors in a large independent replication study. Ann Rheum Dis. 2013;72(4):602–7.
Allanore Y, Saad M, Dieude P, Avouac J, Distler JH, Amouyel P, et al. Genome-wide scan identifies TNIP1, PSORS1C1, and RHOB as novel risk loci for systemic sclerosis. PLoS Genet. 2011;7(7):e1002091.
Tan FK, Zhou X, Mayes MD, Gourh P, Guo X, Marcum C, et al. Signatures of differentially regulated interferon gene expression and vasculotrophism in the peripheral blood cells of systemic sclerosis patients. Rheumatology. 2006;45(6):694–702.
York MR, Nagai T, Mangini AJ, Lemaire R, van Seventer JM, Lafyatis R. A macrophage marker, Siglec-1, is increased on circulating monocytes in patients with systemic sclerosis and induced by type I interferons and toll-like receptor agonists. Arthritis Rheum. 2007;56(3):1010–20.
Bos CL, van Baarsen LG, Timmer TC, Overbeek MJ, Basoski NM, Rustenburg F, et al. Molecular subtypes of systemic sclerosis in association with anti-centromere antibodies and digital ulcers. Genes Immun. 2009;10(3):210–8.
Kim D, Peck A, Santer D, Patole P, Schwartz SM, Molitor JA, et al. Induction of interferon-alpha by scleroderma sera containing autoantibodies to topoisomerase I: association of higher interferon-alpha activity with lung fibrosis. Arthritis Rheum. 2008;58(7):2163–73.
Ciechomska M, Huigens CA, Hugle T, Stanly T, Gessner A, Griffiths B, et al. Toll-like receptor-mediated, enhanced production of profibrotic TIMP-1 in monocytes from patients with systemic sclerosis: role of serum factors. Ann Rheum Dis. 2013;72(8):1382–9.
van Bon L, Popa C, Huijbens R, Vonk M, York M, Simms R, et al. Distinct evolution of TLR-mediated dendritic cell cytokine secretion in patients with limited and diffuse cutaneous systemic sclerosis. Ann Rheum Dis. 2010;69(8):1539–47.
Agarwal SK, Wu M, Livingston CK, Parks DH, Mayes MD, Arnett FC, et al. Toll-like receptor 3 upregulation by type I interferon in healthy and scleroderma dermal fibroblasts. Arthritis Res Ther. 2011;13(1):R3.
Fang F, Ooka K, Sun X, Shah R, Bhattacharyya S, Wei J, et al. A synthetic TLR3 ligand mitigates profibrotic fibroblast responses by inducing autocrine IFN signaling. J Immunol. 2013;191(6):2956–66.
Farina A, Cirone M, York M, Lenna S, Padilla C, McLaughlin S, et al. Epstein-Barr virus infection induces aberrant TLR activation pathway and fibroblast-myofibroblast conversion in scleroderma. J Investig Dermatol. 2014;134(4):954–64. These findings demonstrated that EBV infection in mesenchymal, endothelial, and immune cells in the skin of SSc patients may underlie fibrotic and vascular complications mediated via TLR signaling.
Farina GA, York MR, Di Marzio M, Collins CA, Meller S, Homey B, et al. Poly(I:C) drives type I IFN- and TGFbeta-mediated inflammation and dermal fibrosis simulating altered gene expression in systemic sclerosis. J Investig Dermatol. 2010;130(11):2583–93.
Stifano G, Affandi AJ, Mathes AL, Rice LM, Nakerakanti S, Nazari B, et al. Chronic toll-like receptor 4 stimulation in skin induces inflammation, macrophage activation, transforming growth factor beta signature gene expression, and fibrosis. Arthritis Res Ther. 2014;16(4):R136. This study showed how chronic TLR4 activation in the skin cells can enhance TGF-β signaling in mice, providing a potential mechanism for TLR4/myeloid differentiation factor 88 (MyD88)-dependent fibrosis.
Fineschi S, Goffin L, Rezzonico R, Cozzi F, Dayer JM, Meroni PL, et al. Antifibroblast antibodies in systemic sclerosis induce fibroblasts to produce profibrotic chemokines, with partial exploitation of toll-like receptor 4. Arthritis Rheum. 2008;58(12):3913–23.
Milano A, Pendergrass SA, Sargent JL, George LK, McCalmont TH, Connolly MK, et al. Molecular subsets in the gene expression signatures of scleroderma skin. PLoS One. 2008;3(7):e2696.
White ES, Baralle FE, Muro AF. New insights into form and function of fibronectin splice variants. J Pathol. 2008;216(1):1–14.
Li J, Wang X, Zhang F, Yin H. Toll-like receptors as therapeutic targets for autoimmune connective tissue diseases. Pharmacol Ther. 2013;138(3):441–51. This review highlighted modulating TLR signaling as an important strategy for the treatment of connective tissue diseases.
Compliance with Ethics Guidelines
Conflict of Interest
Swati Bhattacharyya declares no conflict of interest.
John Varga reports the receipt of grants from the NIH and Takeda, as well as a pending patent on a TLR4 inhibitor.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Scleroderma
Rights and permissions
About this article
Cite this article
Bhattacharyya, S., Varga, J. Emerging Roles of Innate Immune Signaling and Toll-Like Receptors in Fibrosis and Systemic Sclerosis. Curr Rheumatol Rep 17, 2 (2015). https://doi.org/10.1007/s11926-014-0474-z
Published:
DOI: https://doi.org/10.1007/s11926-014-0474-z