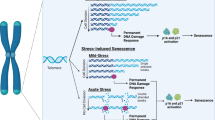
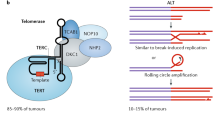
Abstract
What began as a study of the "end-replication problem" took on a new dimension as it became clear that telomeres are a "molecular clock" of replication in human somatic cells. Here we review the biology of telomeres in vitro and in vivo, in mice and humans. We suggest that, in humans, telomeres are involved in the biology of aging and pathobiology of disorders of aging, including cancer and cardiovascular disease. We also propose that the underlying dynamics of telomere biology is in line with broad principles of evolutionary theories.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References and Recommended Reading
Kirkwood TB, Austad SN: Why do we age? Nature 2000, 408:233–238.
Finkel T, Holbrook NJ: Oxidants, oxidative stress and the biology of aging. Nature 2000, 408:239–247. Review of the roles of ROS and defense mechanisms against ROS in the biology of aging.
DePinho RA: The age of cancer. Nature 2000, 408:248–254. Review of the links between aging and cancer in mice and humans, including the role of telomere dysfunction in cancer.
Guarente L, Kenyon C: Genetic pathways that regulate aging in model organisms. Nature 2000, 408:255–262.
Martin GM, Oshima J: Lessons from human progeroid syndromes. Nature 2000, 408:263–266.
Hayflick L: The future of ageing. Nature 2000, 408:267–269.
Medwar PB: An Unsolved Problem in Biology. The Uniqueness of the Individual. New York: Dover; 1952.
Williams GC: Pleitrophy, natural selection and the evolution of senescence. Evolutions 1957, 11:398–411.
Jazwinski SM: Longevity, genes and aging. Science 1996, 273:54–59.
Finch CE, Kirkwood TBL: Chance, Development and Aging. New York, Oxford: Oxford Univesity Press; 2000. Tour de force account of the biology of aging, with numerous examples and illustrations.
Kirkwood TB, Holliday R: The evolution of ageing and longevity. Proc R Soc Lond B Biol Sci 1979, 205:531–546.
Kirkwood TB, Rose MR: Evolution of senescence: late survival sacrificed for reproduction. Philos Trans R Soc Lond B Biol Sci 1991, 332:15–24.
Holliday R: The evolution of human longevity. Perspect Biol Med 1996, 40:100–107.
Blackburn EH: Structure and function of telomeres. Nature 1991, 350:569–573.
Olovnikov AM: A theory of marginotomy: the incomplete copying of template margin in enzymic synthesis of pronucleotides and biological significance of the phenomenon. J Theor Biol 1973, 41:181–190.
Watson JD: Origin of concatemeric T7 DNA. Nature New Biol 1972, 239:197–201.
Levy MZ, Allsopp RC, Futcher AB, et al.: Telomere end-replication problem and cell aging. J Mol Biol 1992, 225:951–960.
Greider CW, Blackburn EH: Identification of a specific telomere terminal transferase enzyme with two kinds of primer specificity. Cell 1985, 51:405–413.
Morin GB: The human telomere terminal transferase enzyme is a ribonucleoprotein that synthesizes TTAGGG repeats. Cell 1989, 59:521–529.
Nakamura TM, Morin GB, Chapman KB, et al.: Telomerase catalytic subunit homologs from fission yeast and human. Science 1997, 277:955–959.
Feng J, Funk WD, Wang S-S, et al.: The RNA component of human telomerase. Science 1995, 269:1236–1241.
Allsopp RC, Vaziri H, Patterson C, et al.: Telomere length predicts replicative capacity of human fibroblasts. Proc Natl Acad Sci U S A 1992, 89:10114–10118.
Chang E, Harley CB: Telomere length and replicative aging in human vascular tissues. Proc Natl Acad Sci U S A 1995, 92:11190–11194.
Effros RB, Allsopp R, Chiu C-P, et al.: Shortened telomeres in the expanded CD28-CD8+T cell subset in HIV disease implicate replicative senescence in HIV pathogenesis. AIDS 1996, 10:F17-F22.
Harley CB, Vaziri H, Counter CM, et al.: The telomere hypothesis of cellular aging. Exp Gerontol 1992, 27:375–382.
Hastie ND, Dempster M, Dunlop MG, et al.: Telomere reduction in human colorectal carcinoma and with ageing. Nature 1990, 346:866–868.
Kojima H, Yokosuka O, Imazeki F, et al.: Telomerase activity and telomere length in hepatocellular carcinoma and chronic liver disease. Gastroenterology 1997, 112:493–500.
Lindsey J, McGill NI, Lindsey LA, et al.: In vivo loss of telomeric repeats with age in humans. Mutat Res 1991, 256:45–48.
Urabe Y, Nouso K, Higashi T, et al.: Telomere length in human liver diseases. Liver 1996, 16:293–297.
Vaziri H, Schachter F, Uchida I, et al.: Loss of telomeric DNA during aging of normal and trisomy 21 human lymphocytes. Am J Hum Genet 1993, 52:661–667.
Weng N-P, Palmer LD, Levine BL, et al.: Tales of tails: regulation of telomere length and telomerase activity during lymphocyte development, differentiation, activation, and aging. Immunol Rev 1997, 160:43–54.
Counter CM, Avilion AA, LeFeuvre CE, et al.: Telomere shortening associated with chromosome instability is arrested in immortal cells which express telomerase activity. EMBO J 1992, 11:1921–1929.
Kim NW, Piatyszek MA, Prowse KR, et al.: Specific association of human telomerase activity with immortal cells and cancer. Science 1994, 266:2011–2014.
Shay JW, Bacchetti S: A survey of telomerase activity in human cancer. Eur J Cancer 1997, 33:777–791.
Wright WE, Piatyszek MA, Rainey WE, et al.: Telomerase activity in human germline and embryonic tissues and cells. Dev Genet 1996, 18:173–179.
Bodnar A, Kim NW, Effros RB, et al.: Mechanism of telomerase induction during T cell activation. Exp Cell Res 1996, 228:58–64.
Broccoli D, Young JW, de Lange T: Telomerase activity in normal and malignant hematopoietic cells. Proc Natl Acad Sci U S A 1995, 92:9082–9086.
Chiu C-P, Dragowska W, Kim NW, et al.: Differential expression of telomerase activity in hematopoietic progenitors from adult human bone marrow. Stem Cells 1996, 14:239–248.
Hiyama K, Hirai Y, Kyoizumi S, et al.: Activation of telomerase in human lymphocytes and hematopoietic progenitor cells. J Immunol 1995, 155:3711–3715.
Tahara H, Nakanishi T, Kitamoto M, et al.: Telomerase activity in human liver tissues: comparison between chronic liver disease and hepatocellular carcinomas. Cancer Res 1995, 55:2734–2736.
Taylor RS, Ramirez RD, Ogoshi M, et al.: Detection of telomerase activity in malignant and nonmalignant skin conditions. J Invest Dermatol 1996, 106:759–765.
Harley CB: Telomere loss: mitotic clock or genetic time bomb? Mut Res 1991, 256:271–282.
Bodnar AG, Ouellette M, Frolkis M, et al.: Extension of life-span by introduction of telomerase into normal human cells. Science 1998, 279:349–352. Telomerase-negative somatic cells from humans were transfected with vectors encoding the human telomerase catalytic subunit. These cells exhibited a normal karyotype, extended life-span, and no evidence of telomeric attrition. In contrast, telomerase-negative cells showed telomeric attrition and replicative senescence.
Hahn WC, Stewart SA, Brooks MW, et al.: Inhibition of telomerase limits the growth of human cancer cells. Nature Med 1999, 5:1164–1170.
Hahn WC, Counter CM, Lundberg AS, et al.: Creation of human tumour cells with defined genetic elements. Nature 1999, 400:464–468.
Vaziri H, Benchimol S: Reconstitution of telomerase activity in normal human cells leads to elongation of telomeres and extended replicative life span. Curr Biol 1998, 8:279–282.
Zhang X, Mar V, Harrington L, et al.: Telomere shortening and apoptosis in telomerase-inhibited human tumor cells. Genet Dev 1999, 13:2388–2399.
Prowse KR, Greider CW: Developmental and tissue specific regulation of mouse telomerase and telomere length. Proc Natl Acad Sci U S A 1995, 92:4818–4822.
Starling JA, Maule J, Hastie ND, et al.: Extensive telomere repeat arrays in mouse are hypervariable. Nucleic Acids Res 1990, 18:6881–6888.
Wright WE, Shay JW: Telomere dynamics in cancer progression and prevention: fundamental differences in human and mouse telomere biology. Nature Med 2000, 6:849–851.
Blasco MA, Lee HW, Hande MP, et al.: Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell 1997, 91:25–34.
Chin L, Artandi SE, Shen Q, et al.: p53 Deficiency rescues the adverse effects of telomere loss and cooperates with telomere dysfunction to accelerate carcinogenesis. Cell 1999, 97:527–538.
Greenberg RA, Chin L, Femino A, et al.: Short dysfunctional telomeres impair tumorigenesis in the INK4a 2/3 cancer-prone mouse. Cell 1999, 97:515–525.
Herrera E, Martinez-A C, Blasco MA: Impaired germinal center reaction in mice with short telomeres. EMBO J 2000, 19:472–481.
Herrera E, Samper E, Martin-Caballero J, Flores, et al.: Disease states associated with telomerase deficiency appear earlier in mice with short telomeres. EMBO J 1999, 18:2950–2960.
Lee H-W, Blasco MA, Gottlieb G, et al.: Essential role of mouse telomerase in highly proliferative organs. Nature 1998, 392:569–574.
Rudolph KL, Chang S, Lee H-W, et al.: Longevity, stress response, and cancer in aging telomerase-deficient mice. Cell 1999, 96:701–712.
Rudolph KL, Chang S, Millard M, et al.: Inhibition of experimental liver cirrhosis in mice by telomerase gene delivery. Science 2000, 287:1253–1258. This article suggests that experimental liver cirrhosis in the telomerase knockout mouse can be reversed by the introduction of the telomerase RNA gene.
Gonzalez-Suarez, E, Samper E, Flores JM, et al.: Telomerasedeficient mice with short telomeres are resistant to skin tumorigenesis. Nature Genet 2000, 26:1–4.
Artandi SE, Chang S, Lee S-L, et al.: Telomere dysfunction promotes non-reciprocal translocations and development of epithelial cancers in mice. Nature 2000, 406:641–645.
Beckman KB, Ames BN: The free radical theory of aging matures. Physiol Rev 1998, 78:547–581. Review of the role of ROS in biological aging.
Barja G: Mitochonderial free radical production and aging in mammals and birds. Ann N Y Acad Sci 1998, 854:224–238.
Allen RG, Tresini M: Oxidative stress and gene regulation. Free Rad Biol Med 2000, 28:463–499.
Berk BC: Redox signals that regulate the vascular response to injury. Thromb Haemost 1999, 82:810–817.
Lu S: Regulation of glutathione synthesis. Curr Top Cel Regul 2000, 26:95–116.
Halliwell B, Gutteridge JM: Free Radicals in Biology and Medicine. Oxford: Oxford University Press; 1989.
Pearl R: The Rate of Living. London: University of London Press; 1928.
Sohl RS: Metabolic rate and life span. In Cellular Aging: Concepts and Mechanisms. Edited by Witler R. Basel, Switzerland: Karger; 1976:25–40.
Ames BN, Gold LS, Willet WC: The causes and prevention of cancer. Proc Natl Acad Sci U S A 1995, 92:5258–5265.
Griendling KK, Sorescu D, Ushio-Fukai M: NAD(P)H oxidase role in cardiovascular biology and disease. Circ Res 2000, 86:494–501.
Smith MA, Sayre LM, Monnier VM, et al.: Radical ageing in Alzheimer’ disease. Trends Neurosci 1995, 18:172–176.
Dean RT, Fu S, Stocker R, et al.: Biochemistry and pathobiology of radical-mediated protein oxidation. Biochem J 1997, 324:1–18.
Stein GH, Drulinger LF, Soulard A, et al.: Differential roles of cyclin-dependent kinase inhibitors P21 and P16 in the mechanisms of senescence and differentiation in human fibroblasts. Mol Cell Biol 1999, 19:2109–2117.
Vaziri H, West MD, Alsopp RC, et al.: ATM-dependent telomere loss in aging human diploid fibroblasts and DNA damage lead to the post-translateral activation of p53 protein involving poly (AR-ribose) polymerase. EMBO J 1997, 16:6018–6033.
Sitte N, Saretzki G, von Zglinicki T: Accelerated telomere shortening in fibroblasts after extended periods of confluency. Free Radic Biol Med 1998, 24:885–893.
von ZglinickiT: Role of oxidative stress in telomere length regulation and replicative senescence. Ann N Y Acad Sci 2000, 908:99–110. Review of the role of ROS in telomere biology.
Swei A, Lacy F, DeLano FA, et al.: Oxidative stress in the Dahl hypertensive rat. Hypertension 1997, 30:1628–1633.
Suzuki H, DeLano FA, Parks DA, et al.: Xanthine oxidase activity associated with arterial blood pressure in spontaneously hypertensive rats. Proc Natl Acad Sci U S A 1998, 95:4754–4759.
Suzuki H, Swei A, Zweifach BW, et al.: In vivo evidence for microvascular oxidative stress in spontaneously hypertensive rats: hydroethidine microfluorography. Hypertension 1995, 25:1083–1089.
Vaziri ND, Oveisi F, Ding Y: Role of increased free radical activity in the pathogenesis of uremic hypertension. Kidney Int 1998, 53:1748–1754.
Vaziri ND, Liang K, Ding Y: Increased nitgric oxide inactivation by reactive oxygen species in lead-induced hypertension. Kidney Int 1998, 53:1748–1754.
Lopez-Ongil S, Hernandez-Perera O, Navarro-Antolin J, et al.:Role of reactive oxygen species in the signaling cascade of cyclosporine A-mediated up-regulation of eNOS in vascular endothelial cells. Br J Pharmacol 1998, 124:447–454.
Vaziri ND, Wang XQ, Oveisi F, et al.: Induction of oxidative stress by glutathione depletion causes severe hypertension in normal rats. Hypertension 2000, 36:142–146.
Lacy F, O’Connor DT, Schmid-Schonbein GW: Plasma hydrogen peroxide production in hypertensives and normotensive subjects at genetic risk of hypertension. J Hypertens 1998, 16:291–303.
Lacy F, Kailasam MT, O’Connor DT, et al.: Plasma hydrogen peroxide production in human essential hypertension. Role of heredity, gender, and ethnicity. Hypertension 2000, 36:878–884.
Bolzan AD, Bianchi MS, Bianchi NO: Superoxide dismutase, catalase and glutathione peroxidase activities in human blood: influence of sex, age and cigarette smoking. Clin Biochem 1997, 30:449–454.
Tang M, Subbiah MT: Estrogens protect against hydrogen peroxide and arachidonic acid induced DNA damage. Biochem Biophys Acta 1996, 1299:155–159.
Romer W, Oettel M, Menzenbach B, et al.: Novel estrogens and their radical scavenging effects, iron-chelating and total antioxidative activities: 17 alpha-substituted analogs of delta 9 (11)-dehydro-17 beta estradiol. Steroids 1997, 62:688–694.
Okuda K, Khan MY, Skurnick J, et al.: Telomere attrition of the human abdominal aorta: relationships with age and atherosclerosis. Atherosclerosis 2000, 152:391–398.
Xu D, Neville R, Finkel T: Homocysteine accelerates endothelial cell senescence. FEBS Lett 2000, 470:20–24.
O’Rourke M, Frohlich ED: Pulse pressure: is this a clinically useful risk factor? Hypertension 1999, 34:372–374.
Izzo JL Jr, Levy D, Black HR: Clinical Advisory Statement. Importance of systolic blood pressure in older Americans. Hypertension 2000, 35:1021–1024.
Zhang Y, Stewart KG, Dandge ST: Estrogen replacement reduces age-associated remodeling in rat mesenteric arteries. Hypertension 2000, 36:970–974.
Jeanclos E, Schork NJ, Kyvik KO, et al.: Telomere length inversely correlates with pulse pressure and is highly familial. Hypertension 2000, 36:195–200.
Benetos A, Okuda K, Lajemi M, et al.: Telomere length as an indicator of biological aging: the gender effect and relation with pulse pressure and pulse wave velocity. Hypertension 2001, in press.
von Zglinicki T, Serra V, Lorenz M, et al.: Short telomeres in patients with vascular dementia: an indicator of low antioxidative capacity and possible risk factor? Lab Invest 2000, 80:1739–1747.
Slagboom PE, Droog S, Boomsma DI: Genetic determination of telomere size in humans: a twin study of three age groups. Am J Hum Genet 1994, 55:876–882.
Kyo S, Takakura M, Kanaya T, et al.: Estrogen activates telomerase. Cancer Res 1999, 59:5917–5921.
O’Brien W, Stenman G, Sager R: Suppression of tumor growth by senescence in virally transformed human fibroblasts. Proc Natl Acad Sci U S A 1986, 83:8659–8663.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Aviv, A., Harley, C.B. How long should telomeres be?. Current Science Inc 3, 145–151 (2001). https://doi.org/10.1007/s11906-001-0029-3
Issue Date:
DOI: https://doi.org/10.1007/s11906-001-0029-3