Abstract
Purpose of Review
Differentiated service delivery (DSD) models were initially developed as a means to combat suboptimal long-term retention in HIV care, and to better titrate limited health systems resources to patient needs, primarily in low-income countries. The models themselves are designed to streamline care along the HIV care cascade and range from individual to group-based care and facility to community-based health delivery systems. However, much remains to be understood about how well and for whom DSD models work and whether these models can be scaled, are sustainable, and can reach vulnerable and high-risk populations. Implementation science is tasked with addressing some of these questions through systematic, scientific inquiry. We review the available published evidence on the implementation of DSD and suggest further health systems innovations needed to maximize the public health impact of DSD and future implementation science research directions in this expanding field.
Recent Findings
While early observational data supported the effectiveness of various DSD models, more recently published trials as well as evaluations of national scale-up provide more rigorous evidence for effectiveness and performance at scale. Deeper understanding of the mechanism of effect of various DSD models and generalizability of studies to other countries or contexts remains somewhat limited. Relative implementability of DSD models may differ based on patient preference, logistical complexity of model adoption and maintenance, human resource and pharmacy supply chain needs, and comparative cost-effectiveness. However, few studies to date have evaluated comparative implementation or cost-effectiveness from a health systems perspective.
Summary
While DSD represents an exciting and promising “next step” in HIV health care delivery, this innovation comes with its own set of implementation challenges. Evidence on the effectiveness of DSD generally supports the use of most DSD models, although it is still unclear which models are most relevant in diverse settings and populations and which are the most cost-effective. Challenges during scale-up highlight the need for accurate differentiation of patients, sustainable inclusion of a new cadre of health care worker (the community health care worker), and substantial strengthening of existing pharmacy supply chains. To maximize the public health impact of DSD, systems need to be patient-centered and adaptive, as well as employ robust quality improvement processes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Health systems in sub-Saharan Africa have continued to evolve in response to the changing global HIV/AIDS epidemic. Early in the epidemic response, health systems were challenged to rapidly scale-up antiretroviral therapy (ART) and care for acutely ill patients. As the response gathered momentum, decentralization and task-shifting were implemented to address emerging systems constraints. In the current era of universal ART, health systems have the dual challenge of initiating greater numbers of individuals on treatment while also providing chronic, life-long care to a growing number of healthy, and yet diverse groups of people. As a result, sustained retention in care has emerged as a key challenge in the current HIV/AIDS response and in achieving the “90-90-90” goals set forth by UNAIDS. Differentiated service delivery (DSD) was developed as a means to combat suboptimal long-term retention by “simplifying and adapting care” along the cascade, guided by patient preferences and needs, while at the same time, “reducing unnecessary burdens” on individuals and the health system [1].
Although some DSD models represent a continuation of decentralization (to health posts, communities, and patient homes) and task-shifting (to lay health care workers and patients), not all models follow this pattern. DSD models run the gamut from community to facility-based, client-driven to health care worker-driven, and group-based to individual strategies (Fig. 1) [1]. What all models do have in common is that they attempt to vary the intensity, frequency, and location of services based on patient preference and need [3]. Although initially designed to provide care for stable adults on ART, DSD models have since evolved to include patients with advanced or virologically detectable disease as well as children, adolescents, pregnant patients, and patients with comorbidities (e.g., tuberculosis and non-communicable diseases).
Summary of differentiated service delivery models (reprinted with permission from Bemelmans M, Baert S, Goemaere E, et al. Community-supported models of care for people on HIV treatment in sub-Saharan Africa. Trop Med Int Health. 2014;19 [8]:968–977) [2]
While the intent of DSD models is to titrate service intensity to meet patient needs without compromising service quality and health outcomes, much remains to be understood about whether and how well specific DSD interventions actually work (effectiveness), how they work (mechanism of intervention effect), for whom they work (generalizability and transportability), whether they work at scale, and whether they help those at greatest risk of falling out of care (reach). It is precisely these types of real-world questions that the burgeoning field of implementation science is concerned with answering. We review the available published evidence on the implementation of DSD and suggest further health systems innovations needed to maximize the public health impact of DSD and future implementation science research directions in this expanding field.
Do DSD Interventions Work? (Effectiveness)
Data supporting the effectiveness of DSD interventions were initially limited to observational studies of pilot programs [4,4,5,6,7,8,9,10,12]. Some of these early findings have since been corroborated by evaluations at-scale in both Mozambique and South Africa [13, 14] as well as in experimental trials [15]. Most studies have focused on evaluating intervention effect on retention, a smaller number on viral load suppression, and even fewer on mortality. While the majority of these studies provide support for the effectiveness of differentiating service delivery, not all evidence is positive.
Group-Based/Client-Managed Models: Community ART Groups
Although evaluations of Community ART Groups (CAGs) have consistently shown substantial reductions in loss to follow-up, potential selection bias limits our ability to comment on model effectiveness. CAGs are small groups of patients who rotate clinical follow-up care and drug pick-up duties at the facility and meet in the community monthly for group drug distribution and peer support. The earliest CAG analyses from Mozambique reported 1-year retention > 97% and 4-year retention at 91.8% [4, 16]. A major limitation of these early studies was the lack of a comparator group and the risk of selection bias as only patients electing to join the CAG model were evaluated and these patients may inherently be more likely to be retained in care. CAG uptake has been shown to be higher among women and clients with lower education/socioeconomic status and lower among men, migrant populations, and clients unwilling to disclose HIV status, with fears of inadvertent disclosure in the community, or with weak social networks [13, 17,17,19].
Since then, a large cohort study at 68 facilities in Mozambique utilized propensity score matching to compare outcomes among patients in CAGs to matched controls who were eligible, but not in CAGs [20]. These data showed non-CAG participants were more than twice as likely to be lost to follow-up (LTFU) compared with CAG participants (HR 2.356; p = 0.04) but no difference in mortality was found between groups. The covariates included in the propensity score, however, likely did not predict the likelihood of choosing to join a CAG and may therefore have not fully addressed selection bias. A complementary analysis utilizing the same CAG participant cohort, but this time comparing CAGs to non-CAG participants at 170 of 288 (60%) of all adult ART facilities in Mozambique and including CAG participation as a time-varying covariate, yielded similar findings [13]. A growing body of literature from other countries (Lesotho, Swaziland, Kenya, Haiti) provides additional observational evidence to support CAG effectiveness [21,21,22,24]. Randomized data on the effectiveness of CAGs compared with standard of care (Community ART study in Zambia) and of 3-vs 6-monthly ART dispensation in CAGs (MMSD study in Zimbabwe) are forthcoming.
Group-Based/Health Care Worker–Managed Models: Adherence Clubs
Effectiveness data on adherence clubs (AC) comes primarily from South Africa, and although generally very promising, has recently yielded mixed results. ACs are groups of approximately 30 patients who meet every 2 to 3 months for group-based drug pick-up and counseling either in the facility or community and who receive clinical follow-up care every 6 or 12 months. Early pilot studies in Cape Town, South Africa, demonstrated a 57% reduction in the risk of loss to follow-up (LTFU) and 67% reduction in the risk of virologic rebound compared with facility-based care [5, 25, 26]. These were conducted at single sites, leaving some question as to the generalizability of study findings to other countries and at scale. However, by 2015, ACs had been scaled to over 32,000 patients and in an analysis of a representative sample of 10% of this population, retention paralleled those found in the earlier pilots: 95.2% and 89.3% retention at 1 and 2 years respectively [14]. ACs were also effective in reducing incidence of late drug pick-up (greater than 7 days late) by fourfold in a cluster randomized trial in Zambia [15].
However, our understanding of AC model effectiveness has been recently complicated by reports of high LTFU and sub-optimal meeting attendance. Observational analyses in South Africa and Kenya have reported 36-month LTFU from ACs as high as 26% [27,27,29]. In the Kenyan study, the most common reasons for withdrawal were relocation (31%), perceived stigma associated with attending meetings (17%), and patient request to discontinue participation (14%) [29]. A randomized trial in South Africa comparing facility-based versus community-based ACs also showed poor retention in club-based care in both arms at 24 months (57% vs 48% respectively) [30]. Meeting non-attendance with failed drug pick-up was the most common reason for club dismissal. However, retention in any care exceeded 88% in both arms despite poor retention in a club, which may represent a “settling out” effect in which people ultimately select the care that works best for them over time.
Individual, Facility-Based Models: Visit Spacing, Multi-month Refills, Streamlined ART Pick-up
Data from individual, facility-based models have been universally positive, albeit some studies demonstrated small effect sizes and the wide variety of effectiveness outcomes measured make it difficult to compare outcomes between studies [6,6,7,8,10, 31,31,33]. These models include any combination of clinical visit spacing, multi-month drug refills (pharmacy visit spacing), and streamlined (fast-track) facility-based drug pick-up. Observational data from multiple settings are available (Uganda, Malawi, DRC, Guinea, Zambia, Nigeria) which highlight decreases in LTFU, missed visits, and medication gaps as well as improvements in patient wait time and provider/patient ratios with increased visit spacing. An ongoing cluster RCT in Malawi and Zambia, the INTERVAL study, will provide the first experimental evidence for the effectiveness of standard of care vs 3- and 6-month ART refills in improving retention in care and virologic suppression [34].
Individual, Community-Based Models: Home Delivery, Community Drug Distribution Points, Mobile ART Delivery
Published effectiveness data on individual, community-based models, which span home delivery, fixed community drug distribution points, and mobile ART delivery, have yielded mixed results in pilots and are yet to be widely scaled. Non-inferiority of health outcomes with home-based care compared with facility-based care was established early-on with two randomized trials in Uganda and Kenya [35, 36] and more recently in Ethiopia [37]. Lack of feasibility of home-based care as client numbers grew prompted TASO in Uganda to develop Community Drug Distribution Points (CDDP) (e.g., drug distribution at community pharmacies, health posts). [11, 38, 39] Cross-sectional assessment of a cohort of patients initiated on ART and enrolled in CDDP between 2004 and 2009 revealed retention of only 69% and death of 17% after 5 years of treatment [11]. Additional analyses showed more promising results but are yet to be published [38, 39]. More favorable results were seen in an MSF pilot of community drug distribution in the Democratic Republic of Congo (PODI) where overall attrition (death, LTFU, transfer out) was 5.66 per 100 person-years after 24 months of follow-up [12, 40, 41]. ATM-like ART dispensation is also being piloted in South Africa but effectiveness data is still forthcoming [42]. Data on mobile ART delivery is limited to two abstracts: one from Swaziland [22], which demonstrated only 77% retention at 12 months, and the second from South Africa, [43] which reported 91.2% virologic suppression and little LTFU (2%) among migrant farm workers on the South African-Zimbabwean border. Randomized data on mobile ART strategies is forthcoming from the DOART trial in Uganda and South Africa in which three models will be compared: (i) Home ART initiation and mobile van ART monitoring and resupply, (ii) Clinic ART initiation and mobile van ART monitoring and resupply, and (iii) Clinic ART initiation, monitoring, and resupply.
How Do DSD Interventions Work? (Mechanism of Intervention Effect)
While published data generally support the effectiveness of DSD models, inadequate understanding of how DSD interventions actually work has limited our ability to determine whether findings from a given study can be generalized and/or replicated across heterogenous socioeconomic, geographical, and cultural settings. There also remain important questions about the most critical elements needed for the success of individual models and about which models are best suited for different settings. Intervention mechanism of effect refers to the steps or processes through which the intervention translates into events or actions that lead to the outcome, or precisely what was altered that led to behavior change. Identification of DSD mechanism of effect has been qualitatively explored for CAGs [17, 18, 20, 21, 44, 45] and ACs [46,46,47,48,49,50,51,53]. However, no studies have yet quantitatively measured potential mediators of effect (factors which shows a statistical association and causal link between the intervention and outcome) or moderators of effect (factors which influence the direction or magnitude of the relation between the intervention and outcome).
In theory, DSD models work on multiple levels to improve retention in care and viral load suppression. Adherence and retention barriers have been previously well studied and include a wide range of factors that are “structural” or due to the external environment (e.g., transportation costs, work); “clinic-based” or due to the healthcare environment (e.g., waiting times, provider attitudes); or “patient-based” or due to knowledge, beliefs, or attitudes of patients within a given social context (e.g., denial, stigma, preference for spiritual healing) [54, 55]. Heretofore, adherence and retention interventions have primarily focused on addressing one of these barriers at a time (e.g., peer counseling, SMS, economic incentives). DSD theoretically offers a multi-modal strategy to address multiple barriers at the same time.
Structural Barriers
DSD models may address structural barriers by decreasing transport and opportunity costs from lost work by reducing visit frequency (in all models) and by bringing care closer to home (in community-based models) [17, 44, 45]. CAGs may additionally reduce costs through pooled financial resources for rotating CAG member travel to the facility [44, 45].
Clinic-Based Barriers
DSD models appear to address clinic-based barriers by reducing clinic congestion (via reduced visit frequency) and thereby waiting time [7]. Decreased provider workload and better monitoring of acutely ill patients (or those with WHO-defined “advanced disease”) may also reduce provider fatigue and improve provider attitudes, thereby altering patient experience and satisfaction at the clinic [44, 45]. Qualitative studies suggest that client-driven models (CAGs) foster greater patient activation, empowerment, and disease self-management, thus altering the previously hierarchal doctor-patient relationship [20, 44, 45].
Patient-Based Barriers
DSD models reduce stigma-related patient barriers due to decreased contact with the facility [17]. Group-based models are thought to additionally foster motivation through peer support. This may play a larger role in CAGs than ACs, which were created for “Fast. Friendly. Two months [sic] supply of ARVs” and to a lesser extent for social support [56••]. A unique potential mechanism for CAGs is greater accountability due to shared drug pick-up responsibilities. Membership in an “exclusive” club and feeling treated as a “VIP,” emerged as a motivator for some AC participants early-on, but dissipated as the number of clubs per clinic increased during scale-up in South Africa [47, 56••].
While many of these mechanisms are supported by existing qualitative data, quantitatively identifying their causal relationship to observed outcomes is important to understanding which mechanisms are most crucial to intervention success, and ultimately to aid policymakers and program developers in deciding which models (or model components) should be scaled or adapted for different individuals and/or populations. For example, while patient empowerment and disease self-management play a theoretical role in CAG intervention effect, it is unclear how important this is compared with the peer support received or the structural barriers addressed through the model. Similarly, while the group-based structure of ACs theoretically provides an avenue for peer support [47], quantitative data showing a large number of missed visits [15, 30] and qualitative data implying that suboptimal implementation of a supportive group environment leads to disobeyance of club rules [51], suggest that ACs may currently be acting more through convenient drug pick-up then via social support. Given this, the added complexities and cost of group formation and management may afford little benefit over the structural barriers addressed through visit spacing and fast-tracking in an individual, facility-based model. Ultimately, well-implemented group-based models may be most effective for patients with psychosocial barriers to care while patients reporting mostly structural barriers may do best with individual-based models.
The optimal way to obtain and leverage this kind of information is to investigate potential mediating factors along the causal pathway between intervention and outcome using mediation analysis. This analytic approach has already been employed to improve understanding of multicomponent interventions in the HIV/AIDS field [57].
For Whom Do DSD Interventions Work? (Generalizability and Transportability)
Knowledge of mechanism of effect is the first step in understanding the generalizability and transportability of study findings, i.e., to what extent can we use published DSD studies to infer or make conclusions about the effectiveness of these interventions in other settings. For example, can we assume that ACs (primarily studied in South Africa) will work the same way and be as effective in Rwanda or Kenya?
Historically, assessments of generalizability and transportability have been limited to subjective interpretations and expert opinion; however, a more sophisticated understanding could be obtained through a transportability framework and causal inference analysis. For example, if we first assume that the effect of a CAG on retention in care is (1) mediated by reduction in opportunity costs which is driven by distance to facility and current visit frequency and is (2) moderated by social capital. And if we then measure distance, visit frequency, and social capital, then, the observed effects can be transported to a new setting in which these same factors are measured, without further experimentation. While this type of analysis requires making other major assumptions (i.e., that there are no other differences in any variables that may affect retention in care), novel application of unique implementation science methods such as these holds some promise.
Do DSD Interventions Work at Scale and Can They Be Sustained?
While a robust theoretical understanding of how and where DSD interventions work is still nascent, key lessons have already been learned through DSD scale-up.
Differentiating Patients Based on Clinical Stability
A central aspect of DSD innovation is the concept of differentiating patients based on need. Initially, the intent was to identify clinically stable patients to receive reduced intensity and frequency of health services. However, an implementation science study of 30 purposively selected facilities in Malawi highlights the challenges with accurate differentiation in a real-world setting: 27% of patients who were eligible for multi-month scripting (based on national criteria for clinical stability) did not receive it while 42% who were ineligible did [58••]. The need to differentiate patients has also drawn attention to deficiencies in laboratory testing as access to CD4 count and viral load testing are required to identify patients with advanced disease and those who are not virologically suppressed. In an IeDeA Southern Africa network study, the authors found that in viral load monitoring sites (but not in CD4-only monitoring sites), the rate of clinic visits in stable patients was substantially lower than in unstable patients. [59] Furthermore, observational data in Zambia suggest that patient stability itself is quite dynamic and cross-sectional assessments of stability do little to reflect the complex nature of patients who transition back and forth between stability and instability [60]. Even as DSD models now strive to include clinically unstable patients, they face the same challenge of differentiating “instability.”
Model Uptake and Adoption
Studies of individual patient uptake of various models have been evaluated both qualitatively and quantitatively and suggest preference for individual, facility-based models. In Nigeria, Kenya, and Zimbabwe, multi-month scripting and fast-track models were preferred over client-driven or community-based models [61,61,63]. Required disclosure in group-based models and inadvertent disclosure in the community were key factors cited in these studies. Discrete choice experiments are a novel analytic method that can allow for quantification of patient preferences for specific DSD model attributes. In both Kenya and Zambia (FACES and CIDRZ unpublished data), the strongest overall preference across all patients was for a reduction in clinic visit frequency. There were additional preferences for individualized care, with urban populations preferring facility-based care and rural populations favoring community-based drug collection.
DSD model adoption at the facility-level also seems to favor individual, facility-based models over group-based or community-based models. In Malawi, multi-month scripting, fast-track drug refills, and CAGs were simultaneously scaled up [58••]. At facilities offering fast-track, 77% of eligible patients were enrolled, while at facilities offering CAGs, only 6% of eligible patients were enrolled. In a study mapping the scale-up of DSD in 722 NGO-supported facilities in 13 countries, facility-based individual models were most commonly implemented [64]. One possible explanation is that group-based model expansion faces additional hurdles due to the complexities and human resource needs of group formation and ongoing group management. In Mozambique, a key anticipated challenge during national scale-up of CAGs was the human resource requirement for a new workforce cadre of community health workers and supervisory staff to assemble and monitor the CAGs [18, 65]. A health system evaluation of AC scale-up in South Africa revealed that although initial widespread adoption of the AC model was successful, after more than 40 clubs at a facility were formed, logistical complexities of group management became unmanageable (this was termed the “forty club hurdle”) [56••].
Human Resource Needs
Nearly all DSD models strive to more efficiently utilize limited high-level health care worker (HCW) resources through task-shifting and are thereby increasingly dependent on community HCW. Both inadequate forecasting of HCW needs within DSD models as well as inadequate training, coordination, and compensation of community HCW have been seen as challenges to DSD scale-up [18, 66]. While several studies have already demonstrated the benefit of DSD models in reducing formal HCW provider workload ratios through decreased visit frequency [67, 68], in some settings, increased absolute numbers of ART patients (due to newly initiating clients) and new tasks such as pre-packaging of medications have offset the direct time-savings benefits experienced by health care workers [56••].
Pharmacy Supply Chain
Another key vulnerability that has emerged with DSD scale-up is gaps in pharmacy supply chain management. Programs that are able to pre-emptively plan and strengthen drug delivery systems (i.e., quantification, forecasting, and stock control) [69] prior to scale-up have fared well [70], while those that lacked this pre-planning have experienced delays in implementation, stock-outs, and client drop-outs [33, 71].
Comparative Costs and Cost-effectiveness
DSD implementation has the potential to mitigate the crisis in HIV treatment financing by providing care for more clients at the same cost [72]. While the fast-track and AC models have been found to be cost-effective when compared with standard of care [6, 73], a few studies have provided valuable costing data comparing different DSD models [58••, 74]. In Malawi, multi-month scripting, fast track, and CAGs all reduced average ART unit costs by 10% compared with standard of care, but costs across all three models were similar. In Uganda, TASO found that among three models of care, standard of care was cheapest ($257) compared with mobile ART delivery ($404) or community drug distribution points ($332). In Zambia, annual costs were most sensitive to salary levels of pharmacy technologists and the fast-track model was found to be the most costly ($92.72 annually) compared with CAG ($85.47) and AC ($27.46) [75]. Importantly, these unit costs are country-specific and do not account for possible differences in model effectiveness. Cost-effectiveness data are urgently needed and randomized comparisons are forthcoming from the DoART study in Uganda and South Africa, the INTERVAL study in Zambia and Malawi, and the MMSD study in Zimbabwe.
Relative Implementability of DSD Models
The relative implementability of any particular DSD model is likely a result of the aforementioned factors: patient preference, differential logistical complexity of model adoption and maintenance, comparative human resource and pharmacy supply chain needs, and relative cost-effectiveness. Thus far, there are few published studies directly examining implementation from a health systems perspective [56••] or comparative implementation of several DSD models within the same context [58••]. In Malawi, Prust et al., conducted a process evaluation of three different models of DSD care (multi-month scripting (MMS), fast track refills (FTR), and community adherence groups (CAGs)). MMS had been universally scaled up but the other two models were not offered at all clinics. The authors documented better differentiation of patient stability at FTR sites and lower uptake of CAGs among eligible patients, and concluded that expanding FTRs and CAGs did not afford additional cost savings compared with MMS alone. However, reasons for low CAG uptake and additional benefits to patient satisfaction or clinical outcomes have not yet been fully explored.
Do DSD Interventions Reach Vulnerable and Key Populations?
A plethora of pilots targeting children, adolescents, and pregnant or post-partum mothers provides early evidence that DSD models can close gaps in treating these vulnerable populations. For children, fast track programs [76] and multi-month scripting [77] were found to be effective. For adolescents, ACs in South Africa and “teen clubs” in Malawi [78], which include Saturday clinical visits to reduce school absenteeism and enhanced psychosocial support activities, have been successful in improving retention. ACs have also been employed for families [79] and post-partum women [80,80,82]. A relative advantage of group-based interventions is the added psychosocial support which may be of particular benefit to adolescents and pregnant/post-partum women.
Similarly, DSD strategies for key populations (men who have sex with men (MSM) [83], intravenous drug abusers (IVDA) [84, 85], sex workers [86,86,88], and transgender patients) are being piloted globally. Some of these programs allow for engagement in care along the entire HIV cascade (from HIV testing to ART initiation to ART maintenance) which is crucial for marginalized populations with decreased contact with health facilities.
Migrant populations and prisoners are two other traditionally difficult to treat populations in whom DSD models are being explored [43, 89]. In a small pilot study at the South Africa-Zimbabwean border, a successful targeted strategy for migrant workers was developed where those planning travel greater than 2 weeks were characterized as temporary transfer outs and were given a 3-month drug supply and a referral letter.
What Health Systems Innovations Are Needed to Maximize Benefits of DSD Implementation?
A crucial factor that has emerged in considering the benefits of a DSD approach is whether DSD is as truly adaptive to individual patient needs as it seeks to be, or whether there is a risk of simply replacing one rigid system with another. Inherent to this discussion is the tension between a one-size-fits-all public health approach and the risk of over-targeting. Although DSD cannot accommodate every individual’s circumstances, redesign can better meet the needs of many. We suggest here several key health systems innovations which are needed to maximize the public health impact of DSD service delivery.
Patient-Centered Care
While the commonly used definition of DSD references a desire to provide patient-centered care, the two concepts are not synonymous. Patient centeredness is a concept well established in high-income countries that has been shown to improve health outcomes and reduce costs [90, 91]. Key dimensions of patient centeredness include improved patient-provider relationships, recognition that a patient’s care exists in the context of their entire life (biopsychosocial perspective), and shared responsibility and decision-making. Patient-centeredness training, such as that developed in the PRIME intervention in Uganda for malaria care, should be adapted and scaled for HIV care and treatment [92].
Quality Improvement
Implementation of quality improvement strategies is essential for DSD systems to achieve the stated goal of maximizing efficiency without compromising service quality. If there are current gaps in facility-based service provision (i.e., poor laboratory testing infrastructure, frequent pharmacy stock-outs) then DSD will not fix these problems, and in fact may amplify them. Increased deployment of community HCW also poses a new challenge in ensuring health care quality [93, 94]. Application of quality-improvement methods, including iterative use of local data to identify gaps in care and test solutions, is critical.
Health System Responsiveness
It is already evident that patients’ needs and preferences within DSD models are dynamic and health systems must be sufficiently flexible and adaptive to allow for titration of service intensity and frequency over time. Migration, job changes, pregnancy, and development of opportunistic infections or comorbidities are frequent causes for disengagement from DSD models of care. Successful monitoring and evaluation platforms will need to account for frequent migration into and out of DSD models.
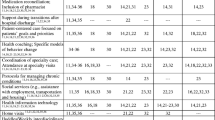
Conclusions
While DSD represents an exciting and promising “next step” in HIV health care delivery, it has already become clear that it is not a panacea and comes with its own set of challenges. Evidence on the effectiveness of DSD generally supports the use of most DSD models, although it is still unclear which models are most relevant in diverse settings and populations and which are the most cost-effective. Further research is needed in these and several other key areas (Table 1). Challenges during scale-up highlight the need for accurate differentiation of patients, sustainable inclusion of a new cadre of health care worker (the community HCW), and substantial strengthening of existing pharmacy supply chains. Understanding reasons for patient disengagement from various DSD models of care and long-term outcome data will additionally help determine DSD model sustainability. Finally, to maximize the public health impact of DSD, systems need to be patient-centered and adaptive, as well as employ robust quality improvement processes.
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
IAS. Differentiated care for HIV: a decision framework for antiretroviral therapy delivery. 2016.
Bemelmans M, Baert S, Goemaere E, Wilkinson L, Vandendyck M, van Cutsem G, et al. Community-supported models of care for people on HIV treatment in sub-Saharan Africa. Tropical Med Int Health. 2014;19(8):968–77.
Duncombe C, Rosenblum S, Hellmann N, Holmes C, Wilkinson L, Biot M, et al. Reframing HIV care: putting people at the centre of antiretroviral delivery. Tropical Med Int Health. 2015;20(4):430–47.
Decroo T, Telfer B, Biot M, Maïkéré J, Dezembro S, Cumba LI, et al. Distribution of antiretroviral treatment through self-forming groups of patients in Tete Province, Mozambique. J Acquir Immune Defic Syndr. 2011;56(2):e39–44.
Luque-Fernandez MA, Van Cutsem G, Goemaere E, et al. Effectiveness of patient adherence groups as a model of care for stable patients on antiretroviral therapy in Khayelitsha, Cape Town, South Africa. PLoS One. 2013;8(2):e56088.
Babigumira JB, Castelnuovo B, Stergachis A, Kiragga A, Shaefer P, Lamorde M, et al. Cost effectiveness of a pharmacy-only refill program in a large urban HIV/AIDS clinic in Uganda. PLoS One. 2011;6(3):e18193.
Alamo ST, Wagner GJ, Ouma J, Sunday P, Marie L, Colebunders R, et al. Strategies for optimizing clinic efficiency in a community-based antiretroviral treatment programme in Uganda. AIDS Behav. 2013;17(1):274–83.
Obua C, Kayiwa J, Waako P, Tomson G, Balidawa H, Chalker J, et al. Improving adherence to antiretroviral treatment in Uganda with a low-resource facility-based intervention. Glob Health Action. 2014;7:24198.
Cawley C NS, Szumilin E, Perry S, Amoros Quiles I, Masiku C, Wringe A. Six-monthly appointments as a strategy for stable antiretroviral therapy patients: evidence of its effectiveness from seven years of experience in a Medecins Sans Frontieres supported programme in Chiradzulu district, Malawi. International AIDS Conference; July 18–22, 2016; Durban, South Africa.
Bekolo CE, Diallo A, Philips M, Yuma JD, di Stefano L, Drèze S, et al. Six-monthly appointment spacing for clinical visits as a model for retention in HIV Care in Conakry-Guinea: a cohort study. BMC Infect Dis. 2017;17(1):766.
Okoboi S, Ding E, Persuad S, Wangisi J, Birungi J, Shurgold S, et al. Community-based ART distribution system can effectively facilitate long-term program retention and low-rates of death and virologic failure in rural Uganda. AIDS Res Ther. 2015;12:37.
Vogt F, Kalenga L, Lukela J, Salumu F, Diallo I, Nico E, et al. Brief report: decentralizing ART supply for stable HIV patients to community-based distribution centers: program outcomes from an urban context in Kinshasa, DRC. J Acquir Immune Defic Syndr. 2017;74(3):326–31.
Auld AF, Shiraishi RW, Couto A, Mbofana F, Colborn K, Alfredo C, et al. A decade of antiretroviral therapy scale-up in Mozambique: evaluation of outcome trends and new models of service delivery among more than 300,000 patients enrolled during 2004-2013. J Acquir Immune Defic Syndr. 2016;73(2):e11–22.
Tsondai PR, Wilkinson LS, Grimsrud A, Mdlalo PT, Ullauri A, Boulle A. High rates of retention and viral suppression in the scale-up of antiretroviral therapy adherence clubs in Cape Town, South Africa. J Int AIDS Soc. 2017;20(Suppl 4):51–7.
Roy Monika B-MC, Sikazwe I, Mukumbwa-Mwenechanya M, Efronson E, Somwe P, Kalunkumya E, Lumpa M, Sharma A, Pry Jake, Padian N, Geng E, Holmes C. Urban adherence clubs in Zambia: findings from model implementation (FRAE0104). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Decroo T, Koole O, Remartinez D, dos Santos N, Dezembro S, Jofrisse M, et al. Four-year retention and risk factors for attrition among members of community ART groups in Tete, Mozambique. Tropical Med Int Health. 2014;19(5):514–21.
Pellecchia U, Baert S, Nundwe S, et al. “We are part of a family”. Benefits and limitations of community ART groups (CAGs) in Thyolo, Malawi: a qualitative study. J Int AIDS Soc. 2017;20(1):21374.
Rasschaert F, Decroo T, Remartinez D, Telfer B, Lessitala F, Biot M, et al. Sustainability of a community-based anti-retroviral care delivery model - a qualitative research study in Tete, Mozambique. J Int AIDS Soc. 2014;17:18910.
Roy M, Mukumbwa-Mwenechanya M, Efronson E, Lumpa M, Sharma A, Sikazwe I, Padian N, Bolton-Moore C, Geng E, Holmes C. Uptake and adaption of community adherence groups in Zambia (#1004). Conference on Retroviruses and Opportunistic Infections (CROI); February 13-16, 2017; Seattle, Washington, USA.
Jobarteh K, Shiraishi RW, Malimane I, Samo Gudo P, Decroo T, Auld AF, et al. Community ART support groups in Mozambique: the potential of patients as partners in care. PLoS One. 2016;11(12):e0166444.
Vandendyck MMM, Mubanga M. Community-based ART resulted in excellent retention and can leverage community empowerment in rural Lesotho: a mixed method study. HIV/AIDS Research and Treatment. 2015;2(2):44–50.
Pasipamire L KB, I. Zabsonre I, Ndlovu S, Sibanda G, Mamba S, Lukhele N, Mazibuko S, Pasipamire M, Kabore SM, Rusch B. Implementation of combination ART refills models in rural Swaziland. International AIDS Conference; July 18–22, 2016; Durban, South Africa.
Konah S. Rolling out a community differentiated care model: Lessons learned from a high-volume health facility in Taita Taveta County, Kenya (WEPEE762). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Naslund JA, Dionne-Odom J, Junior Destine C, et al. Adapting and implementing a community program to improve retention in care among patients with HIV in southern Haiti: “group of 6”. AIDS Research and Treatment. 2014;2014:137545.
Grimsrud A, Sharp J, Kalombo C, Bekker LG, Myer L. Implementation of community-based adherence clubs for stable antiretroviral therapy patients in Cape Town, South Africa. J Int AIDS Soc. 2015;18:19984.
Grimsrud A, Lesosky M, Kalombo C, Bekker LG, Myer L. Implementation and operational research: community-based adherence clubs for the management of stable antiretroviral therapy patients in Cape Town, South Africa: a cohort study. J Acquir Immune Defic Syndr. 2016;71(1):e16–23.
Nofemela A, Kalombo, C, Orrell, C, Myer, L. Discontinuation from community-based antiretroviral adherence clubs in Gugulethu, Cape Town, South Africa International AIDS Conference; July 22–26, 2016; Durban, South Africa.
Cassidy T OCR, Euvrard J, Ndlovu S, Duran LT, Steele SJ, Oba J, Daho S, Boulle A, Gerstenhaber R, Schulz T. A “quick pick-up” differentiated model of ART delivery shows good retention in care (WEPEE751). 22nd International AIDS Conference; July 23-27, 2018; Amsterdam, Netherlands.
Goodrich S SA, Mwangi A, Spira T, Bateganya M, Toroitich-Ruto C, Otieno-Nyunya B, Naanyu V, Wools-Kaloustian K. Effectiveness of a community-based model of HIV care in western Kenya (LBPEE050). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Hanrahan C KV, Schwartz S, Mudavanhu M, West N, Mutunga L, Steingo J, Bassett J, Van Rie A. Retention in community versus clinic-based adherence clubs for stable ART patients in South Africa: 24 month final outcomes from an RCT. 9th IAS Conference on HIV Science; July 23–26, 2017; Paris, France.
Mody A, Roy M, Sikombe K, Savory T, Holmes C, Bolton-Moore C, et al. Improved retention with 6-month clinic return intervals for stable human immunodeficiency virus-infected patients in Zambia. Clin Infect Dis. 2018;66(2):237–43.
Pry J RM, Sikazwe I, Mukumbwa-Mwenechanya M, Somwe P, Kalunkumya E, Lumpa M, Sharma A, Padian N, Geng E, Holmes C, Bolton C. Does faster service equal better retention: assessing fast track services in Lusaka, Zambia (LBPEC030). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Attah M MA, Hassan S, Askederin F, Osueke L, Innocent I, Haanongon PD, Esemokhai E, Ajulo V, Field-Nguer ML. Nigeria ARV multimonth scripting: impact on public health service delivery, infrastructure and supply chain management systems across “high volume” ART clinics (WEPEE740). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Balakasi K PP, Bardon A, Hubbard J, Moyo C, Siwale Z, Kakwesa G, Haambokoma M, Phiri J, Fox M, Kalua T, Xulu T, Rosen S, Sanne I, Hoffman R. Characteristics of adults in Malawi and Zambia eligible for 6-monthly antiretroviral therapy refills: the INTERVAL study (WEPEE763). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Jaffar S, Amuron B, Foster S, Birungi J, Levin J, Namara G, et al. Rates of virological failure in patients treated in a home-based versus a facility-based HIV-care model in Jinja, southeast Uganda: a cluster-randomised equivalence trial. Lancet. 2009;374(9707):2080–9.
Selke HM, Kimaiyo S, Sidle JE, Vedanthan R, Tierney WM, Shen C, et al. Task-shifting of antiretroviral delivery from health care workers to persons living with HIV/AIDS: clinical outcomes of a community-based program in Kenya. J Acquir Immune Defic Syndr. 2010;55(4):483–90.
Msafiri Francis J GP, Sando D, Asmus G, Lema IA, Mboggo E, Koda H, Lwezaula S, Ambikapathi R, Fawzi W, Ulenga N, Bärnighausen T. ARV community delivery: a non-inferiority cluster-randomized pragmatic trial in Dar es Salaam. 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Mpima D BJ, Makabayi R, Kanters S, Luzze C. . Community antiretroviral therapy (ART) delivery models for high patient retention and sustaining good adherence: The AIDS Support Organisation (TASO) operational reseach findings, CDC/PEPFAR funded project in Uganda. 7th IAS Conference on HIV Pathogenesis, Treatment and Prevention; June 30 - July 3, 2013; Kuala Lumpur, Malaysia.
Musinguzi Rugamba K GP, Senyimba C, Mukasa B The role of community drug distribution point of care model on retention of HIV-positive individuals from a pastoral community in Kigaju, Kalungu district (TUPED1323). 9th IAS conference on HIV Science; July 23–26, 2017; Paris, France.
Tchissambou T JA, Dane S, Dibulundu D, Matsumaini S, Madevu-Matson C, Ngalamulume-Roberts B, Ilunga Y, Malele F, J SL. Community ART distribution center (PODI) as a differentiated service delivery measure in the Democratic Republic of Congo (DRC) (WEPEE735). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Mothibi E MJ, Kabamba D, Monteith L, N’siesi FX, Firth J, Kiluba J, Kamhere D, Chirowa F, Makumbe B, Grimwood A, Fatti G Improving retention, viral suppression, and facility decongestion through community-based individual drug distribution (PODIs) in DRC (WEPEE754). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
S B. Pharmacy dispensing units (PDUs): improving access to chronic medicines. Reaching 90-90-90 in South Africa Part III: Best practices and innovations in linkage, treatment, and viral suppression; May 31–June 1, 2016; Pretoria, South Africa.
Matambo T HK, Wilkinson L, Van Cutsem G, Bauemfeind A et al. Targeted adherence strategies for provision of cross border antiretroviral therapy (ART) to migrant farm workers in Musina, South Africa. 19th International AIDS Conference; July 22–25, 2012; Washington D.C., USA.
Rasschaert F, Decroo T, Remartinez D, Telfer B, Lessitala F, Biot M, et al. Adapting a community-based ART delivery model to the patients’ needs: a mixed methods research in Tete, Mozambique. BMC Public Health. 2014;14:364.
Rasschaert F, Telfer B, Lessitala F, Decroo T, Remartinez D, Biot M, et al. A qualitative assessment of a community antiretroviral therapy group model in Tete, Mozambique. PLoS One. 2014;9(3):e91544.
Venables E, Edwards JK, Baert S, Etienne W, Khabala K, Bygrave H. “They just come, pick and go.” The acceptability of integrated medication adherence clubs for HIV and non communicable disease (NCD) patients in Kibera, Kenya. PLoS One. 2016;11(10):e0164634.
Venables E, Towriss C, Rini Z, et al. If I’m not in the club, I have to move from one chair to another.” A qualitative evaluation of patient experiences of adherence clubs in Khayelitsha and Gugulethu, South Africa. IAS; 2017; Paris, France.
Mukumbang FC, Mwale JC, van Wyk B. Conceptualising the factors affecting retention in care of patients on antiretroviral treatment in Kabwe District, Zambia, using the ecological framework. AIDS Research and Treatment. 2017;2017:7356362.
Mukumbang FC, Van Belle S, Marchal B, van Wyk B. An exploration of group-based HIV/AIDS treatment and care models in sub-Saharan Africa using a realist evaluation (intervention-context-actor-mechanism-outcome) heuristic tool: a systematic review. Implement Sci. 2017;12(1):107.
Mukumbang FC, Van Belle S, Marchal B, van Wyk B. Exploring 'generative mechanisms’ of the antiretroviral adherence club intervention using the realist approach: a scoping review of research-based antiretroviral treatment adherence theories. BMC Public Health. 2017;17(1):385.
Mukumbang FC, Marchal B, Van Belle S, van Wyk B. “Patients are not following the [adherence] club rules anymore”: a realist case study of the antiretroviral treatment adherence Club, South Africa. Qual Health Res. 2018;28(12):1839–57.
Mukumbang FC, Marchal B, Van Belle S, van Wyk B. A realist approach to eliciting the initial programme theory of the antiretroviral treatment adherence club intervention in the Western Cape Province, South Africa. BMC Med Res Methodol. 2018;18(1):47.
Mukumbang FC, Marchal B, Van Belle S, van Wyk B. Unearthing how, why, for whom and under what health system conditions the antiretroviral treatment adherence club intervention in South Africa works: a realist theory refining approach. BMC Health Serv Res. 2018;18(1):343.
Geng EH, Odeny TA, Lyamuya R, Nakiwogga-Muwanga A, Diero L, Bwana M, et al. Retention in care and patient-reported reasons for undocumented transfer or stopping care among HIV-infected patients on antiretroviral therapy in Eastern Africa: application of a sampling-based approach. Clin Infect Dis. 2016;62(7):935–44.
Haberer JE, Sabin L, Amico KR, Orrell C, Galárraga O, Tsai AC, et al. Improving antiretroviral therapy adherence in resource-limited settings at scale: a discussion of interventions and recommendations. J Int AIDS Soc. 2017;20(1):21371.
•• MacGregor H, McKenzie A, Jacobs T, Ullauri A. Scaling up ART adherence clubs in the public sector health system in the Western Cape, South Africa: a study of the institutionalisation of a pilot innovation. Glob Health. 2018;14(1):40. This study provides unique information about the challenges of scaling-up a particular DSD model from a health systems perspective.
Bryan A, Schmiege SJ, Broaddus MR. Mediational analysis in HIV/AIDS research: estimating multivariate path analytic models in a structural equation modeling framework. AIDS Behav. 2007;11(3):365–83.
•• Prust ML, Banda CK, Nyirenda R, et al. Multi-month prescriptions, fast-track refills, and community ART groups: results from a process evaluation in Malawi on using differentiated models of care to achieve national HIV treatment goals. J Int AIDS Soc. 2017;20(Suppl 4):41–50. This publication is one of the only studies evaluating the relative implementability of various DSD models and provides valuable insights into DSD implementation challenges.
Haas A JL, Sikazwe I, Ford N, Fox M, Prozesky H, Chimbetete C, Euvrard J, Egger M. Is Differentiated care implemented? Analysis of seven African treatment programs. Conference on Retroviruses and Opportunistic Infections (CROI); March 4–7, 2018; Boston, Massachusetts, USA.
Roy M, Holmes C, Sikazwe I, Savory T, Mwanza M, Bolton Moore C, et al. Application of a multistate model to evaluate visit burden and patient stability to improve sustainability of human immunodeficiency virus treatment in Zambia. Clin Infect Dis. 2018;67(8):1269–77.
Katchy UI EA, Ibrahim S, Usman AB, Magana MR, Ogundehin D, Ezekiel J. Client and health worker perception to differentiated HIV care: a qualitative study from Kebbi State, North West Nigeria (WEPEE766). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Obunga J OL, Kanyango B, Burmen B. Experiences and lessons learned in implementing the differentiated care model in HIV clinics in Nyamira County, Western Kenya, Jan -Sept 2017 (WEPEB135). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Mashungu V MJ, Khembo W, Nkawu B, Kumani B, Mugurungi O, Ngorima-Mabhena N Acceptability of community differentiated models of care: a patient and service provider’s perspective on community art refill groups in Zimbabwe (WEPEE759). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
T H. Mapping the scale-up of DSD: 13-country health facility survey. 22nd International AIDS Conference: Pre-conference Symposia; July 23, 2018; Amsterdam, Netherlands.
Decroo TLJ, Rasschaert F, Bermudez-Aza EH, Couto A, Candrinho B, Biot M, et al. Scaling up community ART groups in Mozambique. International STD Research and Reviews. 2013;1(2):49–59.
De Neve JW, Garrison-Desany H, Andrews KG, et al. Harmonization of community health worker programs for HIV: a four-country qualitative study in Southern Africa. PLoS Med. 2017;14(8):e1002374.
Geza S KP, Mdala J, McQuide P, Nepela A, Shipena R, Omukongo S, Kandume P, Velishavo F, Zegeye A, Hamunime N, Mazibuko G, Mwinga S, Sagwa E. The effect of differentiated care interventions on the workload of facilities providing ART in Onandjokwe district in Northern Namibia (THPED402). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Kamerhe D TC, Mwamba R, Thior I, Canagasabey D, Kiluba JC, Mueller Scott L. Impact of offering differentiated HIV care on treatment retention and health facility workload: results from Kenya health zone in the Democratic Republic of Congo (WEPEE749). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Nsanzimana S, Remera E, Ribakare M, Burns T, Dludlu S, Mills EJ, et al. Phased implementation of spaced clinic visits for stable HIV-positive patients in Rwanda to support treat all. J Int AIDS Soc. 2017;20(Suppl 4):21635.
Fusire TT ZM, Murangandi S, Fatti G, Ngorima-Mabhena N Supporting antiretroviral therapy multi-month scripting and dispensing at facilities in Zimbabwe: effect on supply chain management (WEPEE630). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Joseph Davey D SC, Luthuli T, Darkoh E. Uptake of HIV differentiated care models for patients on antiretroviral therapy in South Africa (WEPEE756). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Barker C DA, Klein K Can differentiated care models solve the crisis in treatment financing? Analysis of prospects for 38 high-burden countries in sub-Saharan Africa (WEAD0204). 9th IAS Conference on HIV Science; July 23–26, 2017; Paris, France.
Bango F, Ashmore J, Wilkinson L, van Cutsem G, Cleary S. Adherence clubs for long-term provision of antiretroviral therapy: cost-effectiveness and access analysis from Khayelitsha, South Africa. Tropical Med Int Health. 2016;21(9):1115–23.
Vu L, Waliggo S, Zieman B, Jani N, Buzaalirwa L, Okoboi S, et al. Annual cost of antiretroviral therapy among three service delivery models in Uganda. J Int AIDS Soc. 2016;19(5 Suppl 4):20840.
CIDRZ. Community ART for retention in Zambia: health economics evaluations. 2018; http://www.cidrz.org/wp-content/toolkits/commart/publications.html. Accessed 12 Dec 2018.
Bacha J AL, Wanless S, Ngo K, Campbell L, Shutze G. The standardized pediatric expedited encounters for ART drugs initiative: description and evaluation of an innovative pediatric ART health service delivery model in Tanzania (TUPED1321). 9th IAS Conference on HIV Science; July 23–26, 2017; Paris, France.
Kim M WR, Ahmed S, Mhango J, Damba D, Kayabu A, Chodota M, Dlamini S, Chida N, Mokhali M, Calles N, Amzel A, Golin R, Abrams EJ. Multi-month prescription of antiretroviral therapy and its feasibility: experiences from the Baylor International Pediatric AIDS initiative (BIPAI) in six southern African countries (MOAD0105). 9th IAS Conference on HIV Science; July 23–26, 2017; Paris, France.
MacKenzie RK, van Lettow M, Gondwe C, et al. Greater retention in care among adolescents on antiretroviral treatment accessing “Teen Club” an adolescent-centred differentiated care model compared with standard of care: a nested case-control study at a tertiary referral hospital in Malawi. J Int AIDS Soc. 2017;20(3).
Tsondai P WL, Henwood R, Ullauri A, Cassidy T, Tutu S, Davies MA. Retention and viral suppression outcomes of patients enrolled in family ART adherence clubs in Cape Town, South Africa (TUPED1325). 9th IAS Conference on HIV Science; July 23–26, 2017; Paris, France.
Myer L, Iyun V, Zerbe A, Phillips TK, Brittain K, Mukonda E, et al. Differentiated models of care for postpartum women on antiretroviral therapy in Cape Town, South Africa: a cohort study. J Int AIDS Soc. 2017;20(Suppl 4):21636.
Zerbe A BK, Phillips T, Iyun VO, Mukonda E, Abrams EJ, Myer L for the MCH-ART Study. Breastfeeding practices and infant care engagement among HIV-infected postpartum women on antiretroviral therapy (ART) attending community-based adherence clubs (ACs) in Cape Town, South Africa. 9th IAS Conference on HIV Science; July 23–26, 2017; Paris, France.
Nelson AK CT, Trivino Duran L, Hoxha A, Buchanan K, Shroufi A, Uenishi R, Matangana N, Mdani L, Mohr E, De Azevedo V, Abrahams S, Hofmeyr C, Igumbor J Post natal clubs: a differentiated model of care integrating maternal, child health and prevention of mother to child transmission (PMTCT) in Khayelitsha, South Africa (THPEE660). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Barrington C ILMM, Northbrook S, William J, Alvis JP, Leonardo Santa Luce R, Guzman Guevara K, Pinzon Meza R, Anton Urbina ME, Ricardo Calderon M. Decentralizing and differentiating HIV care for MSM living with HIV in Guatemala City: acceptability and retention in care (FRAE0103). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Alao O AA, Osisami O, Kalaiwo A, Tewobola O. Engaging community-based model of care for differentiated anti-retroviral therapy (ART) service delivery for people who inject drugs (PWIDS) in Nigeria: lessons and challenges (WEPED456). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Petlele R ML, Kganakga C Differentiated service delivery for harm reduction strategies for people who inject drugs_learnings from the Mauritian context (WEPED452). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Kamtimaleka M MB, Kamtumbiza F, Kumwenda G, Sikwese S, Mpunga E, Kamanga G, Ruberintwari M, Akolo C. Differentiated service delivery models for HIV testing among female sex workers: lessons from the LINKAGES project in Blantyre, Malawi. 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Oucul L BJ, David K Differentiated ART delivery model for female sex workers in Uganda: community client led ART delivery to improve outcomes (THPDD0206). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Kagimu D EJ, Oucul L, Senyonga E. Overcoming barriers to access of HIV/AIDS services among female sex workers through differentiated service delivery models, TASO Entebbe experience (WEPEE764). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Mwanda K CM, Mwila E, Lungu Y, Phiri D, Kawanga L, Banda M, Chibesakunda K, Chikondi D, Jose J ART behind bars: differentiated service delivery results in high viral load suppression among prisoners in Zambia (WEPEE750). 22nd International AIDS Conference; July 23–27, 2018; Amsterdam, Netherlands.
Beach MC, Keruly J, Moore RD. Is the quality of the patient-provider relationship associated with better adherence and health outcomes for patients with HIV? J Gen Intern Med. 2006;21(6):661–5.
Stewart MA. Effective physician-patient communication and health outcomes: a review. CMAJ. 1995;152(9):1423–33.
DiLiberto DD, Staedke SG, Nankya F, et al. Behind the scenes of the PRIME intervention: designing a complex intervention to improve malaria care at public health centres in Uganda. Glob Health Action. 2015;8:29067.
Horwood C, Butler L, Barker P, Phakathi S, Haskins L, Grant M, et al. A continuous quality improvement intervention to improve the effectiveness of community health workers providing care to mothers and children: a cluster randomised controlled trial in South Africa. Hum Resour Health. 2017;15(1):39.
Horwood CM, Youngleson MS, Moses E, Stern AF, Barker PM. Using adapted quality-improvement approaches to strengthen community-based health systems and improve care in high HIV-burden sub-Saharan African countries. AIDS. 2015;29(Suppl 2):S155–64.
Acknowledgments
The authors thank Megha Mehrotra for her technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Implementation Science
Rights and permissions
About this article
Cite this article
Roy, M., Bolton Moore, C., Sikazwe, I. et al. A Review of Differentiated Service Delivery for HIV Treatment: Effectiveness, Mechanisms, Targeting, and Scale. Curr HIV/AIDS Rep 16, 324–334 (2019). https://doi.org/10.1007/s11904-019-00454-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11904-019-00454-5