Abstract
Overt hepatic encephalopathy (OHE) and covert hepatic encephalopathy (CHE) are two clinically distinct entities that are at the end of the spectrum of cognitive impairment due to portal hypertension. They are fraught with diagnostic and treatment challenges, but both could result in serious complications if not intervened upon in a timely fashion. CHE has been recognized as a harbinger to OHE providing an opportunity to intervene. There are numerous diagnostic strategies for CHE, and most require specialized equipment, trained personnel, and time. Diagnosis of OHE is significantly easier compared to CHE. As such, the psychometric tests have been more favored due to less dependence on specialized equipment and personnel, and their ability to be equally reliable compared to neuropsychological tests. While we have learned more about the pathophysiology of hepatic encephalopathy, our treatment modalities have not evolved as much. A collaborative approach to management, with development of easier reliable tests, will help us combat this epidemic.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatic encephalopathy (HE) is a widely prevalent [1] serious complication of decompensated chronic liver disease (CLD) representing the neurological and psychiatric manifestation of porto-systemic shunting (PSS). It has been defined as “A brain dysfunction caused by liver insufficiency and or porto-systemic shunting” by the 2014 practice guidelines of the AALSD and EASL [2••]. The commonly practiced classification system that is based on clinical manifestations and specialized testing divides HE into OHE, the more obvious and recognizable form, and CHE, the more prevalent but not readily diagnosable form [3, 4]. CHE includes grade 1 and minimal HE (MHE) on the West Haven Scale (WHS) for HE [5••]. The other common classification systems for HE is based on underlying etiology [6]. Another classification for HE defines it based on duration into episodic, recurrent with repeated bouts in a 6-month time frame, and persistent, which defines a baseline altered behavior with interspersed relapses of OHE [6]. Since grade 1 HE and MHE are often difficult to distinguish clinically and have the same diagnostic approach, we shall be talking about CHE in this article. HE, in general, has an unpredictable clinical course in terms of onset and response to therapy, and it is a well-known independent predictor of mortality in cirrhotics. The estimated incidence of OHE in cirrhotics is 30–40 % during their lifetime [7] and MHE/CHE occur in 20–80 % of cirrhotics [4, 8–10].
Given the complexities in diagnosing and grading OHE, in an attempt to standardize management, the recent AASLD/EASL guidelines suggest evaluating HE as a spectrum along four axes—Axis 1: underlying reason: could be acute liver failure (type A), porto-systemic bypass (type B), and cirrhosis (type C); Axis 2: severity of the current episode: covert (minimal + grade I), overt (grades II–IV of the West Haven Criteria); Axis 3: time course (episodic, one episode in 6 months; recurrent, >1 episodes within 6 months; and persistent, never resolved fully); and Axis 4: spontaneous or precipitated based on the identification of precipitating factors. Axes 3 and 4 only apply to those with overt HE.
Despite optimal lactulose treatment, OHE can recur in 40 % of patients in 6 months [11•]. No data regarding CHE recurrence has been documented so far. CHE has been explored by various groups over the past decade and is now a well-recognized clinical entity. The importance and urgency of diagnosis of MHE/CHE lie in the fact that it is a predictor of impending OHE [9, 12, 13]. Therefore, the epidemiological and clinical burden of MHE/CHE is equally important as that of OHE, and its diagnosis forebears a poor prognosis as it has been shown as an independent predictor of mortality in cirrhosis itself [14].
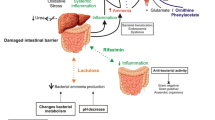
In this article, we shall be discussing the diagnosis of OHE based on current standards, the diagnostic challenges of MHE and CHE, and subsequently discuss the current therapeutic options for treating HE (Fig. 1).
OHE—Diagnosis and WHS Testing
Diagnosis
Diagnosis of OHE is primarily by clinical manifestations based on the WHS which grades impaired mental function and impaired neuromotor functions. The spectrum of impaired mental function varies from agitation to coma, while the presence of asterixis is commonly noted in early stages of OHE. As such, HE is a diagnosis of exclusion and hence clinicians need to perform a detailed history, examination, and pertinent laboratory investigations to exclude the extensive differentials of HE.
Biochemical Tests
HE is associated with upregulated neuro-inflammation and neurotoxicity related to hyperammonemia and other toxic metabolites. Elevated levels of interleukins have been documented in acute and chronic liver failure and MHE [15, 16], but do not really have any clinical significance for testing. Serum ammonia, on the other hand, is a widely accepted screening test for HE though it lacks specificity. Venous ammonia levels are directly affected by the method of obtaining the sample and if the sample was placed on ice. Additionally, there are multiple other non-liver-related causes of hyperammonemia such as rare congenital urea cycle disorders, to more commonly heavy exercise, GI bleeding, shock, renal disease, total parenteral nutrition, and drugs (valproic acid, salicylates, narcotics, diuretics). An increased ammonia level does not add to changes in mental functioning for the diagnosis of HE; however, in a confused cirrhotic patient, a normal ammonia should lead to reconsideration of the HE diagnosis.
Grading
West Haven Criteria (Conn Score)
The WHC is the most commonly practiced grading system which was envisaged as a semi-quantitative system. Therefore, it has subjective portions which can impair its reliability in lower grades of HE. In order to improve inter-observer variability, covert HE, which combines minimal and grade I (the most subjective stage), has been defined, while overt HE has been defined as patients with disorientation and asterixis who require additional or modification of current therapy. Therefore, the staging of grade II–IV is more reliable, although the Glasgow Coma Scale could be used to define the advanced stages further. Over the years, the need for a more objective scoring system has led to the development of alternatives.
Hepatic Encephalopathy Scoring Algorithm (HESA)
This scoring algorithm developed by Hassenein et al. refines the WHS into four grades through the incorporation of neuropsychological testing and clinical indicators. It is significantly more detailed, objective, has lower inter-observer variability, and hence is an effective tool for HE diagnosis in the research setting [17]. It was validated by the same group in a large clinical trial across multiple sites and proves to be a reliable diagnostic tool for grade I/II HE for the diagnosis of initial and recurrent HE. Though it is a simple, consistent test with good sensitivity, it is a long test to administer and hence is limited in use to clinical trials and not day-to-day clinical practice.
Modified-Orientation Log (MO-log)
This MO-log was developed from the traumatic brain injury literature to refine the overt HE severity [18]. It focuses on a standardized orientation questionnaire, keeping in mind that disorientation to time and person are early cognitive defects in HE, therefore exploring the level of disorientation [19•]. The strengths of the MO-log test lie in its reproducibility, sensitivity to cognitive impairment, predictive ability for HE patients that are at risk for in hospital mortality and at risk for decompensation, and most importantly clinical applicability. The disadvantage is the reliance on orientation as the sole definition of altered mental status.
Clinical Hepatic Encephalopathy Staging Scale (CHESS)
The CHESS was developed by Ortiz et al. to standardize observations, incorporating the WHC and GCS, being more definitive [20•]. It is relatively simpler to administer and is useful to monitor HE severity on a scale where 0 (minimal score) signifies normalcy and 9 (maximal score) signifies coma. It has been validated by two large RCTs to be effective in earlier stages of HE [21•, 22].
Full Outline of UnResponsiveness (FOUR) Score
Assessment of the level of consciousness in HE does not have a standardized method. The FOUR score was developed and has been endorsed to assess the level of consciousness in comatose patients in general [23]. The FOUR score incorporates four components—visual and motor reflexes, eliminates verbal response, but incorporates assessment of brainstem reflex and breathing patterns. Mouri et al. in a prospective trial compared the Glasgow Coma Scale (GCS) to the FOUR score and validated this score showing that it is not only effective to diagnose HE but also predicts HE (WHC grade 2–4) and has the ability to discriminate between the grades of OHE. The study also showed that the score effectively predicts mortality for scores <16 [24].
Mini-Mental Status Examination (MMSE)
The MMSE was initially developed in 1975 as a test to assess the neurocognitive status to diagnose dementia [25]. It has since then been studied extensively in outpatients with HE to ensure that they are able to understand cognitive tests and to exclude dementia. It is not a test for inpatients with OHE [26].
Glasgow Coma Scale (GCS)
The GCS is a scale that is utilized for all comatose patients and can be adapted for assessing mental status in advanced grades of HE. It is an objective scoring system that has not been studied extensively in HE per se but can still be used in grade III and IV HE. It has no diagnostic value for grade II HE and below [27•].
Treatment
A four-pronged approach should be taken to manage OHE [2••].
1. Initiation of care for patients with altered consciousness. 2. Evaluation for alternative etiologies of altered mental status and treating them. 3. Identification of precipitating factors and their correction, as a majority of patients can be managed with just correction of the precipitating factor [28]. 4. Initiation of empirical HE therapy. Treatment strategies (Table 1) discussed will be for an initial episode of OHE and for recurrent OHE. Management of post-TIPS HE and HE related to porto-systemic shunts are beyond the scope of this article.
-
1.
Non-absorbable disaccharides:
The most widely used drug of this class is lactulose. It has been studied extensively and works primarily by reducing the production and absorption of ammonia in the gut. Lactulose is the backbone of therapy for HE and is used in the management of initial and recurrent HE for secondary prophylaxis [11•]. General consensus for dosing of lactulose is 15–30 ml two to three times a day with a goal of having two to three soft or loose bowels a day. One needs to be cautious in directions of use as overuse can result in dehydration and can rarely precipitate HE itself [29].
-
2.
Antibiotics:
-
(a)
Rifaximin: Rifaximin is a broad-spectrum gut-specific antimicrobial agent. It Is FDA approved for the prevention of recurrent HE in conjunction with lactulose. It has very limited gut absorption and achieves a great concentration in the gastrointestinal tract. It works by inhibiting bacterial protein synthesis without grossly affecting the composition of the normal gut flora [30]. It has also been studied (along with lactulose) versus lactulose [31] and versus placebo [32••] for the management of recurrent HE and has shown to be effective, but has not been validated independently. It additionally has been shown to improve HRQOL in OHE [33].
-
(b)
Aminoglycosides: The drugs in this class that have been studied, i.e., neomycin and ribostamycin, are FDA approved for OHE treatment, but have multiple systemic side effects (nephrotoxicity, ototoxicity) and hence are not recommended.
-
(c)
Miscellaneous antibiotics: The other non-FDA-approved antibiotics that have been studied in OHE are metronidazole, vancomycin, and paromomycin. Metronidazole was studied against neomycin [34], vancomycin was studied against lactulose [35], and paromomycin was studied against rifaximin [36, 37], but side effects, cost, potential for resistant bacteria, and limited data make these options less favorable.
-
(a)
-
3.
Other non-FDA-approved treatment strategies:
Many novel approaches to management of HE have been investigated. Many of them have been proven to have some effect on control of symptoms, but lack sufficient evidence. These drugs could be tried in the appropriate setting, along with ongoing current standards of treatment. Glycerol phenyl butyrate (GPB) [21•], branched chain amino acids (BCAA) [38, 39], l-ornithine l-aspartate (LOLA) [40], and even albumin [22] have been studied with minimal side effects. Liver assist devices have not demonstrated any mortality benefit in chronic liver disease.
-
4.
Liver transplant: This should always be considered in a patient with HE given that this is the only lasting cure for cirrhosis.
CHE—Diagnosis and Testing
CHE is the equally relevant, more prevalent but unfortunately lesser diagnosed form of HE. It faces many diagnostic challenges owing to the difficulty in diagnosing it using simple clinical examination, and hence often goes unrecognized [41]. It, however, has a significant impact on the health-related quality of life (HRQOL), development rate of overt HE, and on the hospitalization and survival in cirrhotic patients [42, 43]. Therefore, efforts must be made to diagnose this entity to the fullest extent [44]. Clinicians are gradually getting more aware to this entity, but testing modalities are not yet standardized and are limited by availability, cost, and local population’s normative data [45] and as such are not really suitable for regular outpatient clinics. Alternative strategies are to use high-sensitivity tests in clinics that can then be used to guide further diagnosis at a later stage in patients with cirrhosis.
The diagnostic testing would be most productive when focused on patients who would suffer the most if diagnosed with CHE. This includes patients with cognitive complaints, those with poor HRQOL, those who are current drivers, and those who are currently employed. Screening strategies could be the use of four questions of the Sickness Impact Profile [46] or the use of the Stroop EncephalApp.
Broadly, tests for CHE are divided into psychometric and neurophysiological tests. Psychometric tests are divided into simple paper-and-pencil tests and computerized tests (Table 2). The majority of tests are easy to administer after training appropriate staff, but are lengthy for day-to-day clinical practice. Per the International Society for Hepatic Encephalopathy and Nitrogen Metabolism (ISHEN) 2 tests, the Psychometric Hepatic Encephalopathy Score (PHES) and the Repeatable Battery for the Assessment of Neurophysiological status are the preferable paper-and-pencil tests [45]. Computerized tests, clinically, are slightly more advantageous over paper-and-pencil tests owning to standardization and lesser use of motor functions. Since the changes to cognition and QOL are fine, no clinically valid grading system has been developed yet. For the purpose of practicality, we shall discuss the tests based on the need for specialized equipment and specialists.
Diagnosis
Biochemical Tests
There is no role for serum markers for the diagnosis of CHE, as patients are often normal on general examination.
Test That Do Not Require Specialized Equipment and Trained Personnel
Inhibitory Control Test (ICT)
The ICT is a computerized test originally developed to detect deficits in attention and response inhibition in schizophrenia, traumatic brain injury, and attention deficit disorder in children. It has been applied successfully to diagnose CHE and predict OHE in CLD patients, with a sensitivity and specificity of 90 % [47]. It essentially is a computerized test that shows a sequence of letters on a computer screen every 500 ms without any intervals. The subject is then instructed to respond to predetermined alternating patterns of the letters X and Y (targets) and non-alternating presentations of the letters X and Y (lures), and the response times and responses are monitored and scored. This test has good external validity and is easy to administer, but requires a fully functional patient. The ICT has been improved by Amodio et al. to detect MHE based on target accuracy and lures weighted by target accuracy, and appeared to be more sensitive for MHE detection than the original test studies by Bajaj et al. [48]. A positive test correlates with traffic accidents related to MHE [49]. It is readily available for download at the website www.hecme.tv.
Cognitive Drug Research (CDR)
The CDR test is a computerized neuropsychometric test that is not available in the USA. It is a computerized test that was originally developed to assess the cognitive functions such as choice reaction time and word and picture recognition in demented patients. It was adapted to CLD patients and assessed five major areas—power and continuity of attention, speed and quality of working memory, and quality of episodic memory. It has been assessed in HE patients [50] and has proven to be a simple test, not requiring any special expertise, but takes up to 20 min requiring functional patients. It does not have norms for the USA.
Scan Test
The Scan test is also a computerized test that measures reaction time to visual stimuli, reaction time to choice, and scan reaction time based on the Sternberg paradigm. It is not available in the USA, but has been validated in Europe as a good psychometric test that prognosticates cirrhosis [51]. It has also been endorsed as a test that is clinically useful in CHE diagnosis [52].
EncephalApp Stroop Test
The EncephalApp Stroop test is another neuropsychometric test of psychomotor speed and cognitive flexibility, with good sensitivity in the diagnosis of CHE [53].
EncephalApp_Stroop is a reliable and valid test that can be downloaded and administered on any Apple smartphone, in any clinic, with an excellent sensitivity for the detection of the cognitive changes of CHE [54••]. It is limited to use in patients who are not color blind.
Tests That Require Specialized Equipment and/or Trained Personnel
Psychometric Hepatic Encephalopathy Score (PHES/PSE)
The PHES, developed by Weissenborn and colleagues at the Hanover Medical School in Germany [55••], is the most recommended test by various consensus groups [2••, 6]. It essentially is a battery of five paper-and-pencil tests, namely (1) Number connection test A (NCT A), (2) Number connection test B (NCT B), (3) Line tracing test, (4) Serial dotting test, and (5) Digital symbol test. These subtests effectively evaluate cognitive motor abilities and visuo-motor coordination, functions that are affected early in HE. To be positive for CHE, either the score must be significant or two out of four of the individual tests need to be abnormal [6]. The PSE, unfortunately, has limited use outside Germany, Spain, Mexico, and Italy due to the absence of comparative data from the normal population. The test has a good sensitivity (96 %) and great specificity (100 %), but lacks test-retest reliability and is prone to multiple biases during testing [56••]. In an ideal setting, the test should be completed within 20–25 min.
Critical Flicker Frequency (CFF) Test
The CFF test is a neurophysiological test where visual signals and cognitive functions are evaluated. The CFF itself is the frequency at which steady fused light begins to flicker. A CFF below 39 Hz is diagnostic for CHE [57]. It also has application in the assessment of recovery of cirrhotic patients post-sedation for endoscopy [58]. The CFF test had excellent diagnostic value and predictive value for grade II and I HE [57], but requires specialized equipment, binocular vision, absence of red-green color blindness, and cooperation. Barring this, it is a simple-to-administer, reliable tool for CHE diagnosis.
Recently, based on the principle that eye movements are affected in HE, Cunniffe et al. showed that saccadic eye movements are altered in CHE and can prove as an accurate, simple diagnostic tool with 75 % sensitivity and specificity [59].
Electroencephalogram
The EEG is a simple test that does not require patient involvement. It is a neurophysiological test that studies the cortical cerebral activity, and in the advanced stages of OHE, a triphasic wave is classically seen [60]. EEG classifies HE into five grades of severity as well and requires the expertise of a neurologist to read the tracings. This can lead to inter-observer variability, and hence to eliminate this computerized spectral analysis of the readings is preferred [61, 62]. Spectral analysis of EEG can predict CHE by detecting cerebral alterations when no clinical symptomatology is seen [63]. The limitations of using EEG in routine practice are the requirement of specialized equipment and the presence of underlying confounding metabolic disturbances.
Evoked Potentials
Evoked potentials (auditory, visual, and somatosensory) are specialized tests with limited value for OHE diagnosis as they require more patient involvement as opposed to a simple EEG. The principle is that on appropriate stimuli, the brain will show a corresponding EEG response. A delayed response would suggest brain impairment. It has been studied in CHE but have not gained favor due to various drawbacks [64].
Treatment of CHE
The AASLD and other organizations do not recommend routine treatment of CHE [2••]. The current recommendation is evaluation of individual cases and then offering the current treatment standards for OHE. However, rifaximin has been studied in CHE and has been shown to improve cognitive performance and HRQOL in patients screening positive for CHE [65]. It has also been shown to improve driving simulator performance in CHE patients [66]. Apart from its known mechanism of action on OHE, it has been shown to reduce endotoxemia and regulate the metabiome in CHE patients [67].
Conclusions
The diagnosis and management of HE is an evolving field. Due to the complexity of pathophysiology of this disease process, there is not one single test that can be nominated as a true gold standard. Riggio et al. recently proposed a possible model for prediction of OHE [68]. This model only highlighted that there is not a single test that could predict the clinical course of HE. This is reflective of the spectrum of neurocognitive impairment in cirrhosis (SONIC) and the challenges researchers face. There is also a need to develop alternative cost-effective therapies to lactulose and rifaximin. Over the last decade, the focus has shifted on MHE/CHE as it presents an opportunity to intervene earlier. Given the subtle nature of CHE, the majority of tests require specialized equipment and expertise, posing a challenge in routine clinics to accurately and timely diagnose CHE. Despite extensive research here, there are no guidelines for management of CHE. Given the risks of CHE, it is our advice that patients should be screened for CHE and, if needed, appropriate referrals be given to higher centers that have the right tools for diagnosis. In time as our understanding deepens, we can conclude that the future of HE definitely looks promising.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Albrecht J. Hepatic encephalopathy in our genes? Ann Intern Med. 2010;153(5):335–6. doi:10.7326/0003-4819-153-5-201009070-00008.
Vilstrup H, Amodio P, Bajaj J, Cordoba J, Ferenci P, Mullen KD, et al. Hepatic encephalopathy in chronic liver disease: 2014 Practice Guideline by the American Association for the Study of Liver Diseases and the European Association for the Study of the Liver. Hepatology. 2014;60(2):715–35. doi:10.1002/hep.27210. AASLD/EASL guidelines on hepatic encephalopathy released in 2014.
Saxena N, Bhatia M, Joshi YK, Garg PK, Tandon RK. Auditory P300 event-related potentials and number connection test for evaluation of subclinical hepatic encephalopathy in patients with cirrhosis of the liver: a follow-up study. J Gastroenterol Hepatol. 2001;16(3):322–7.
Schomerus H, Hamster W. Quality of life in cirrhotics with minimal hepatic encephalopathy. Metab Brain Dis. 2001;16(1–2):37–41.
Bajaj JS, Cordoba J, Mullen KD, Amodio P, Shawcross DL, Butterworth RF, et al. Review article: The design of clinical trials in hepatic encephalopathy—an International Society for Hepatic Encephalopathy and Nitrogen Metabolism (ISHEN) consensus statement. Aliment Pharmacol Ther. 2011;33(7):739–47. doi:10.1111/j.1365-2036.2011.04590.x. ISHEN guidelines for conducting clinical trials in HE introducing the concept of covert HE.
Ferenci P, Lockwood A, Mullen K, Tarter R, Weissenborn K, Blei AT. Hepatic encephalopathy—definition, nomenclature, diagnosis, and quantification: final report of the working party at the 11th World Congresses of Gastroenterology, Vienna, 1998. Hepatology. 2002;35(3):716–21. doi:10.1053/jhep.2002.31250.
Amodio P, Del Piccolo F, Petteno E, Mapelli D, Angeli P, Iemmolo R, et al. Prevalence and prognostic value of quantified electroencephalogram (EEG) alterations in cirrhotic patients. J Hepatol. 2001;35(1):37–45.
Groeneweg M, Moerland W, Quero JC, Hop WC, Krabbe PF, Schalm SW. Screening of subclinical hepatic encephalopathy. J Hepatol. 2000;32(5):748–53.
Romero-Gomez M, Cordoba J, Jover R, del Olmo JA, Ramirez M, Rey R, et al. Value of the critical flicker frequency in patients with minimal hepatic encephalopathy. Hepatology. 2007;45(4):879–85. doi:10.1002/hep.21586.
Lauridsen MM, Jepsen P, Vilstrup H. Critical flicker frequency and continuous reaction times for the diagnosis of minimal hepatic encephalopathy: a comparative study of 154 patients with liver disease. Metab Brain Dis. 2011;26(2):135–9. doi:10.1007/s11011-011-9242-1.
Sharma BC, Sharma P, Agrawal A, Sarin SK. Secondary prophylaxis of hepatic encephalopathy: an open-label randomized controlled trial of lactulose versus placebo. Gastroenterology. 2009;137(3):885–91, 91.e1. doi:10.1053/j.gastro.2009.05.056. Randomized trial showing lactulose is better than placebo in preventing OHE recurrence
Das A, Dhiman RK, Saraswat VA, Verma M, Naik SR. Prevalence and natural history of subclinical hepatic encephalopathy in cirrhosis. J Gastroenterol Hepatol. 2001;16(5):531–5.
Saxena N, Bhatia M, Joshi YK, Garg PK, Dwivedi SN, Tandon RK. Electrophysiological and neuropsychological tests for the diagnosis of subclinical hepatic encephalopathy and prediction of overt encephalopathy. Liver. 2002;22(3):190–7.
Amodio P, Del Piccolo F, Marchetti P, Angeli P, Iemmolo R, Caregaro L, et al. Clinical features and survival of cirrhotic patients with subclinical cognitive alterations detected by the number connection test and computerized psychometric tests. Hepatology. 1999;29(6):1662–7. doi:10.1002/hep.510290619.
Genesca J, Gonzalez A, Segura R, Catalan R, Marti R, Varela E, et al. Interleukin-6, nitric oxide, and the clinical and hemodynamic alterations of patients with liver cirrhosis. Am JGastroenterol. 1999;94(1):169–77. doi:10.1111/j.1572-0241.1999.00790.x.
Montoliu C, Piedrafita B, Serra MA, del Olmo JA, Urios A, Rodrigo JM, et al. IL-6 and IL-18 in blood may discriminate cirrhotic patients with and without minimal hepatic encephalopathy. J Clin Gastroenterol. 2009;43(3):272–9. doi:10.1097/MCG.0b013e31815e7f58.
Hassanein TI, Hilsabeck RC, Perry W. Introduction to the Hepatic Encephalopathy Scoring Algorithm (HESA). Dig Dis Sci. 2008;53(2):529–38. doi:10.1007/s10620-007-9895-0.
Jackson WT, Novack TA, Dowler RN. Effective serial measurement of cognitive orientation in rehabilitation: the Orientation Log. Arch Phys Med Rehabil. 1998;79(6):718–20.
Salam M, Matherly S, Farooq IS, Stravitz RT, Sterling RK, Sanyal AJ, et al. Modified-orientation log to assess hepatic encephalopathy. Aliment Pharmacol Ther. 2012;35(8):913–20. doi:10.1111/j.1365-2036.2012.05038.x. Modified Orientation log and outcomes in cirrhosis.
Ortiz M, Cordoba J, Doval E, Jacas C, Pujadas F, Esteban R, et al. Development of a clinical hepatic encephalopathy staging scale. Aliment Pharmacol Ther. 2007;26(6):859–67. doi:10.1111/j.1365-2036.2007.03394.x. Clinical Hepatic Encephalopathy Staging Scale for outcomes.
Rockey DC, Vierling JM, Mantry P, Ghabril M, Brown Jr RS, Alexeeva O, et al. Randomized, double-blind, controlled study of glycerol phenylbutyrate in hepatic encephalopathy. Hepatology. 2014;59(3):1073–83. doi:10.1002/hep.26611. Glyceryl phenylbutyrate for prevention of recurrent HE after the second episode.
Simon-Talero M, Garcia-Martinez R, Torrens M, Augustin S, Gomez S, Pereira G, et al. Effects of intravenous albumin in patients with cirrhosis and episodic hepatic encephalopathy: a randomized double-blind study. J Hepatol. 2013;59(6):1184–92. doi:10.1016/j.jhep.2013.07.020.
Wijdicks EF, Bamlet WR, Maramattom BV, Manno EM, McClelland RL. Validation of a new coma scale: the FOUR score. Ann Neurol. 2005;58(4):585–93. doi:10.1002/ana.20611.
Mouri S, Tripon S, Rudler M, Mallet M, Mayaux J, Thabut D, et al. FOUR score, a reliable score for assessing overt hepatic encephalopathy in cirrhotic patients. Neurocrit Care. 2015;22(2):251–7. doi:10.1007/s12028-014-0078-5.
Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189–98.
Koziarska D, Wunsch E, Milkiewicz M, Wojcicki M, Nowacki P, Milkiewicz P. Mini-Mental State Examination in patients with hepatic encephalopathy and liver cirrhosis: a prospective, quantified electroencephalography study. BMC Gastroenterol. 2013;13:107. doi:10.1186/1471-230X-13-107.
Hassanein T, Blei AT, Perry W, Hilsabeck R, Stange J, Larsen FS, et al. Performance of the hepatic encephalopathy scoring algorithm in a clinical trial of patients with cirrhosis and severe hepatic encephalopathy. Am J Gastroenterol. 2009;104(6):1392–400. doi:10.1038/ajg.2009.160. Hepatic Encephalopathy Scoring Algorithm for refining HE grading.
Strauss E, Tramote R, Silva EP, Caly WR, Honain NZ, Maffei RA, et al. Double-blind randomized clinical trial comparing neomycin and placebo in the treatment of exogenous hepatic encephalopathy. Hepato-Gastroenterology. 1992;39(6):542–5.
Bajaj JS, Sanyal AJ, Bell D, Gilles H, Heuman DM. Predictors of the recurrence of hepatic encephalopathy in lactulose-treated patients. Aliment Pharmacol Ther. 2010;31(9):1012–7. doi:10.1111/j.1365-2036.2010.04257.x.
Brigidi P, Swennen E, Rizzello F, Bozzolasco M, Matteuzzi D. Effects of rifaximin administration on the intestinal microbiota in patients with ulcerative colitis. J Chemother. 2002;14(3):290–5. doi:10.1179/joc.2002.14.3.290.
Sharma BC, Sharma P, Lunia MK, Srivastava S, Goyal R, Sarin SK. A randomized, double-blind, controlled trial comparing rifaximin plus lactulose with lactulose alone in treatment of overt hepatic encephalopathy. Am J Gastroenterol. 2013;108(9):1458–63. doi:10.1038/ajg.2013.219.
Bass NM, Mullen KD, Sanyal A, Poordad F, Neff G, Leevy CB, et al. Rifaximin treatment in hepatic encephalopathy. N Engl J Med. 2010;362(12):1071–81. doi:10.1056/NEJMoa0907893. Pivotal trial with rifaximin in preventing recurrence after the second HE episode.
Sanyal A, Younossi ZM, Bass NM, Mullen KD, Poordad F, Brown RS, et al. Randomised clinical trial: rifaximin improves health-related quality of life in cirrhotic patients with hepatic encephalopathy—a double-blind placebo-controlled study. Aliment Pharmacol Ther. 2011;34(8):853–61. doi:10.1111/j.1365-2036.2011.04808.x.
Morgan MH, Read AE, Speller DC. Treatment of hepatic encephalopathy with metronidazole. Gut. 1982;23(1):1–7.
Tarao K, Ikeda T, Hayashi K, Sakurai A, Okada T, Ito T, et al. Successful use of vancomycin hydrochloride in the treatment of lactulose resistant chronic hepatic encephalopathy. Gut. 1990;31(6):702–6.
De Marco F, Sanatamaria Amato P, D’arienzo A. Rifaximin in collateral treatment of port-systemic encephalopathy: a preliminary report. Curr Ther Res. 1984;36(4):668–74.
Parini P, Cipolla A, Ronchi M, Salzetta A, Mazzella G, Roda E. Effect of rifaximin and paromomycin in the treatment of portal-systemic encephalopathy. Curr Ther Res. 1992;52(1):34–9.
Gluud LL, Dam G, Borre M, Les I, Cordoba J, Marchesini G, et al. Lactulose, rifaximin or branched chain amino acids for hepatic encephalopathy: what is the evidence? Metab Brain Dis. 2013;28(2):221–5. doi:10.1007/s11011-012-9372-0.
Gluud LL, Dam G, Borre M, Les I, Cordoba J, Marchesini G, et al. Oral branched-chain amino acids have a beneficial effect on manifestations of hepatic encephalopathy in a systematic review with meta-analyses of randomized controlled trials. J Nutr. 2013;143(8):1263–8. doi:10.3945/jn.113.174375.
Kircheis G, Nilius R, Held C, Berndt H, Buchner M, Gortelmeyer R, et al. Therapeutic efficacy of L-ornithine-L-aspartate infusions in patients with cirrhosis and hepatic encephalopathy: results of a placebo-controlled, double-blind study. Hepatology. 1997;25(6):1351–60. doi:10.1002/hep.510250609.
Bajaj JS, Etemadian A, Hafeezullah M, Saeian K. Testing for minimal hepatic encephalopathy in the United States: an AASLD survey. Hepatology. 2007;45(3):833–4. doi:10.1002/hep.21515.
Groeneweg M, Quero JC, De Bruijn I, Hartmann IJ, Essink-bot ML, Hop WC, et al. Subclinical hepatic encephalopathy impairs daily functioning. Hepatology. 1998;28(1):45–9. doi:10.1002/hep.510280108.
Prakash RK, Mullen KD. Is poor quality of life always present with minimal hepatic encephalopathy? Liver Int. 2011;31(7):908–10. doi:10.1111/j.1478-3231.2011.02532.x.
Bajaj JS, Pinkerton SD, Sanyal AJ, Heuman DM. Diagnosis and treatment of minimal hepatic encephalopathy to prevent motor vehicle accidents: a cost-effectiveness analysis. Hepatology. 2012;55(4):1164–71. doi:10.1002/hep.25507.
Randolph C, Hilsabeck R, Kato A, Kharbanda P, Li YY, Mapelli D, et al. Neuropsychological assessment of hepatic encephalopathy: ISHEN practice guidelines. Liver Int. 2009;29(5):629–35. doi:10.1111/j.1478-3231.2009.02009.x.
Nabi E, Thacker LR, Wade JB, Sterling RK, Stravitz RT, Fuchs M, et al. Diagnosis of covert hepatic encephalopathy without specialized tests. Clin Gastroenterol Hepatol. 2014;12(8):1384.e2–9.e2. doi:10.1016/j.cgh.2013.12.020.
Bajaj JS, Saeian K, Verber MD, Hischke D, Hoffmann RG, Franco J, et al. Inhibitory control test is a simple method to diagnose minimal hepatic encephalopathy and predict development of overt hepatic encephalopathy. Am J Gastroenterol. 2007;102(4):754–60. doi:10.1111/j.1572-0241.2007.01048.x.
Amodio P, Ridola L, Schiff S, Montagnese S, Pasquale C, Nardelli S et al. Improving the inhibitory control task to detect minimal hepatic encephalopathy. Gastroenterology. 2010;139(2):510–8, 8 e1-2. doi:10.1053/j.gastro.2010.04.057.
Bajaj JS, Hafeezullah M, Zadvornova Y, Martin E, Schubert CM, Gibson DP, et al. The effect of fatigue on driving skills in patients with hepatic encephalopathy. Am JGastroenterol. 2009;104(4):898–905. doi:10.1038/ajg.2009.7.
Mardini H, Saxby BK, Record CO. Computerized psychometric testing in minimal encephalopathy and modulation by nitrogen challenge and liver transplant. Gastroenterology. 2008;135(5):1582–90. doi:10.1053/j.gastro.2008.06.043.
Montagnese S, Schiff S, Turco M, Bonato CA, Ridola L, Gatta A, et al. Simple tools for complex syndromes: a three-level difficulty test for hepatic encephalopathy. Dig Liver Dis. 2012;44(11):957–60. doi:10.1016/j.dld.2012.06.010.
Sakamoto M, Perry W, Hilsabeck RC, Barakat F, Hassanein T. Assessment and usefulness of clinical scales for semiquantification of overt hepatic encephalopathy. Clin Liver Dis. 2012;16(1):27–42. doi:10.1016/j.cld.2011.12.005.
Amodio P, Schiff S, Del Piccolo F, Mapelli D, Gatta A, Umilta C. Attention dysfunction in cirrhotic patients: an inquiry on the role of executive control, attention orienting and focusing. Metab Brain Dis. 2005;20(2):115–27.
Bajaj JS, Thacker LR, Heuman DM, Fuchs M, Sterling RK, Sanyal AJ, et al. The Stroop smartphone application is a short and valid method to screen for minimal hepatic encephalopathy. Hepatology. 2013;58(3):1122–32. doi:10.1002/hep.26309. Stroop application for the diagnosis of minimal HE.
Weissenborn K, Ennen JC, Schomerus H, Ruckert N, Hecker H. Neuropsychological characterization of hepatic encephalopathy. J Hepatol. 2001;34(5):768–73. Describing the Psychometric Hepatic Encephalopathy Score for minimal HE.
Torlot FJ, McPhail MJ, Taylor-Robinson SD. Meta-analysis: The diagnostic accuracy of critical flicker frequency in minimal hepatic encephalopathy. Aliment Pharmacol Ther. 2013;37(5):527–36. doi:10.1111/apt.12199. Critical flicker frequency for the diagnosis of minimal HE.
Sharma P, Sharma BC, Sarin SK. Critical flicker frequency for diagnosis and assessment of recovery from minimal hepatic encephalopathy in patients with cirrhosis. Hepatobiliary Pancreat Dis Int. 2010;9(1):27–32.
Sharma P, Singh S, Sharma BC, Kumar M, Garg H, Kumar A, et al. Propofol sedation during endoscopy in patients with cirrhosis, and utility of psychometric tests and critical flicker frequency in assessment of recovery from sedation. Endoscopy. 2011;43(5):400–5. doi:10.1055/s-0030-1256182.
Cunniffe N, Munby H, Chan S, Saatci D, Edison E, Carpenter RH, et al. Using saccades to diagnose covert hepatic encephalopathy. Metab Brain Dis. 2015. doi:10.1007/s11011-014-9647-8.
Van der Rijt CC, Schalm SW, De Groot GH, De Vlieger M. Objective measurement of hepatic encephalopathy by means of automated EEG analysis. Electroencephalogr Clin Neurophysiol. 1984;57(5):423–6.
Amodio P, Pellegrini A, Ubiali E, Mathy I, Piccolo FD, Orsato R, et al. The EEG assessment of low-grade hepatic encephalopathy: comparison of an artificial neural network-expert system (ANNES) based evaluation with visual EEG readings and EEG spectral analysis. Clin Neurophysiol. 2006;117(10):2243–51. doi:10.1016/j.clinph.2006.06.714.
Amodio P, Marchetti P, Del Piccolo F, de Tourtchaninoff M, Varghese P, Zuliani C, et al. Spectral versus visual EEG analysis in mild hepatic encephalopathy. Clin Neurophysiol. 1999;110(8):1334–44.
Kullmann F, Hollerbach S, Lock G, Holstege A, Dierks T, Scholmerich J. Brain electrical activity mapping of EEG for the diagnosis of (sub)clinical hepatic encephalopathy in chronic liver disease. Eur J Gastroenterol Hepatol. 2001;13(5):513–22.
Montagnese S, Amodio P, Morgan MY. Methods for diagnosing hepatic encephalopathy in patients with cirrhosis: a multidimensional approach. Metab Brain Dis. 2004;19(3–4):281–312.
Sidhu SS, Goyal O, Mishra BP, Sood A, Chhina RS, Soni RK. Rifaximin improves psychometric performance and health-related quality of life in patients with minimal hepatic encephalopathy (the RIME Trial). Am J Gastroenterol. 2011;106(2):307–16. doi:10.1038/ajg.2010.455.
Bajaj JS, Heuman DM, Wade JB, Gibson DP, Saeian K, Wegelin JA, et al. Rifaximin improves driving simulator performance in a randomized trial of patients with minimal hepatic encephalopathy. Gastroenterology. 2011;140(2):478–87. doi:10.1053/j.gastro.2010.08.061.
Bajaj JS, Heuman DM, Sanyal AJ, Hylemon PB, Sterling RK, Stravitz RT, et al. Modulation of the metabiome by rifaximin in patients with cirrhosis and minimal hepatic encephalopathy. PLoS One. 2013;8(4), e60042. doi:10.1371/journal.pone.0060042.
Riggio O, Amodio P, Farcomeni A, Merli M, Nardelli S, Pasquale C, et al. A model for predicting development of overt hepatic encephalopathy in patients with cirrhosis. Clin Gastroenterol Hepatol. 2015. doi:10.1016/j.cgh.2014.12.025.
Compliance with Ethics Guidelines
Conflict of Interest
Chathur Acharya declares no conflict of interest.
Jasmohan S. Bajaj has been a consultant for Salix, Merz, Norgine, and Grifols, unrelated to this article.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Portal Hypertension
Rights and permissions
About this article
Cite this article
Acharya, C.A., Bajaj, J.S. Covert and Overt Hepatic Encephalopathy: Current Options for Diagnosis and Treatment. Curr Hepatology Rep 14, 234–242 (2015). https://doi.org/10.1007/s11901-015-0277-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11901-015-0277-3