Abstract
Purpose of Review
Type 2 diabetes is a growing concern worldwide with increasing incidence in youth. Development of preventive strategies in earlier stages of life is crucial. We aimed to examine epidemiological evidence of early-life exposures and their associations with childhood and later risk of obesity and diabetes, and to discuss potential mechanisms.
Recent Findings
Parental obesity and diabetes in the preconception period may influence offspring’s obesity risk via epigenetic mechanisms influencing gametogenesis and early development that could have significant transgenerational effects. A more comprehensive understanding of these effects is needed to identify possible avenues for interventions in both fathers and mothers to be. In addition, current evidence suggests that growth and body weight trajectories in infancy and childhood are useful indicators of later obesity and type 2 diabetes. Moreover, the composition and variations in the microbiome in early life are associated with long-term health and could mediate associations between several early-life exposures and later risk of diseases.
Summary
Altogether, the epidemiological evidence supports the need for preconception and early-life interventions to reduce the obesity and diabetes burden in later life.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The prevalence of type 2 diabetes has nearly doubled in adults over the past 30 years [1]. It has also increased in children and parallels the increasing prevalence of obesity and overweight in youth [2,3,4]. Established risk factors for type 2 diabetes include ethnicity, family history of diabetes, previous gestational diabetes, older age, overweight and obesity, and unhealthful lifestyle behaviors including poor diet quality, insufficient physical activity, and smoking [1]. Over the past decades, several epidemiological studies have provided insights into the pathways by which environmental exposures in sensitive periods of early development predict risk for adult metabolic disorders, including diabetes [5], a concept referred to as the developmental origins of health and disease [6].
The objectives of this review are to examine epidemiological evidence of early-life exposures and their associations with childhood and later risk of obesity and diabetes, and to discuss potential mechanisms. We categorize early development into three exposure periods: preconception, gestation, and early childhood (Fig. 1). The gestation period and associations of fetal growth with later health outcomes have been more extensively studied, as summarized previously [7,8,9], and thus, this review will primarily focus on recent evidence in the associations of preconception paternal and maternal health with childhood obesity and on early childhood growth patterns as indicators of future child’s risk. We also discuss potential mechanisms for the associations of risk factors during preconception and early childhood with later obesity and diabetes, mainly through alternations in the microbiome and epigenetic regulation of gene expression.
Preconception Risk Factors
Abundant evidence has linked maternal obesity and diabetes during pregnancy with offspring obesity and diabetes risks in later life. However, in epidemiologic studies, it is challenging to distinguish the effects of maternal diabetes and obesity in the preconception compared to the gestational period, as these characteristics generally track over time. However, some studies have suggested that effects, and associated mechanisms, would differ for preconception vs. gestational exposure, resulting in a complex interaction between the effects of both periods [10]. Although most studies have focused on maternal-child associations and mechanisms in the developmental origins of child’s health, more recent studies have investigated the potential paternal metabolic contribution to his child’s later risk of disease.
Parental Diabetes in the Preconception Period
The heredity of diabetes has been well-established, with an additive effect on risk for diabetes in the offspring if both parents are affected [11]. For example, in the Framingham Offspring Study, maternal and paternal diabetes over the life course conveyed equivalent risk for type 2 diabetes in offspring in adulthood, suggesting transmission of genetic factors of similar strengths [12]. This study was unable to distinguish whether diabetes occurred prior or after pregnancy; yet, offspring of mothers with diabetes onset before 50 years of age had a markedly higher risk of diabetes and an earlier age at onset [12]. Other studies that looked at parental diabetes prior to conception specifically have showed associations with the risks of obesity, type 2 diabetes, and associated metabolic alterations in the offspring [13,14,15].
Given that many women may not have been screened for diabetes prior to conception, it is often difficult to distinguish pre-gestational type 2 diabetes first diagnosed during pregnancy from true gestational diabetes. Most of the evidence of an increased risk for obesity and type 2 diabetes in children from mothers who had developed diabetes prior to pregnancy come from either offspring of women with type 1 diabetes [14] or from the Pima Indians [13], a population at elevated risk for early-onset type 2 diabetes.
A few studies have suggested an increase in body mass index (BMI) z-score in offspring of women with type 1 diabetes prior to pregnancy as compared to children not exposed to diabetes in utero [14]. For example, Lindsay and colleagues reported no difference in glucose tolerance, but higher BMI, waist circumference, sum of skinfolds, and prevalence of overweight and obesity, in 7-year-old offspring from mothers with type 1 diabetes compared to offspring of non-diabetic mothers, a relationship that appeared to be related to higher fetal leptin concentrations [16]. An increased risk for type 2 diabetes was also reported in a small cohort (N = 75) of offspring from mothers with type 1 diabetes (predicted RR = 3.2) [17] and from a larger cohort (N = 597) including mothers with both gestational and type 1 diabetes (OR specifically in mothers with type 1 diabetes = 4.02, 95% CI [1.31, 12.33]) [18]. However, given that women with pre-gestational diabetes are usually hyperglycemic both prior to and during pregnancy, the sensitive exposure windows are difficult to disentangle.
The prevalence of type 2 diabetes in women of reproductive age (18–44 years old) remains fairly low in the USA (< 3%) [19]; yet it has been increasing over the past years especially among pregnant women [20], likely driven by increased obesity rates and older ages at pregnancy. Women with pre-gestational type 2 diabetes are more likely to present adverse maternal and fetal outcomes [21], but little is known about the child’s long-term health risk. In a sub-analysis of siblings from the Pima Indians of Arizona longitudinal study, mean BMI in adolescence (age 13) and diabetes risk in early adulthood (age 22–24) in offspring were found to be higher in siblings born after the mother’s diagnosis of type 2 diabetes compared to those born before [13, 22]. Considering that women of childbearing age do not typically undergo routine screening for type 2 diabetes, its presence in women prior to conception remains difficult to identify; however, the specific maternal and child risk associated with pre-gestational type 2 diabetes should be further investigated in diverse populations. Also, efficient implementation of the American Diabetes Associations’ recommendation to screen high-risk women for gestational diabetes in the first trimester [23] could contribute to identify cases of pre-gestational diabetes; however, we do not know the extent to which this recommendation is followed in clinical settings.
Little is known about the contributing role of paternal diabetes to children’s long-term risk of obesity and diabetes. Paternal type 2 diabetes or insulin resistance at conception has been linked to fetal growth restriction and lower birth weight [24, 25]. One study reported that lower birth weight predicted later development of diabetes in the offspring in the presence of paternal type 2 diabetes, but not maternal type 2 diabetes, suggesting that paternally derived genetic or epigenetic differences could be responsible for the association [15]. In the sub-analysis of siblings from the Pima Indians of Arizona longitudinal study, no association was found with timing of paternal diabetes diagnosis and offspring diabetes risk in early adulthood [13, 22]. A study in mice showed that offspring from fathers with pre-gestational diabetes could have increased susceptibility to diabetes through epigenetic alterations in gametes and modified expression of genes in the pancreatic islets, leading to altered glucose metabolism regulation and insulin signaling [26].
Parental Obesity in the Preconception Period
Parental obesity is also a strong predictor of childhood obesity and some studies showed greater effects of maternal BMI on childhood obesity risk as compared to paternal BMI; yet, risk for childhood obesity appears to be greatest when both parents are obese [27, 28]. In the Northern Finland Birth Cohort 1986, both maternal and paternal BMI were independently associated with overweight and obesity in childhood, and obesity persistence through adolescence [29]. In the US Project Viva cohort, compared to offspring of non-obese parents, maternal and paternal obesity, individually or together, were associated with an earlier age at adiposity rebound (11.4 months earlier for maternal obesity only, 6.5 months earlier for paternal obesity only, and 12.2 months earlier for obesity in both parents); in turn, such earlier adiposity rebound predicts later obesity [30]. Obesity of both parents was associated with the highest BMI into early adolescence (Fig. 2) [30].
Child BMI trajectories during infancy and childhood by parental obesity status in Project Viva (from [30] with permission from Elsevier)
Children of parents with obesity likely share not only genetic risks but also extra-uterine environmental and lifestyle-related exposures (shared household dietary habits and meal patterns, familial physical activities, similar home and neighborhood characteristics including access to grocery stores, green spaces, and exposure to pollution) that could explain some of the associations observed with parental preconception obesity and offspring’s obesity risk. Beyond these shared factors, preconception parental obesity could impact child’s obesity risk via epigenetic mechanisms influencing gametogenesis and early development [31,32,33,34]. For example, Soubry and colleagues found relationships of preconception parental obesity, specific to maternal or paternal obesity, with DNA methylation profiles (in DNA extracted from umbilical cord blood leukocytes at birth) of genes implicated in normal growth and development [31, 32]. Specifically, paternal obesity was associated with hypomethylation of the insulin-like growth factor 2 (IGF2) gene [31], suggesting a susceptibility to “reprogramming” of imprint marks during spermatogenesis, and both maternal and paternal obesity were associated with altered DNA methylation patterns at different imprinted genes [32].
In women, pre-conception obesity is also associated with increased risk of gestational diabetes [35] which could partly mediate the observed effects in offspring. Kral and colleagues compared the rates of obesity and overweight in siblings born before or after malabsorptive bariatric surgery [36]. Interestingly, male offspring whose mothers had their surgery before rather than after pregnancy, and thus entered pregnancy at lower BMI (mean ± SD pre-pregnancy BMI 48 ± 8 vs. 31 ± 9 kg/m2), had lower rates of obesity and overweight in childhood and adolescence [36]. The investigators did not evaluate whether these benefits were mediated by restricted gestational weight gain, improved glucose tolerance (before and/or during pregnancy), or epigenetic differences. Recent findings from a Swedish cohort study showed that pregnancies after a bariatric surgery were associated with reduced risks of gestational diabetes and large for gestational age infants, but also with increased risks of small for gestational age infants and possibly increased neonatal mortality [37].
Parental Nutrition in the Preconception Period
Recent studies in animal models have also shown epigenetic effects of paternal diet-induced obesity or nutritional inadequacies [38, 39••]. For example, in male rodents (mice and rats) fed a high-fat diet prior to conception, changes in the sperm epigenome were observed and were linked to offspring metabolism [40, 41]. Both maternal and paternal folate deficiency have been associated with offspring DNA methylation changes [42]. These dietary effects could likely persist through generations [38] and should be further investigated to better understand the paternal-specific role in the developmental origins of obesity and diabetes. Maternal nutrition prior to conception has also been shown to influence DNA methylation in offspring, resulting in lasting phenotypic effects that are not fully characterized [43]. In addition, effects of dietary methyl donors (before and during pregnancy) on DNA methylation changes in the offspring have been reported (e.g., associations of maternal folic acid in the periconceptional period have been associated with methylation of the IGF2 gene in offspring), as reviewed recently [44].
Preconception Environmental Exposures
A number of epidemiologic studies have reported associations of paternal exposures to various drugs and environmental toxicants with altered metabolism in offspring [38, 45, 46]. Recent reviews described how environmental chemical exposures in fathers can affect sperm quality and epigenetic profiles, affecting offspring’s metabolism and later risk of obesity and chronic diseases [38, 39••, 45]. In women, environmental exposures pre-pregnancy can have a variety of effects, depending of the type, timing, and magnitude of exposure, including altered fetal growth and gestational length, and functional and structural abnormalities [47]. Toxicant exposures in utero can also influence offspring’s later risk of chronic diseases, including diabetes [48].
In summary, both maternal and paternal pre-conception obesity and diabetes and nutritional and environmental exposures contribute to child’s later risk for disease. Paternal influences would be limited, in the pre-conception period, to effects through gametogenesis. However, the long-term and even transgenerational impact of these epigenetic alterations is biologically plausible, and fathers can also influence the postnatal family environment (e.g., dietary and physical activity patterns). Influences of maternal pre-gestational diabetes and obesity are likely to be more extensive, given the duration of exposure from pre-conception through gestation, and also during postnatal life. Epigenetic research in both animal and human studies will likely contribute to a better understanding of the associations between parental preconception risk factors and obesity and diabetes risk in offspring, and more research is needed in the field of transgenerational epigenetics, especially for a possible paternal role in the transmission of risk factors to offspring [45].
Gestational Risk Factors
Maternal Nutrition and Diabetes in Pregnancy
Adequate fetal growth and development are highly dependent on maternal nutritional status [49]. Several studies, mainly related to famine exposure, support associations between nutritional deprivation in utero and offspring risk of diabetes in adult life [50]. It remains unclear whether these associations are related to insufficient intake of total energy or of specific macro- or micronutrients. On the other hand, fetal overnutrition is also associated with later risk of obesity and metabolic disturbances in offspring, likely through different pathways [51, 52]. Common causes of fetal overnutrition include excessive gestational weight gain or maternal diabetes during pregnancy. Excessive pregnancy weight gain is highly prevalent: a 2017 meta-analysis including 23 observational studies and > 1 million women found that excessive gestational weight gain occurs in 47% of pregnant women in developed countries [53]. Women with weight gain above recommendations are at higher risk for having large for gestational age babies [53] and their children have a 1.4-fold higher risk of obesity into adolescence and adulthood [51]. Interestingly, maternal weight gain in early pregnancy seems to have a stronger association with later obesity compared with third trimester gain [54]; this observation suggests that intervention studies aiming to limit gestational weight gain should begin early in pregnancy if aiming to reduce the long-term risk in offspring.
Maternal hyperglycemia during pregnancy (with diabetes prior to pregnancy or first occurrence in pregnancy) contributes to fetal overnutrition, and observational studies suggest higher risk of obesity and diabetes in exposed offspring [14, 55,56,57,58,59], although studies often lack adjustment for important confounding factors, most notably maternal and paternal BMI [60]. Current evidence linking gestational diabetes to childhood obesity raises several questions as to whether hyperglycemia per se is a direct cause for childhood obesity, and randomized controlled trials with long-term follow-up as well as mechanistic studies remain needed.
Environmental Exposures
There is some evidence that environmental exposures during pregnancy influence fetal growth and later risk of obesity and metabolic disorders. One particularly well-characterized exposure is maternal smoking during pregnancy, which is associated with restricted fetal growth [61]. However, in later childhood, children of mothers who smoked during pregnancy have 1.5-fold greater risk for overweight and obesity as compared to children of mothers who did not smoke [61] and a dose-response relationship of maternal smoking and offspring overweight has been observed [62]. A number of environmental chemical exposures have been linked to obesity and metabolic perturbations, and earlier life exposure to these chemicals and contaminants could have a long-lasting effect [63,64,65]. Studied chemical exposures include, but are not limited to, smoking, arsenic, persistent organic pollutants (POPs), peroxisome proliferator-activated receptor (PPAR) activators (organotins and phthalates), bisphenol A (BPA), and pesticides [64]. Thus, many exposures could influence child’s risk of obesity and diabetes through fetal growth, although we do not fully understand all the drivers and their interactions.
Early Childhood Risk Factors
Growth and Body Weight Patterns
Several indicators of childhood growth have been associated with higher risks of insulin resistance and type 2 diabetes later in life [66, 67]. In infancy, rapid weight gain, defined as a change in weight z-score > 0.67 (corresponding to the width of percentile bands on growth charts), is associated with overweight and obesity through adulthood [68]. The odds ratio for overweight and obesity was 3.66 [95% CI 2.59–5.17] in the presence of rapid weight gain before 2 years of age based on a recent meta-analysis (2018; 17 studies), with higher odds when rapid weight gain occurred before 1 year of age [69]. Some studies have also suggested that rapid weight gain in the first year of life, and particularly in the first 3 months of life [70, 71], could be associated with decreased insulin sensitivity in adolescence and early adulthood although other studies have found either the opposite [72, 73] or null findings [74].
Growth trajectories in most children include a peak in BMI around 1 year of age, with a decrease through 4 to 6 years of age after which adiposity starts to increase again. The BMI nadir has been labeled the “adiposity rebound” [75], although BMI is not a direct measure of adiposity. Different childhood growth patterns have been associated with elevated risk of type 2 diabetes and obesity later in life. An infancy BMI peak at a later age and of greater magnitude was associated with adiposity in later childhood in two prospective birth cohorts [76, 77•]. In addition, an earlier adiposity rebound has been associated with higher risk of obesity [78, 79] and cardiometabolic disorders [80,81,82] in adolescence and adulthood. For example, in the Helsinki cohort, an earlier age at adiposity rebound was found to be associated with a higher BMI at age 12 years and an increased cumulative incidence of type 2 diabetes later in life [81].
Current evidence suggests that growth and body weight trajectories are useful indicators of risks for later obesity and related metabolic disorders. However, studies remain needed to identify determinants of early adiposity rebound and different growth patterns in childhood and to provide insight on mechanisms linking childhood growth patterns to cardiometabolic risk in adulthood. Differences in growth patterns are likely the result of numerous interactions between pre-conception and gestational exposures, and disentangling these effects will require detailed longitudinal assessments. In Project Viva, a pre-birth longitudinal cohort in the USA, maternal glucose tolerance status in pregnancy was associated with the timing and magnitude of BMI peak while higher maternal first trimester weight gain, smoking during pregnancy, no breastfeeding, parental obesity, and no university education were associated with higher BMI at rebound [30].
Early-Life Feeding Practices
The associations of early-life feeding practices (breastfeeding or not, and timing of introduction and type of complementary feeding) and obesity and diabetes risk have been investigated in numerous observational studies. Although some observational studies suggest a lower prevalence of overweight and obesity in children who were fed breast milk compared with infant formula [83], optimal duration and intensity of breastfeeding to provide substantial benefits remain unknown and several confounding factors such as parental feeding styles, socioeconomic status, and maternal health should be accounted for in future studies to fully support these associations [84]. Moreover, results from the Promotion of Breastfeeding Intervention Trial (PROBIT) study, a randomized controlled trial promoting increased duration and exclusivity of breastfeeding, showed that although the intervention succeeded in increasing the duration of exclusive breastfeeding, it did not lower risk of overweight and obesity [85] or influence cardiometabolic risk factors [86] in early adolescence. Timing of introduction and type/order of complementary food could also contribute to childhood obesity or metabolic risk through microbiome alterations and related epigenetic alterations, although the evidence in this research area is scarce and inconsistent [87,88,89].
Microbiome
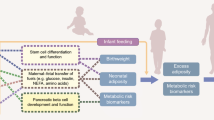
The potential role of the gut microbiome, either as an exposure or as a mechanism, in metabolic disorders such as type 2 diabetes has recently been identified and has been the subject of intense investigation [90]. Beyond the importance of the gut microbiota in the development of infant’s immune systems, there is now growing evidence for the role of the microbiome in postnatal programming of obesity and auto-immune and metabolic disorders [91, 92]. Early-life microbiome’s composition and variations are associated with long-term health and could mediate the associations observed between several early-life exposures and later risk of diseases [93••]. Evidence of perinatal factors affecting the microbiome in early life has accumulated, and relevant exposures include maternal-fetal transmission, birth mode, infant feeding, antibiotic use, and diet (Fig. 3) [92, 94].
Factors shaping the microbiome in early life (reprinted by permission from Springer Nature [92])
Results from animal studies and from human prospective studies suggest that gut microbiome variations in early life precede the development of obesity [93••]. Dogra and colleagues showed that gut microbiota composition at 6 months was associated with adiposity at 18 months of age [95]. Although limited, there is evidence suggesting that maternal obesity status in pregnancy and weight gain, as well as maternal diabetes, may be associated with offspring microbiome in infancy [91, 96]. However, these effects appear to differ according to child’s sex, ethnicity, and geographical location [91]. Mechanistic studies are needed to understand the contributing or mediating roles of the microbiome to the developmental origins of obesity and diabetes [92], including investigations of related epigenetic modifications in the host that are likely one of the key mechanisms to explain these associations [97].
Conclusions
A deeper understanding of early-life risk factors linked to risk of diabetes, and their associated mechanisms, could help tailor interventions for susceptible populations or vulnerable periods of development. Improving maternal and paternal metabolic health, including through diet, before conception could play a significant role in preventing later disease risk in offspring. Recent evidence has increased the awareness of paternal metabolic health in the preconception period, but additional studies remain needed. The preconception period presents a unique opportunity for intervention, which could possibly result in more effective interventions compared to those beginning in pregnancy only [98]. Nonetheless, healthy nutrition during pregnancy remains crucial, together with appropriate gestational weight gain and control of maternal hyperglycemia to favor adequate fetal growth. Finally, strategies are needed to prevent obesity and promote a healthy lifestyle among all sectors of the population. Recent studies have identified several windows of opportunity, from preconception to childhood, during which interventions could have long-lasting effects and that could halt the transgenerational cycle of obesity and type 2 diabetes.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
World Health Organization. Global report on diabetes. Geneva, Switzerland; 2016. http://www.who.int/diabetes/global-report/en/.
Mayer-Davis EJ, Lawrence JM, Dabelea D, Divers J, Isom S, Dolan L, et al. Incidence trends of type 1 and type 2 diabetes among youths, 2002–2012. N Engl J Med. 2017;376(15):1419–29. https://doi.org/10.1056/NEJMoa1610187.
Williams DE, Cadwell BL, Cheng YJ, Cowie CC, Gregg EW, Geiss LS, et al. Prevalence of impaired fasting glucose and its relationship with cardiovascular disease risk factors in US adolescents, 1999–2000. Pediatrics. 2005;116(5):1122–6. https://doi.org/10.1542/peds.2004-2001.
Fryar CD, Carroll MD, Ogden CL. Prevalence of overweight and obesity among children and adolescents aged 2–19 years: United States, 1963–1965 through 2013–2014. http://www.cdc.gov/nchs/data/hestat/obesity_child_13_14/obesity_child_13_14.htm. Published July 2016. Accessed May 2018.
Godfrey KM, Barker DJ. Fetal programming and adult health. Public Health Nutr. 2001;4(2B):611–24.
Gillman MW, Barker D, Bier D, Cagampang F, Challis J, Fall C, et al. Meeting report on the 3rd International Congress on Developmental Origins of Health and Disease (DOHaD). Pediatr Res. 2007;61(5 Pt 1):625–9. https://doi.org/10.1203/pdr.0b013e3180459fcd.
Gluckman PD, Hanson MA, Cooper C, Thornburg KL. Effect of in utero and early-life conditions on adult health and disease. N Engl J Med. 2008;359(1):61–73. https://doi.org/10.1056/NEJMra0708473.
Ozanne SE, Fernandez-Twinn D, Hales CN. Fetal growth and adult diseases. Semin Perinatol. 2004;28(1):81–7.
Oken E, Gillman MW. Fetal origins of obesity. Obes Res. 2003;11(4):496–506. https://doi.org/10.1038/oby.2003.69.
Freeman DJ. Effects of maternal obesity on fetal growth and body composition: implications for programming and future health. Semin Fetal Neonatal Med. 2010;15(2):113–8. https://doi.org/10.1016/j.siny.2009.09.001.
Stumvoll M, Goldstein BJ, van Haeften TW. Type 2 diabetes: principles of pathogenesis and therapy. Lancet. 2005;365(9467):1333–46. https://doi.org/10.1016/S0140-6736(05)61032-X.
Meigs JB, Cupples LA, Wilson PW. Parental transmission of type 2 diabetes: the Framingham Offspring Study. Diabetes. 2000;49(12):2201–7.
Dabelea D, Pettitt DJ. Intrauterine diabetic environment confers risks for type 2 diabetes mellitus and obesity in the offspring, in addition to genetic susceptibility. J Pediatr Endocrinol Metab. 2001;14(8):1085–91.
Philipps LH, Santhakumaran S, Gale C, Prior E, Logan KM, Hyde MJ, et al. The diabetic pregnancy and offspring BMI in childhood: a systematic review and meta-analysis. Diabetologia. 2011;54(8):1957–66. https://doi.org/10.1007/s00125-011-2180-y.
Lindsay RS, Dabelea D, Roumain J, Hanson RL, Bennett PH, Knowler WC. Type 2 diabetes and low birth weight: the role of paternal inheritance in the association of low birth weight and diabetes. Diabetes. 2000;49(3):445–9.
Lindsay RS, Nelson SM, Walker JD, Greene SA, Milne G, Sattar N, et al. Programming of adiposity in offspring of mothers with type 1 diabetes at age 7 years. Diabetes Care. 2010;33(5):1080–5. https://doi.org/10.2337/dc09-1766.
Weiss PA, Scholz HS, Haas J, Tamussino KF, Seissler J, Borkenstein MH. Long-term follow-up of infants of mothers with type 1 diabetes: evidence for hereditary and nonhereditary transmission of diabetes and precursors. Diabetes Care. 2000;23(7):905–11.
Clausen TD, Mathiesen ER, Hansen T, Pedersen O, Jensen DM, Lauenborg J, et al. High prevalence of type 2 diabetes and pre-diabetes in adult offspring of women with gestational diabetes mellitus or type 1 diabetes: the role of intrauterine hyperglycemia. Diabetes Care. 2008;31(2):340–6. https://doi.org/10.2337/dc07-1596.
Center for Disease Control and Prevention. National Diabetes Statistics Report, 2017. https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Page consulted 06–06-2018.
Peng TY, Ehrlich SF, Crites Y, Kitzmiller JL, Kuzniewicz MW, Hedderson MM, et al. Trends and racial and ethnic disparities in the prevalence of pregestational type 1 and type 2 diabetes in Northern California: 1996–2014. Am J Obstet Gynecol. 2017;216(2):177 e1–8. https://doi.org/10.1016/j.ajog.2016.10.007.
Feig DS, Palda VA. Type 2 diabetes in pregnancy: a growing concern. Lancet. 2002;359(9318):1690–2. https://doi.org/10.1016/S0140-6736(02)08599-9.
Dabelea D, Hanson RL, Lindsay RS, Pettitt DJ, Imperatore G, Gabir MM, et al. Intrauterine exposure to diabetes conveys risks for type 2 diabetes and obesity: a study of discordant sibships. Diabetes. 2000;49(12):2208–11.
American Diabetes A. 2. Classification and diagnosis of diabetes: standards of medical care in diabetes—2018. Diabetes Care. 2018;41(Suppl 1):S13–27. https://doi.org/10.2337/dc18-S002.
Hillman S, Peebles DM, Williams DJ. Paternal metabolic and cardiovascular risk factors for fetal growth restriction: a case-control study. Diabetes Care. 2013;36(6):1675–80. https://doi.org/10.2337/dc12-1280.
Moss JL, Harris KM. Impact of maternal and paternal preconception health on birth outcomes using prospective couples’ data in Add Health. Arch Gynecol Obstet. 2015;291(2):287–98. https://doi.org/10.1007/s00404-014-3521-0.
Wei Y, Yang CR, Wei YP, Zhao ZA, Hou Y, Schatten H, et al. Paternally induced transgenerational inheritance of susceptibility to diabetes in mammals. Proc Natl Acad Sci U S A. 2014;111(5):1873–8. https://doi.org/10.1073/pnas.1321195111.
Danielzik S, Langnase K, Mast M, Spethmann C, Muller MJ. Impact of parental BMI on the manifestation of overweight 5–7 year old children. Eur J Nutr. 2002;41(3):132–8. https://doi.org/10.1007/s00394-002-0367-1.
Whitaker KL, Jarvis MJ, Beeken RJ, Boniface D, Wardle J. Comparing maternal and paternal intergenerational transmission of obesity risk in a large population-based sample. Am J Clin Nutr. 2010;91(6):1560–7. https://doi.org/10.3945/ajcn.2009.28838.
Morandi A, Meyre D, Lobbens S, Kleinman K, Kaakinen M, Rifas-Shiman SL, et al. Estimation of newborn risk for child or adolescent obesity: lessons from longitudinal birth cohorts. PLoS One. 2012;7(11):e49919. https://doi.org/10.1371/journal.pone.0049919.
Aris IM, Rifas-Shiman S, Li LJ, Kleinman K, Coull B.A., Gold DR et al. Pre-, perinatal and parental predictors of body mass index trajectory milestones. J Pediatr. 2018;in press.
Soubry A, Schildkraut JM, Murtha A, Wang F, Huang Z, Bernal A, et al. Paternal obesity is associated with IGF2 hypomethylation in newborns: results from a Newborn Epigenetics Study (NEST) cohort. BMC Med. 2013;11:29. https://doi.org/10.1186/1741-7015-11-29.
Soubry A, Murphy SK, Wang F, Huang Z, Vidal AC, Fuemmeler BF, et al. Newborns of obese parents have altered DNA methylation patterns at imprinted genes. Int J Obes (Lond). 2015;39(4):650–7. https://doi.org/10.1038/ijo.2013.193.
Hur SS, Cropley JE, Suter CM. Paternal epigenetic programming: evolving metabolic disease risk. J Mol Endocrinol. 2017;58(3):R159–R68. https://doi.org/10.1530/JME-16-0236.
McPherson NO, Fullston T, Aitken RJ, Lane M. Paternal obesity, interventions, and mechanistic pathways to impaired health in offspring. Ann Nutr Metab. 2014;64(3–4):231–8. https://doi.org/10.1159/000365026.
Mighty HE, Fahey AJ. Obesity and pregnancy complications. Curr Diab Rep. 2007;7(4):289–94.
Kral JG, Biron S, Simard S, Hould FS, Lebel S, Marceau S, et al. Large maternal weight loss from obesity surgery prevents transmission of obesity to children who were followed for 2 to 18 years. Pediatrics. 2006;118(6):e1644–9. https://doi.org/10.1542/peds.2006-1379.
Johansson K, Cnattingius S, Naslund I, Roos N, Trolle Lagerros Y, Granath F, et al. Outcomes of pregnancy after bariatric surgery. N Engl J Med. 2015;372(9):814–24. https://doi.org/10.1056/NEJMoa1405789.
Soubry A. Epigenetic inheritance and evolution: a paternal perspective on dietary influences. Prog Biophys Mol Biol. 2015;118(1–2):79–85. https://doi.org/10.1016/j.pbiomolbio.2015.02.008.
•• Sales VM, Ferguson-Smith AC, Patti ME. Epigenetic mechanisms of transmission of metabolic disease across generations. Cell Metab. 2017, 25(3):559–71. https://doi.org/10.1016/j.cmet.2017.02.016. This review describes how epigenetic mechanisms could contribute to the intergenerational cycle of metabolic disease, with a focus on paternal lineage effects.
McPherson NO, Owens JA, Fullston T, Lane M. Preconception diet or exercise intervention in obese fathers normalizes sperm microRNA profile and metabolic syndrome in female offspring. Am J Physiol Endocrinol Metab. 2015;308(9):E805–21. https://doi.org/10.1152/ajpendo.00013.2015.
de Castro Barbosa T, Ingerslev LR, Alm PS, Versteyhe S, Massart J, Rasmussen M, et al. High-fat diet reprograms the epigenome of rat spermatozoa and transgenerationally affects metabolism of the offspring. Mol Metab. 2016;5(3):184–97. https://doi.org/10.1016/j.molmet.2015.12.002.
Mejos KK, Kim HW, Lim EM, Chang N. Effects of parental folate deficiency on the folate content, global DNA methylation, and expressions of FRalpha, IGF-2 and IGF-1R in the postnatal rat liver. Nutr Res Pract. 2013;7(4):281–6. https://doi.org/10.4162/nrp.2013.7.4.281.
Dominguez-Salas P, Moore SE, Baker MS, Bergen AW, Cox SE, Dyer RA, et al. Maternal nutrition at conception modulates DNA methylation of human metastable epialleles. Nat Commun. 2014;5:3746. https://doi.org/10.1038/ncomms4746.
Dominguez-Salas P, Cox SE, Prentice AM, Hennig BJ, Moore SE. Maternal nutritional status, C(1) metabolism and offspring DNA methylation: a review of current evidence in human subjects. Proc Nutr Soc. 2012;71(1):154–65. https://doi.org/10.1017/S0029665111003338.
Soubry A. POHaD: why we should study future fathers. Environ Epigenet. 2018;4(2):dvy007. https://doi.org/10.1093/eep/dvy007.
Skinner MK, Manikkam M, Guerrero-Bosagna C. Epigenetic transgenerational actions of endocrine disruptors. Reprod Toxicol. 2011;31(3):337–43. https://doi.org/10.1016/j.reprotox.2010.10.012.
Mattison DR. Environmental exposures and development. Curr Opin Pediatr. 2010;22(2):208–18. https://doi.org/10.1097/MOP.0b013e32833779bf.
Stillerman KP, Mattison DR, Giudice LC, Woodruff TJ. Environmental exposures and adverse pregnancy outcomes: a review of the science. Reprod Sci. 2008;15(7):631–50. https://doi.org/10.1177/1933719108322436.
Wu G, Bazer FW, Cudd TA, Meininger CJ, Spencer TE. Maternal nutrition and fetal development. J Nutr. 2004;134(9):2169–72. https://doi.org/10.1093/jn/134.9.2169.
Hoffman DJ, Reynolds RM, Hardy DB. Developmental origins of health and disease: current knowledge and potential mechanisms. Nutr Rev. 2017;75(12):951–70. https://doi.org/10.1093/nutrit/nux053.
Mamun AA, Mannan M, Doi SA. Gestational weight gain in relation to offspring obesity over the life course: a systematic review and bias-adjusted meta-analysis. Obes Rev. 2014;15(4):338–47. https://doi.org/10.1111/obr.12132.
Taylor PD, Poston L. Developmental programming of obesity in mammals. Exp Physiol. 2007;92(2):287–98. https://doi.org/10.1113/expphysiol.2005.032854.
Goldstein RF, Abell SK, Ranasinha S, Misso M, Boyle JA, Black MH, et al. Association of gestational weight gain with maternal and infant outcomes: a systematic review and meta-analysis. JAMA. 2017;317(21):2207–25. https://doi.org/10.1001/jama.2017.3635.
Hivert MF, Rifas-Shiman SL, Gillman MW, Oken E. Greater early and mid-pregnancy gestational weight gains are associated with excess adiposity in mid-childhood. Obesity (Silver Spring). 2016;24(7):1546–53. https://doi.org/10.1002/oby.21511.
American Diabetes A. 13. Management of diabetes in pregnancy: standards of medical care in diabetes—2018. Diabetes Care. 2018;41(Suppl 1):S137–S43. https://doi.org/10.2337/dc18-S013.
Regnault N, Gillman MW, Rifas-Shiman SL, Eggleston E, Oken E. Sex-specific associations of gestational glucose tolerance with childhood body composition. Diabetes Care. 2013;36(10):3045–53. https://doi.org/10.2337/dc13-0333.
Wright CS, Rifas-Shiman SL, Rich-Edwards JW, Taveras EM, Gillman MW, Oken E. Intrauterine exposure to gestational diabetes, child adiposity, and blood pressure. Am J Hypertens. 2009;22(2):215–20. https://doi.org/10.1038/ajh.2008.326.
Silverman BL, Rizzo TA, Cho NH, Metzger BE. Long-term effects of the intrauterine environment. The Northwestern University Diabetes in Pregnancy Center. Diabetes Care. 1998;21(Suppl 2):B142–9.
Silverman BL, Metzger BE, Cho NH, Loeb CA. Impaired glucose tolerance in adolescent offspring of diabetic mothers. Relationship to fetal hyperinsulinism. Diabetes Care. 1995;18(5):611–7.
Kawasaki M, Arata N, Miyazaki C, Mori R, Kikuchi T, Ogawa Y, et al. Obesity and abnormal glucose tolerance in offspring of diabetic mothers: a systematic review and meta-analysis. PLoS One. 2018;13(1):e0190676. https://doi.org/10.1371/journal.pone.0190676.
Oken E, Levitan EB, Gillman MW. Maternal smoking during pregnancy and child overweight: systematic review and meta-analysis. Int J Obes (Lond). 2008;32(2):201–10. https://doi.org/10.1038/sj.ijo.0803760.
Albers L, Sobotzki C, Kuss O, Ajslev T, Batista RF, Bettiol H, et al. Maternal smoking during pregnancy and offspring overweight: is there a dose-response relationship? An individual patient data meta-analysis. Int J Obes (Lond). 2018; https://doi.org/10.1038/s41366-018-0050-0.
De Long NE, Holloway AC. Early-life chemical exposures and risk of metabolic syndrome. Diabetes Metab Syndr Obes. 2017;10:101–9. https://doi.org/10.2147/DMSO.S95296.
Thayer KA, Heindel JJ, Bucher JR, Gallo MA. Role of environmental chemicals in diabetes and obesity: a National Toxicology Program workshop review. Environ Health Perspect. 2012;120(6):779–89. https://doi.org/10.1289/ehp.1104597.
Braun JM. Early-life exposure to EDCs: role in childhood obesity and neurodevelopment. Nat Rev Endocrinol. 2017;13(3):161–73. https://doi.org/10.1038/nrendo.2016.186.
Barker DJ. The developmental origins of insulin resistance. Horm Res. 2005;64(Suppl 3):2–7. https://doi.org/10.1159/000089311.
Eriksson JG, Kajantie E, Lampl M, Osmond C. Trajectories of body mass index amongst children who develop type 2 diabetes as adults. J Intern Med. 2015;278(2):219–26. https://doi.org/10.1111/joim.12354.
Gillman MW. Early infancy as a critical period for development of obesity and related conditions. Nestle Nutr Workshop Ser Pediatr Program. 2010;65:13–20; discussion −4. https://doi.org/10.1159/000281141.
Zheng M, Lamb KE, Grimes C, Laws R, Bolton K, Ong KK, et al. Rapid weight gain during infancy and subsequent adiposity: a systematic review and meta-analysis of evidence. Obes Rev. 2018;19(3):321–32. https://doi.org/10.1111/obr.12632.
Leunissen RW, Kerkhof GF, Stijnen T, Hokken-Koelega A. Timing and tempo of first-year rapid growth in relation to cardiovascular and metabolic risk profile in early adulthood. JAMA. 2009;301(21):2234–42. https://doi.org/10.1001/jama.2009.761.
Fabricius-Bjerre S, Jensen RB, Faerch K, Larsen T, Molgaard C, Michaelsen KF, et al. Impact of birth weight and early infant weight gain on insulin resistance and associated cardiovascular risk factors in adolescence. PLoS One. 2011;6(6):e20595. https://doi.org/10.1371/journal.pone.0020595.
Eriksson JG, Forsen TJ, Osmond C, Barker DJ. Pathways of infant and childhood growth that lead to type 2 diabetes. Diabetes Care. 2003;26(11):3006–10.
Eriksson JG, Osmond C, Kajantie E, Forsen TJ, Barker DJ. Patterns of growth among children who later develop type 2 diabetes or its risk factors. Diabetologia. 2006;49(12):2853–8. https://doi.org/10.1007/s00125-006-0459-1.
Larnkjaer A, Schack-Nielsen L, Molgaard C, Ingstrup HK, Holst JJ, Michaelsen KF. Effect of growth in infancy on body composition, insulin resistance, and concentration of appetite hormones in adolescence. Am J Clin Nutr. 2010;91(6):1675–83. https://doi.org/10.3945/ajcn.2009.27956.
Wen X, Kleinman K, Gillman MW, Rifas-Shiman SL, Taveras EM. Childhood body mass index trajectories: modeling, characterizing, pairwise correlations and socio-demographic predictors of trajectory characteristics. BMC Med Res Methodol. 2012;12:38. https://doi.org/10.1186/471-2288-12-38.
Aris IM, Bernard JY, Chen LW, Tint MT, Pang WW, Lim WY, et al. Infant body mass index peak and early childhood cardio-metabolic risk markers in a multi-ethnic Asian birth cohort. Int J Epidemiol. 2017;46(2):513–25. https://doi.org/10.1093/ije/dyw232.
• Marinkovic T, Toemen L, Kruithof CJ, Reiss I, van Osch-Gevers L, Hofman A, et al. Early infant growth velocity patterns and cardiovascular and metabolic outcomes in childhood. J Pediatr. 2017;186:57–63.e4. https://doi.org/10.1016/j.jpeds.2017.02.004. This study examined the associations of detailed longitudinal infant weight velocity patterns with childhood cardiovascular and metabolic outcomes.
Hughes AR, Sherriff A, Ness AR, Reilly JJ. Timing of adiposity rebound and adiposity in adolescence. Pediatrics. 2014;134(5):e1354–61. https://doi.org/10.1542/peds.2014-1908.
Rolland-Cachera MF, Deheeger M, Maillot M, Bellisle F. Early adiposity rebound: causes and consequences for obesity in children and adults. Int J Obes (Lond). 2006;30(Suppl 4):S11–7.
Mo-Suwan L, McNeil E, Sangsupawanich P, Chittchang U, Choprapawon C. Adiposity rebound from three to six years of age was associated with a higher insulin resistance risk at eight-and-a-half years in a birth cohort study. Acta Paediatr. 2017;106(1):128–34. https://doi.org/10.1111/apa.13639.
Eriksson JG, Forsen T, Tuomilehto J, Osmond C, Barker DJ. Early adiposity rebound in childhood and risk of type 2 diabetes in adult life. Diabetologia. 2003;46(2):190–4. https://doi.org/10.1007/s00125-002-1012-5.
Koyama S, Ichikawa G, Kojima M, Shimura N, Sairenchi T, Arisaka O. Adiposity rebound and the development of metabolic syndrome. Pediatrics. 2014;133(1):e114–9. https://doi.org/10.1542/peds.2013-0966.
Woo JG, Martin LJ. Does breastfeeding protect against childhood obesity? Moving beyond observational evidence. Curr Obes Rep. 2015;4(2):207–16. https://doi.org/10.1007/s13679-015-0148-9.
Oken E, Fields DA, Lovelady CA, Redman LM. TOS scientific position statement: breastfeeding and obesity. Obesity (Silver Spring). 2017;25(11):1864–6. https://doi.org/10.1002/oby.22024.
Martin RM, Patel R, Kramer MS, Guthrie L, Vilchuck K, Bogdanovich N, et al. Effects of promoting longer-term and exclusive breastfeeding on adiposity and insulin-like growth factor-I at age 11.5 years: a randomized trial. JAMA. 2013;309(10):1005–13. https://doi.org/10.1001/jama.2013.167.
Martin RM, Patel R, Kramer MS, Vilchuck K, Bogdanovich N, Sergeichick N, et al. Effects of promoting longer-term and exclusive breastfeeding on cardiometabolic risk factors at age 11.5 years: a cluster-randomized, controlled trial. Circulation. 2014;129(3):321–9. https://doi.org/10.1161/CIRCULATIONAHA.113.005160.
Pearce J, Taylor MA, Langley-Evans SC. Timing of the introduction of complementary feeding and risk of childhood obesity: a systematic review. Int J Obes (Lond). 2013;37(10):1295–306. https://doi.org/10.1038/ijo.2013.99.
Vail B, Prentice P, Dunger DB, Hughes IA, Acerini CL, Ong KK. Age at weaning and infant growth: primary analysis and systematic review. J Pediatr. 2015;167(2):317–24 e1. https://doi.org/10.1016/j.jpeds.2015.05.003.
Seach KA, Dharmage SC, Lowe AJ, Dixon JB. Delayed introduction of solid feeding reduces child overweight and obesity at 10 years. Int J Obes (Lond). 2010;34(10):1475–9. https://doi.org/10.1038/ijo.2010.101.
Hartstra AV, Bouter KE, Backhed F, Nieuwdorp M. Insights into the role of the microbiome in obesity and type 2 diabetes. Diabetes Care. 2015;38(1):159–65. https://doi.org/10.2337/dc14-0769.
Kozyrskyj AL, Kalu R, Koleva PT, Bridgman SL. Fetal programming of overweight through the microbiome: boys are disproportionately affected. J Dev Orig Health Dis. 2016;7(1):25–34. https://doi.org/10.1017/S2040174415001269.
Tamburini S, Shen N, Wu HC, Clemente JC. The microbiome in early life: implications for health outcomes. Nat Med. 2016;22(7):713–22. https://doi.org/10.1038/nm.4142.
•• Stiemsma LT, Michels KB. The role of the microbiome in the developmental origins of health and disease. Pediatrics. 2018;141(4). https://doi.org/10.1542/peds.2017-2437. This review presents the latest evidence suggesting that the microbiome would be a mechanistic mediator in the developmental origins of health and disease.
Mueller NT, Bakacs E, Combellick J, Grigoryan Z, Dominguez-Bello MG. The infant microbiome development: mom matters. Trends Mol Med. 2015;21(2):109–17. https://doi.org/10.1016/j.molmed.2014.12.002.
Dogra S, Sakwinska O, Soh SE, Ngom-Bru C, Bruck WM, Berger B et al. Dynamics of infant gut microbiota are influenced by delivery mode and gestational duration and are associated with subsequent adiposity. MBio. 2015;6(1). https://doi.org/10.1128/mBio.02419-14.
Singh S, Karagas MR, Mueller NT. Charting the maternal and infant microbiome: what is the role of diabetes and obesity in pregnancy? Curr Diab Rep. 2017;17(2):11. https://doi.org/10.1007/s11892-017-0836-9.
Indrio F, Martini S, Francavilla R, Corvaglia L, Cristofori F, Mastrolia SA, et al. Epigenetic matters: the link between early nutrition, microbiome, and long-term health development. Front Pediatr. 2017;5:178. https://doi.org/10.3389/fped.2017.00178.
Stephenson J, Heslehurst N, Hall J, Schoenaker D, Hutchinson J, Cade JE, et al. Before the beginning: nutrition and lifestyle in the preconception period and its importance for future health. Lancet. 2018;391(10132):1830–41. https://doi.org/10.1016/S0140-6736(18)30311-8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Véronique Gingras, Marie-France Hivert, and Emily Oken declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Pathogenesis of Type 2 Diabetes and Insulin Resistance
Rights and permissions
About this article
Cite this article
Gingras, V., Hivert, MF. & Oken, E. Early-Life Exposures and Risk of Diabetes Mellitus and Obesity. Curr Diab Rep 18, 89 (2018). https://doi.org/10.1007/s11892-018-1050-0
Published:
DOI: https://doi.org/10.1007/s11892-018-1050-0