Abstract
Purpose of Review
Carcinoid heart disease is a rare disorder that is associated with significant morbidity and mortality. In this review of the literature, we will present current concepts in diagnosis and management of carcinoid heart disease.
Recent Findings
Recent expert consensus guidelines highlight the role of echocardiography and screening with NT-proBNP for the evaluation of carcinoid heart disease. Advances in medical therapy along with better surgical outcomes highlight the experience and expertise that has been gained in the treatment of carcinoid heart disease.
Summary
Carcinoid heart disease occurs in patients with neuroendocrine tumors who have carcinoid syndrome. Serotonin appears to play a central role in the development of carcinoid heart disease. Cardiac biomarkers and multimodality imaging can be used to aid in screening and diagnosis. The mainstay of treatment of carcinoid heart disease is surgery.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Carcinoid heart disease is a rare disease that occurs most commonly in patients with neuroendocrine tumors (NETs) that have metastasized to produce carcinoid syndrome. Carcinoid heart disease is the initial presentation of carcinoid syndrome in 20% of patients and occurs in up to half of patients with carcinoid syndrome [1]. Patients with carcinoid syndrome have clinical symptoms of diarrhea, hypotension, and bronchospasm, and in cases of cardiac involvement have signs and symptoms of congestive heart failure. Serotonin appears to play a central role in the development of valvular carcinoid heart disease, and medical therapy has been used to target the serotonin biochemical pathway in an attempt to control symptoms in patients with carcinoid syndrome. While carcinoid heart disease is rare, it is associated with significant morbidity and mortality. In this comprehensive review of the literature, we will present current concepts in the diagnosis and treatment of carcinoid heart disease.
Etiology and Pathophysiology
The incidence of NETs is estimated to be 6.98 per 100,000 in the general population and has increased 6.4-fold between 1973 and 2012 [2]. The highest incidence rates were found to be in gastroenteropancreatic sites (3.56 per 100,000), lung (1.49 per 100,000), and from unknown primary sites (0.84 per 100,000). Survival varies by stage, grade, age at diagnosis, primary site, and decade of diagnosis and has been improving over time, likely due to improvements in therapy [2].
Disseminated NETs can lead to carcinoid syndrome and can cause cardiac symptoms due to the release of vasoactive substances such as histamine, bradykinin, serotonin, and prostaglandins [3]. In patients with carcinoid syndrome, more than 70% of tryptophan is converted to serotonin as compared with 1% in normal situations [4]. Serotonin (5-HT) is circulated in the blood stream and is thought to play a crucial role in the development of carcinoid heart disease. Along with alterations in the metabolism of tryptophan that leads to increased production of serotonin, the activation of 5-hydroxytryptamine (5-HT) 2B receptors (located on heart valves) and interference with 5-HT transporters (which inactivates 5-HT in the lungs) are thought to lead to serotonin-mediated heart disease [5,6,7,8]. Serotonin is eventually metabolized into 5-hydroxyindoleacetic acid (5-HIAA).
Carcinoid heart disease is characterized by the plaque-like deposit of fibrous tissue which is composed of a mixture of myofibroblasts, smooth muscle cells, extracellular matrix, and an overlying endothelial layer. This primarily affects the heart valves, but can also involve other structures of the heart such as the cardiac chambers, the vena cava, pulmonary artery, and the coronary sinus [9].
Clinical Manifestations
Cardiac manifestations of carcinoid heart disease are usually associated with other common symptoms of the carcinoid syndrome like diarrhea, flushing, and bronchospasm [10,11,12,13]. Typically, patients are hypotensive, but others may have labile blood pressure or even be hypertensive [13]. In most cases, the right-sided valves of the heart are involved. In a case series of 74 patients, the majority (> 90%) had tricuspid valve disease showing moderate-to-severe tricuspid regurgitation, with > 80% also showing pulmonic valve involvement [1]. The predominant lesion is regurgitation but it may also cause stenosis [1]. Rarely, left-sided valvular involvement may precede the development of overt right-sided valvular disease [11]. Left-sided valve involvement is usually associated with the presence of a patent foramen ovale or atrial septal defect.
In the early phase of carcinoid heart disease, patients may be asymptomatic. Later, in the course of the disease, patients may develop fatigue and dyspnea on exertion. As the disease progresses, right-sided heart failure ensues with worsening dyspnea, edema, and ascites. Major findings on physical examination include edema and elevated jugular venous pressure. In cases of severe tricuspid regurgitation, the jugular venous pulse may show a prominent “V” wave. A palpable right ventricular impulse may be present, and murmurs of tricuspid and pulmonic valve regurgitation may be audible. In cases of pulmonic stenosis, a systolic murmur is present in the pulmonic area.
The auscultatory findings may be subtle, as the murmurs of tricuspid and pulmonary valve regurgitation are sometimes difficult to detect due to the low pressure in the right heart chambers. These right-sided murmurs are accentuated by inspiration. In the rare instance of left sided disease, murmurs of mitral and aortic disease may be present. In the presence of a significant right to left shunt, patients may have central cyanosis, in addition to other signs of heart failure [14]. In one of the earliest reports on this condition, Biorck K. et al. [15] reported a case of a patient with cyanosis in association with pulmonary stenosis and tricuspid insufficiency, in whom the postmortem analysis showed that in addition to the cardiac valvular lesion, there was a malignant carcinoid tumor of the jejunum with metastases to the liver.
Although valvular involvement appears to be the dominant clinical presentation of carcinoid heart disease, other atypical presentations include coronary artery vasospasm with ST segment elevation [16, 17], atrial fibrillation [18], ventricular tachycardia, ventricular fibrillation, and cardiac arrest [19,20,21]. Carcinoid heart disease has also been reported to provoke coronary artery vasospasm, angina, and in-stent thrombosis in patients with established ischemic heart disease [22]. Infrequently, carcinoid heart disease may present as an isolated intra-cardiac mass, without any valvular involvement [23, 24]. Rarely, patients may present with heart failure due to pericardial disease and constrictive pericarditis, in the absence of significant valvular disease [10, 25].
Diagnosis
Biomarkers
The measurement of serum biomarkers may be useful to identify carcinoid heart disease. Both elevated 5-hydroxyindoleacetic acid (5-HIAA) levels and elevated N-terminal pro b-type natriuretic peptide (NT-proBNP) levels are sensitive and specific predictors of carcinoid heart disease [26]. The European Neuroendocrine Tumor Society (ENETS) recommends screening patients with elevated 5-HIAA levels for carcinoid heart disease using transthoracic echocardiography [27]. Furthermore, one study suggested elevated NT-proBNP levels may be useful to select patients who should undergo diagnostic echocardiogram, allowing for more judicious use of this imaging modality [28]. NT-proBNP levels also correlate well with many of the echocardiographic scoring systems for severity of carcinoid heart disease [26].
Imaging
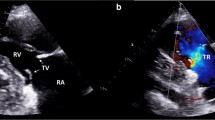
Two-dimensional transthoracic echocardiography (2D TTE) is often the diagnostic imaging test of choice for patients suspected of having carcinoid heart disease. Echocardiography is recommended in all patients with carcinoid syndrome and a high suspicion of carcinoid heart disease, based on a recent expert statement by the American College of Cardiology (ACC) [29••]. The ACC expert consensus statement recommends that for those with established mild valvular disease, a repeat echocardiogram and clinical assessment should occur every 6 months [30]. While for those with established moderate-to-severe valvular disease, a repeat echocardiogram and clinical assessment should occur every 3 months [29••], whereas the ENETS guideline recommends repeating the TTE annually among this group [27]. The TTE often demonstrates fibrous deposition along the pulmonic and tricuspid valves, which are noted to move in a “board-like” pattern [30]. This leads to significant tricuspid regurgitation with a characteristic Doppler pattern (Fig. 1).
Transthoracic echocardiogram in a modified RV inflow view showing thickened, fixed, and non-coapting tricuspid valve leaflets (arrowheads) (a), color flow Doppler imaging of severe tricuspid valve regurgitation (arrow) (b), and the characteristic “dagger-”shaped Doppler pattern (arrow) with an early peak pressure and rapid decline (c)
Three-dimensional transthoracic echocardiography (3D TTE) allows for visualization of all valve leaflets at the same time, which makes it a more sensitive test for diagnosing carcinoid heart disease, as not all valve leaflets are always involved [30, 31]. Transesophageal echocardiography (TEE) may be particularly useful in visualizing the pulmonic valve, which is often poorly visualized on TTE [32].
Cardiac magnetic resonance imaging (CMR) may be useful in identifying carcinoid heart disease, especially when echocardiographic findings are equivocal or there are technical difficulties in assessing the tricuspid valve by 2D echocardiography. Late gadolinium enhancement may allow for the identification of fibrotic changes in the myocardium that may not be apparent on echocardiogram [33]. Assessing the pulmonic valve with TTE might be difficult in adults, for which CMR provides great anatomical visualization and ability to provide an accurate and reproducible quantification of pulmonic regurgitation, regurgitant volume, and regurgitant fraction [34]. Cardiac computerized tomography (CCT) may also be used when echocardiogram is equivocal. Both the CMR and CCT share many advantages over echocardiogram, including improved visualization of valves, ability to identify rare cardiac metastasis, and quantification of chamber sizes [30, 35]. The CCT is a robust modality for structural intervention planning in the tricuspid valve and for appropriate prosthetic valve sizing [36]. These findings make the CMR and CCT particularly useful for surgical selection and planning.
Scoring Systems
Various scoring systems incorporate echocardiographic findings to either diagnose or track the progression of carcinoid heart disease (Table 1). The first echocardiographic scoring system takes into consideration tricuspid valve anatomy, tricuspid regurgitation severity, pulmonic stenosis severity, and pulmonic regurgitation severity [39]. One study recently determined that these tests are quite similar in sensitivity and specificity, but the more complex echocardiographic scoring systems may be particularly useful in tracking disease progression [37].
Invasive Hemodynamic Testing
Cardiac catheterization offers a direct means of analyzing the degree of valve dysfunction via invasive hemodynamic pressurement. A recent report included 13 patients with a known diagnosis of carcinoid heart disease who underwent cardiac catheterization revealed that tricuspid valve regurgitation was the most prevalent pathology (92%), followed by tricuspid stenosis (38%) and mitral regurgitation (38%). Left-sided lesions were less common and less severe than their right-sided counterparts. In one study, right-sided filling pressures tended to be mild to moderately elevated with normal left-sided pressure (no patient had mean pulmonary capillary wedge pressure > 20 mmHg) [38]. These findings were consistent with the greater severity of right-sided valvular pathology (tricuspid and pulmonary) compared with left-sided disease (mitral and aortic). Overall, there was a 91% concordance between cardiac catheterization and echocardiography for the assessment of valvular lesions [38]. These findings are consistent with a prior case series of 7 carcinoid heart disease patients by Himelman et al. which demonstrated hemodynamic findings of an elevated mean right atrial pressure of 15 mmHg, with a V wave of 22 mmHg and a right ventricular pressure of 39/10 mmHg [42].
Treatment
The treatment of carcinoid valvular heart disease is focused on managing symptoms, delaying progression of valve disease, and optimal timing of surgical intervention for improved quality of life and reducing mortality. This requires early and close collaboration between the medical oncologist, cardiologist, and cardiac surgeon. This collaboration and close monitoring is essential, as there may be rapid progression of carcinoid heart disease, and surgical intervention should be considered early before a disease progresses to a point at which surgery would be too high risk [29••].
The management of right-sided heart failure from carcinoid valvular disease primarily consists of careful volume management. Patients should be aware of the symptoms and signs of heart failure such as lower extremity edema and dyspnea so that they may seek medical attention immediately at the onset of symptoms. Most commonly, loop diuretics are used with the addition of aldosterone antagonists or thiazide diuretics, if additional diuresis is needed. In patients having valvular carcinoid disease with or without right ventricular dilation and failure, caution should be taken to avoid intravascular depletion [43]. This will result in decreased cardiac output and symptoms of lightheadedness, syncope, fatigue, and dyspnea with possible hypotension. In selected cases, traditional heart failure medications like angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, and beta-blockers can be considered, but have not been studied or shown to have proven benefit in this specific patient population. All of these cardiovascular medications are for palliation of symptoms and do not address the underlying carcinoid syndrome or carcinoid heart disease with valvular degeneration.
The pharmacological management of valvular disease is largely dependent on control of 5-HIAA levels through the use of somatostatin analogues [44, 45]. For patients refractory to somatostatin analogues, other options include everolimus [46], interferon alpha [47], peptide receptor radionuclide therapy [48], and telotristat etiprate [49]. Unfortunately, control of carcinoid syndrome has not shown to reverse the already established carcinoid valvular disease, but may play a role in slowing its progression [1, 37].
Surgical intervention is the only established effective treatment for carcinoid valvular heart disease. The optimal timing for surgical intervention is not clear, but evidence favors intervening prior to the development of right ventricular dilation and depressed right ventricular systolic function [50]. The current ACC consensus statement recommendations include consideration of valve surgery when symptoms are present and severe valvular disease is established [29••]. Typically, the tricuspid valve is involved, but careful attention should be placed on assessment of the pulmonic valve as often it has significant involvement and requires replacement [51]. When both the tricuspid valve and pulmonic valve are replaced, there appears to be a beneficial effect on right ventricular size and possibly right ventricular remodeling [52]. Consideration must be taken prior to surgery to control the systemic carcinoid syndrome, as uncontrolled carcinoid syndrome can result in bioprosthetic valve dysfunction [53].
The decision to pursue bioprosthetic versus mechanical valve replacement usually favors bioprosthetic valve replacement for right-sided valve pathology. Current literature supports the use of bioprosthetic valves as opposed to mechanical for several reasons [29••, 54]. One of the main considerations is long-term anticoagulation which can be problematic in carcinoid syndrome patients who often have concomitant liver and kidney disease and often need further procedures or non-cardiac surgeries. Also, mechanical valves in the tricuspid position are more prone to thrombosis and newer bioprosthetic valves have improved long-term durability. If patients have a favorable survival from carcinoid disease and bioprosthetic surgical valve deterioration occurs, transcatheter valve-in-valve replacement can be performed. In addition to valve replacement, the ACC expert consensus statement recommends the closure of patent foramen ovale at the time of initial surgery to reduce future left-sided valve involvement [29••].
To prevent perioperative carcinoid crisis, it is important to involve the medical oncologist and anesthesiologist in the preoperative surgical planning and in the perioperative management. Carcinoid crisis develops from the release of biologically active substances resulting in wide fluctuations in hemodynamics at times requiring vasopressor support. Generally, octreotide is started preoperatively and continued until the patient is stable postoperatively [59]. A multidisciplinary team including anesthesiologists, oncologists, surgeons, and cardiologists familiar with carcinoid syndrome is recommended for perioperative management.
Prognosis
The prognosis of valvular carcinoid heart disease without treatment is dismal with a median survival of only 11 months in patients with advanced heart failure [50]. The presence of valvular disease on echocardiography has been shown to be associated with worse survival when compared with those without valvular disease [1]. Once the development of valvular disease is established, the progression of valve disease also portends a worse prognosis [55, 56]. Patients with normal natriuretic peptide levels have a better prognosis even in the presence of established valve disease [58]. Moller et al. reported improved survival over the past two decades and largely attributed this to increased rates of cardiac surgery for these patients [56]. As stated above, the only established effective treatment is valve replacement. Early studies of valve surgery resulted in 30-day perioperative mortality as high as 63% [60], but with improved experience and better surgical techniques, the 30-day perioperative mortality has recently been reported as low as 3.7% [57•].
Conclusion
Carcinoid heart disease is a major cause of morbidity and mortality in patients with NETs. Serotonin appears to play a central role in the development of carcinoid valvular heart disease. Patients with carcinoid heart disease present with heart failure and both biomarkers and multimodality imaging are used for screening and diagnosis. The mainstay of treatment is valve surgery, but still carries substantial risks, and the timing of surgery remains unclear. A multidisciplinary team involving oncologists, cardiologists, cardiac surgeons, and anesthesiologist is essential for the optimal management of patients with carcinoid heart disease.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Pellikka PA, Tajik AJ, Khandheria BK, Seward JB, Callahan JA, Pitot HC, et al. Carcinoid heart disease. Clinical and echocardiographic spectrum in 74 patients. Circulation. 1993;87(4):1188–96.
Dasari A, Shen C, Halperin D, Zhao B, Zhou S, Xu Y, et al. Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the United States. JAMA Oncol. 2017;3(10):1335–42. https://doi.org/10.1001/jamaoncol.2017.0589.
Lundin L, Norheim I, Landelius J, Oberg K, Theodorsson-Norheim E. Carcinoid heart disease: relationship of circulating vasoactive substances to ultrasound-detectable cardiac abnormalities. Circulation. 1988;77(2):264–9.
Kvols LK. Metastatic carcinoid tumors and the malignant carcinoid syndrome. Ann N Y Acad Sci. 1994;733:464–70.
Castillo JG, Silvay G, Solis J. Current concepts in diagnosis and perioperative management of carcinoid heart disease. Semin Cardiothorac Vasc Anesth. 2013;17(3):212–23. https://doi.org/10.1177/1089253212465475.
Etienne N, Schaerlinger B, Jaffre F, Maroteaux L. The 5-HT2B receptor: a main cardio-pulmonary target of serotonin. J Soc Biol. 2004;198(1):22–9.
Hutcheson JD, Setola V, Roth BL, Merryman WD. Serotonin receptors and heart valve disease--it was meant 2B. Pharmacol Ther. 2011;132(2):146–57. https://doi.org/10.1016/j.pharmthera.2011.03.008.
Mekontso-Dessap A, Brouri F, Pascal O, Lechat P, Hanoun N, Lanfumey L, et al. Deficiency of the 5-hydroxytryptamine transporter gene leads to cardiac fibrosis and valvulopathy in mice. Circulation. 2006;113(1):81–9. https://doi.org/10.1161/circulationaha.105.554667.
Modlin IM, Sandor A. An analysis of 8305 cases of carcinoid tumors. Cancer. 1997;79(4):813–29.
Johnston SD, Johnston PW, O’Rourke D. Carcinoid constrictive pericarditis. Heart. 1999;82(5):641–3. https://doi.org/10.1136/hrt.82.5.641.
Marupakula V, Vinales KL, Najib MQ, Lanza LA, Lee HR, Chaliki HP. Occurrence of left-sided heart valve involvement before right-sided heart valve involvement in carcinoid heart disease. Eur J Echocardiogr. 2011;12(3):E18. https://doi.org/10.1093/ejechocard/jeq171.
Pandya UH, Pellikka PA, Enriquez-Sarano M, Edwards WD, Schaff HV, Connolly HM. Metastatic carcinoid tumor to the heart: echocardiographic-pathologic study of 11 patients. J Am Coll Cardiol. 2002;40(7):1328–32.
Tomaszewski A, Matuszek B, Zapolski T, Czekajska-Chehab E, Matuszek M, Rudzki S. Carcinoid heart disease as a first manifestation of midgut carcinoid tumor in a young woman. 2009.
Blick DR, Zoghbi WA, Lawrie GM, Verani MS. Carcinoid heart disease presenting as right-to-left shunt and congestive heart failure: successful surgical treatment. Am Heart J. 1988;115(1 Pt 1):201–3.
Biorck G, Axen O, Thorson A. Unusual cyanosis in a boy with congenital pulmonary stenosis and tricuspid insufficiency. Fatal outcome after angiocardiography. Am Heart J. 1952;44(1):143–8.
Eapen DJ, Clements S Jr, Block P, Sperling L. Metastatic carcinoid disease inducing coronary vasospasm. Tex Heart Inst J. 2012;39(1):76–8.
Petersen KG, Seemann WR, Plagwitz R, Kerp L. Evidence for coronary spasm during flushing in the carcinoid syndrome. Clin Cardiol. 1984;7(8):445–8. https://doi.org/10.1002/clc.4960070805.
Langer C, Piper C, Vogt J, Heintze J, Butz T, Lindner O, et al. Atrial fibrillation in carcinoid heart disease: the role of serotonin. A review of the literature. Clin Res Cardiol. 2007;96(2):114–8. https://doi.org/10.1007/s00392-006-0463-y.
Mehta AC, Rafanan AL, Bulkley R, Walsh M, DeBoer GE. Coronary spasm and cardiac arrest from carcinoid crisis during laser bronchoscopy. Chest. 1999;115(2):598–600. https://doi.org/10.1378/chest.115.2.598.
Rupp AB, Ahmadjee A, Morshedzadeh JH, Ranjan R. Carcinoid syndrome-induced ventricular tachycardia. Case Rep Cardiol. 2016;2016:9142598–3. https://doi.org/10.1155/2016/9142598.
Topol EJ, Fortuin NJ. Coronary artery spasm and cardiac arrest in carcinoid heart disease. Am J Med. 1984;77(5):950–2.
Bourgault C, Bergeron S, Bogaty P, Poirier P. A most unusual acute coronary syndrome. Can J Cardiol. 2006;22(5):429–32. https://doi.org/10.1016/s0828-282x(06)70930-8.
Meurice J, Boule S, Deswarte G, Brigadeau F, Modine T. Solitary atrial myocardial metastasis revealing ileal neuroendocrine carcinoma. Cardiovasc Pathol. 2012;21(4):355–7. https://doi.org/10.1016/j.carpath.2011.10.003.
Sareyyupoglu B, Connolly HM, Schaff HV. Surgical excision of right ventricular carcinoid tumor in a symptomatic patient without carcinoid valve disease. J Thorac Cardiovasc Surg. 2010;140(2):e23–5. https://doi.org/10.1016/j.jtcvs.2009.07.072.
Rich LL, Lisa CP, Nasser WK. Carcinoid pericarditis. Am J Med. 1973;54(4):522–7.
Dobson R, Burgess MI, Banks M, Pritchard DM, Vora J, Valle JW, et al. The association of a panel of biomarkers with the presence and severity of carcinoid heart disease: a cross-sectional study. PLoS One. 2013;8(9):e73679. https://doi.org/10.1371/journal.pone.0073679.
Caplin ME, Baudin E, Ferolla P, Filosso P, Garcia-Yuste M, Lim E, et al. Pulmonary neuroendocrine (carcinoid) tumors: European Neuroendocrine Tumor Society expert consensus and recommendations for best practice for typical and atypical pulmonary carcinoids. Ann Oncol. 2015;26(8):1604–20. https://doi.org/10.1093/annonc/mdv041.
Bhattacharyya S, Toumpanakis C, Caplin ME, Davar J. Usefulness of N-terminal pro-brain natriuretic peptide as a biomarker of the presence of carcinoid heart disease. Am J Cardiol. 2008;102(7):938–42. https://doi.org/10.1016/j.amjcard.2008.05.047.
•• Davar J, Connolly HM, Caplin ME, Pavel M, Zacks J, Bhattacharyya S, et al. Diagnosing and managing carcinoid heart disease in patients with neuroendocrine tumors: an expert statement. J Am Coll Cardiol. 2017;69(10):1288–304. https://doi.org/10.1016/j.jacc.2016.12.030 Expert consensus statement of the diagnosis and management of carcinoid heart disease.
Bhattacharyya S, Toumpanakis C, Burke M, Taylor AM, Caplin ME, Davar J. Features of carcinoid heart disease identified by 2- and 3-dimensional echocardiography and cardiac MRI. Circ Cardiovasc Imaging. 2010;3(1):103–11. https://doi.org/10.1161/circimaging.109.886846.
Bhattacharyya S, Burke M, Caplin ME, Davar J. Utility of 3D transoesophageal echocardiography for the assessment of tricuspid and pulmonary valves in carcinoid heart disease. Eur J Echocardiogr. 2011;12(1):E4. https://doi.org/10.1093/ejechocard/jeq099.
Fazlinezhad A, Moravvej Z, Azari A, Bigdelu L. Carcinoid heart disease and the utility of 3D trans-thoracic and trans-esophageal echocardiography: two clinical cases. J Saudi Heart Assoc. 2014;26(1):51–5. https://doi.org/10.1016/j.jsha.2013.08.004.
Moerman VM, Dewilde D, Hermans K. Carcinoid heart disease: typical findings on echocardiography and cardiac magnetic resonance. Acta Cardiol. 2012;67(2):245–8. https://doi.org/10.2143/ac.67.2.2154218.
Agha AM, Lopez-Mattei J, Donisan T, Balanescu D, Iliescu CA, Banchs J, et al. Multimodality imaging in carcinoid heart disease. Open Heart. 2019;6(1):e001060. https://doi.org/10.1136/openhrt-2019-001060.
Rajiah P, Kanne JP, Kalahasti V, Schoenhagen P. Computed tomography of cardiac and pericardiac masses. J Cardiovasc Comput Tomogr. 2011;5(1):16–29. https://doi.org/10.1016/j.jcct.2010.08.009.
Khalique OK, Cavalcante JL, Shah D, Guta AC, Zhan Y, Piazza N, et al. Multimodality imaging of the tricuspid valve and right heart anatomy. J Am Coll Cardiol Img. 2019;12(3):516–31. https://doi.org/10.1016/j.jcmg.2019.01.006.
Dobson R, Cuthbertson DJ, Jones J, Valle JW, Keevil B, Chadwick C, et al. Determination of the optimal echocardiographic scoring system to quantify carcinoid heart disease. Neuroendocrinology. 2014;99(2):85–93.
Robiolio PA, Rigolin VH, Wilson JS, Harrison JK, Sanders LL, Bashore TM, et al. Carcinoid heart disease. Circulation. 1995;92(4):790–5. https://doi.org/10.1161/01.CIR.92.4.790.
Denney WD, Kemp WE Jr, Anthony LB, Oates JA, Byrd BF 3rd. Echocardiographic and biochemical evaluation of the development and progression of carcinoid heart disease. J Am Coll Cardiol. 1998;32(4):1017–22.
Moller JE, Connolly HM, Rubin J, Seward JB, Modesto K, Pellikka PA. Factors associated with progression of carcinoid heart disease. The New England journal of medicine. 2003;348(11):1005–15.
Mansencal N, Mitry E, Bachet JB, Rougier P, Dubourg O. Echocardiographic follow-up of treated patients with carcinoid syndrome. The American journal of cardiology. 2010;105(11):1588–91.
Himelman RB, Schiller NB. Clinical and echocardiographic comparison of patients with the carcinoid syndrome with and without carcinoid heart disease. Am J Cardiol. 1989;63(5):347–52.
Bernheim AM, Connolly HM, Hobday TJ, Abel MD, Pellikka PA. Carcinoid heart disease. Prog Cardiovasc Dis. 2007;49(6):439–51. https://doi.org/10.1016/j.pcad.2006.12.002.
Rinke A, Muller HH, Schade-Brittinger C, Klose KJ, Barth P, Wied M, et al. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: a report from the PROMID Study Group. J Clin Oncol. 2009;27(28):4656–63. https://doi.org/10.1200/jco.2009.22.8510.
Caplin ME, Pavel M, Ćwikła JB, Phan AT, Raderer M, Sedláčková E, et al. Lanreotide in metastatic enteropancreatic neuroendocrine tumors. N Engl J Med. 2014;371(3):224–33. https://doi.org/10.1056/NEJMoa1316158.
Yao JC, Fazio N, Singh S, Buzzoni R, Carnaghi C, Wolin E, et al. Everolimus for the treatment of advanced, non-functional neuroendocrine tumours of the lung or gastrointestinal tract (RADIANT-4): a randomised, placebo-controlled, phase 3 study. Lancet (London, England). 2016;387(10022):968–77. https://doi.org/10.1016/s0140-6736(15)00817-x.
Faiss S, Pape U-F, Böhmig M, Dörffel Y, Mansmann U, Golder W, et al. Prospective, randomized, multicenter trial on the antiproliferative effect of lanreotide, interferon alfa, and their combination for therapy of metastatic neuroendocrine gastroenteropancreatic tumors—the International Lanreotide and Interferon Alfa Study Group. J Clin Oncol. 2003;21(14):2689–96. https://doi.org/10.1200/JCO.2003.12.142.
Bushnell DL Jr, O’Dorisio TM, O’Dorisio MS, Menda Y, Hicks RJ, Van Cutsem E, et al. 90Y-edotreotide for metastatic carcinoid refractory to octreotide. J Clin Oncol. 2010;28(10):1652–9. https://doi.org/10.1200/jco.2009.22.8585.
Pavel M, Horsch D, Caplin M, Ramage J, Seufferlein T, Valle J, et al. Telotristat etiprate for carcinoid syndrome: a single-arm, multicenter trial. J Clin Endocrinol Metab. 2015;100(4):1511–9. https://doi.org/10.1210/jc.2014-2247.
Connolly HM, Nishimura RA, Smith HC, Pellikka PA, Mullany CJ, Kvols LK. Outcome of cardiac surgery for carcinoid heart disease. J Am Coll Cardiol. 1995;25(2):410–6.
Warner RRP, Castillo JG. Carcinoid heart disease: the challenge of the unknown known∗. J Am Coll Cardiol. 2015;66(20):2197–200. https://doi.org/10.1016/j.jacc.2015.05.086.
Connolly HM, Schaff HV, Mullany CJ, Abel MD, Pellikka PA. Carcinoid heart disease: impact of pulmonary valve replacement in right ventricular function and remodeling. Circulation. 2002;106(12 Suppl 1):I51–i6.
Castillo JG, Filsoufi F, Rahmanian PB, Zacks JS, Warner RR, Adams DH. Early bioprosthetic valve deterioration after carcinoid plaque deposition. Ann Thorac Surg. 2009;87(1):321. https://doi.org/10.1016/j.athoracsur.2008.04.061.
Hassan SA, Banchs J, Iliescu C, Dasari A, Lopez-Mattei J, Yusuf SW. Carcinoid heart disease. Heart. 2017;103(19):1488–95. https://doi.org/10.1136/heartjnl-2017-311261.
Westberg G, Wangberg B, Ahlman H, Bergh CH, Beckman-Suurkula M, Caidahl K. Prediction of prognosis by echocardiography in patients with midgut carcinoid syndrome. Br J Surg. 2001;88(6):865–72. https://doi.org/10.1046/j.0007-1323.2001.01798.x.
Moller JE, Pellikka PA, Bernheim AM, Schaff HV, Rubin J, Connolly HM. Prognosis of carcinoid heart disease: analysis of 200 cases over two decades. Circulation. 2005;112(21):3320–7. https://doi.org/10.1161/circulationaha.105.553750.
• Connolly HM, Schaff HV, Abel MD, Rubin J, Askew JW, Li Z, et al. Early and late outcomes of surgical treatment in carcinoid heart disease. J Am Coll Cardiol. 2015;66(20):2189–96. https://doi.org/10.1016/j.jacc.2015.09.014 Surgical survival rates over time in patients who have had valve replacements for carcinoid heart disease. Surivival rates seemed to be tied to surgical center experience and era when the surgery was performed.
Zuetenhorst JM, Korse CM, Bonfrer JMG, Bakker RH, Taal BG. Role of natriuretic peptides in the diagnosis and treatment of patients with carcinoid heart disease. Br J Cancer. 2004;90(11):2073–9. https://doi.org/10.1038/sj.bjc.6601816.
Ramage JK, Ahmed A, Ardill J, Bax N, Breen DJ, Caplin ME, et al. Guidelines for the management of gastroenteropancreatic neuroendocrine (including carcinoid) tumours (NETs). Gut. 2012;61(1):6–32. https://doi.org/10.1136/gutjnl-2011-300831.
Robiolio PA, Rigolin VH, Harrison JK, Lowe JE, Moore JO, Bashore TM, et al. Predictors of outcome of tricuspid valve replacement in carcinoid heart disease. Am J Cardiol. 1995;75(7):485–8.
Author information
Authors and Affiliations
Ethics declarations
Conflict of Interest
Saamir A. Hassan, Nicolas L. Palaskas, Ali M. Agha, Cezar Iliescu, Juan Lopez-Mattei, Christopher Chen, Henry Zheng, and Syed Wamique Yusuf declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Valvular Heart Disease
Rights and permissions
About this article
Cite this article
Hassan, S.A., Palaskas, N.L., Agha, A.M. et al. Carcinoid Heart Disease: a Comprehensive Review. Curr Cardiol Rep 21, 140 (2019). https://doi.org/10.1007/s11886-019-1207-8
Published:
DOI: https://doi.org/10.1007/s11886-019-1207-8