Abstract
Conventional scintigraphic myocardial perfusion imaging with SPECT/CT or with PET/CT has evolved as an important clinical tool for the diagnostic assessment of flow-limiting epicardial lesions and risk stratification of patients with suspected CAD. By determining the relative distribution of radiotracer-uptake in the left-ventricular (LV) myocardium during stress, the presence of flow-limiting CAD lesions can be identified. While this approach successfully identifies epicardial coronary artery lesions, the presence of subclinical and non-obstructive CAD may go undetected. In this direction, the concurrent ability of PET/CT to assess absolute myocardial blood flow (MBF) in ml/g/min, rather that relative regional distribution of radiotracer-uptake, and myocardial flow reserve (MFR), expands the scope of conventional myocardial perfusion imaging from the identification of more advanced and flow-limiting epicardial lesions to (1) subclinical CAD, (2) an improved characterization of the extent and severity of CAD burden, and (3) the discovery of “balanced” reduction in myocardial blood flow as a consequence of 3 vessel CAD. Concurrent to the PET data, the CT component of the hybrid PET/CT allows the assessment of coronary artery calcification as an indirect surrogate for CAD burden, without contrast, or with contrast angiography to directly denote coronary stenosis and/or plaque morphology with CT. Hybrid PET/CT system, therefore, has the potential to not only identify and characterize flow-limiting epicardial lesions but also subclinical stages of functional and/or structural stages of CAD. Whether the application of PET/CT for an optimal assessment of coronary pathology, its downstream effects on myocardial perfusion, and coronary circulatory function will in effect lead to changes in clinical decision-making process, investiture in preventive health care, and improved long-term outcome, awaits scientific verification.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Despite the increasing use of non-invasive, contrast CT coronary angiography (CTCA) to denote coronary morphology, invasive coronary angiography still remains the ‘gold standard’ for evaluation of the luminal diameter of the epicardial artery and, thus, the severity and extent of obstructive CAD [1]. Previous landmark studies of Gould et al. [2–4] have demonstrated that resting coronary flow virtually remains normal unless the epicardial luminal diameter is decreased by 90 %, which can be related to a compensatory vasodilation of the downstream arteriolar resistance vessels in order to balance an increase in epicardial vascular resistance caused by an obstructive CAD. As to pharmacologically-induced hyperemic flow increases, an inverse relationship has been reported between increasing severity of epicardial coronary artery lesions with ≥50 % narrowing and the reduction of stress-induced regional coronary blood flow, myocardial flow reserve (MFR), and presumably stress-induced regional myocardial perfusion defect [5–9]. However, an increase in epicardial vascular resistance during hyperemic flows due to an advanced, flow-limiting lesion can also be accompanied by collateral flow supply and a relatively preserved MFR [10••]. For these reasons, there is no simple relationship between the morphologic severity of coronary artery lesions and physiologic stress-induced myocardial perfusion defects [5–9, 11–14], and a marked variability may exist in individual patients [5, 7].
Clinical decision-making process with regards to coronary interventional procedures to restore coronary blood flow is commonly based on the findings of stress-induced myocardial ischemia. In a subset of patients, however, with equivocal myocardial perfusion results or suspicion for diffuse ischemia owing to flow-limiting effects of coronary 3 vessel disease or main stem lesion adding non-invasive CT-coronary angiography to denote coronary morphology may be indicated [15]. This review aims to summarize contributions of PET/CT systems in the diagnosis of clinically-manifest and subclinical stages of the CAD, its diagnostic and prognostic implications, its potential influence on clinical decision-making process, and investiture in preventive health care.
Clinical Role of Perfusion and Flow Quantification with PET/CT
The current practice of interpreting nuclear myocardial perfusion studies entails an assessment of the relative left ventricular myocardial distribution of radiotracer-uptake during stress and at rest [16]. Regional reductions in radiotracer uptake during stress signify the flow-limiting consequences of epicardial coronary artery lesions. Conversely, myocardial regions with the highest radiotracer concentration (termed normal reference region) are presumably subtended by normal or non-flow limiting coronary arteries. In patients with multivessel CAD, such assessment of regional differences of the radiotracer-uptake commonly identify the “culprit” or most severe coronary artery luminal narrowing but not necessarily all flow-limiting coronary artery lesions. Such a limitation may be overcome by the concurrent ability of PET/CT to assess regional myocardial blood flow (MBF) in absolute terms (ml/g/min) during stress and rest, which allows to calculate the myocardial flow reserve (MFR) in each vascular territory rather than rely and/or assume that the normal reference region is supplied by a non-flow limiting coronary artery [10••, 17]. This approach widens the scope of conventional myocardial perfusion imaging from the sole identification of “culprit” or most severe CAD lesions to other flow-limiting lesions of intermediate range in multivessel disease. Accordingly, adding the non-invasive assessment of absolute MBF and MFR with PET/CT may improve the identification of the extent and severity of CAD burden, including “balanced” reduction of MBF that may often go undetected with the relative radiotracer uptake approach [10••]. The CT component of the hybrid PET/CT provides additional information on coronary artery calcification as an indirect surrogate for CAD burden. When contrast is added for CT angiography, coronary artery stenosis and/or plaque morphology may be directly visualized. Hybrid PET/CT system, therefore, allows the identification and characterization of flow-limiting effects of advanced epicardial lesions in clinically manifest CAD and subclinical, functional, and/or structural stages of the CAD process, which have important prognostic implications [10••, 18••].
The visual or semiquantitative assessment of stress-induced regional myocardial perfusion defects with PET or PET/CT allows a precise identification of flow-limiting epicardial lesions [10••, 18••, 19, 20]. When compared with conventional SPECT imaging, either with 201Thallium or 99mTc-labeled perfusion tracers, the higher spatial and contrast resolution of PET, with either 13N-ammonia or 82Rubidium, may account for a higher sensitivity in the identification of flow-limiting lesions [9, 21]. In addition, the photon attenuation–free images of PET imaging may explain a relative increase in specificity in the detection of advanced CAD. These advantages of PET perfusion imaging lead to a higher image quality, and a more confident and accurate interpretation of the stress-rest myocardial perfusion studies (Table 1) [21]. Consequently, 13N-ammonia or 82rubidium PET are now increasingly utilized in clinical practice for the detection of the CAD process [18••]. In a highly selected study population with an intermediate-to-high likelihood of CAD, the mean sensitivity and specificity of PET myocardial perfusion imaging for identifying ≥50 % or ≥70 % diameter stenosis on invasive coronary angiographic evaluation is reported to be in 92 % and 90 %, respectively [10••, 18••, 19, 22]. Since PET/CT perfusion studies are commonly free of photon-related artifacts, its use is well suited for the identification of CAD in women with breast attenuation artifact, and in individuals with large body habitus [19]. In these patient population the reported sensitivity and specificity of 82 Rubidium PET/CT for the detection of ≥70 % diameter stenosis, as determined with invasive coronary angiography, was 93 % and 83 %, respectively, with a diagnostic accuracy of 87 %.
Added Value of CT-Coronary Angiography (CTA) to Scintigraphic Myocardial Perfusion Scintigraphy
In the last decade, CTA has evolved as reliable tool to identify and characterize coronary morphology, which may be used clinically,
-
(1)
to clarify equivocal myocardial perfusion imaging results,
-
(2)
unmasking severe 3-vessel CAD or main stem lesions in patients with normal perfusion imaging but clinically suspicion for severe CAD, and
-
(3)
excluding morphologically significant CAD in patients with low-to-intermediate likelihood for the presence of CAD.
As mentioned before, there may be equivocal perfusion imaging results with PET/CT or more pronounced with SPECT/CT in a subset of patients [21], which may not allow one to draw definite conclusion on the presence or absence of flow-limiting epicardial lesions. In these cases, adding CTA may be indicated to verify whether an equivocal stress-induced regional perfusion defect on myocardial scintigraphic images is indeed due to a coronary stenosis ≥50 % diameter, or whether it rather reflects an artifact or false positive finding. Adding CTA to myocardial perfusion scintigraphy in these cases, therefore, can justify or avoid unnecessary invasive coronary angiography. Another scenario where CTA could be added to myocardial perfusion scintigraphy is the clinical suspicion for the presence of “diffuse” or balanced ischemia due to severe 3-vessel coronary disease or main stem lesion. These patients commonly present with anginal symptoms and ST-segment depression during stress testing but having normal or homogeneous radiotracer distribution on stress-rest myocardial perfusion scintigraphy. Under such circumstances, non-invasive CTA should be added in order to unmask these high risk patients with severe 3-vessel CAD or left main disease [10••]. The latter, however, even if PET-determined regional MFR is in all 3 major coronary territories reduced, should be preferentially confirmed by a peak stress transient ischemic cavity dilation of the left ventricle during dobutamine echocardiography or magnetic resonance imaging (MRI). Finally, CTA may be performed instead of stress-rest myocardial perfusion imaging in patients with low-to-intermediate likelihood for the presence of CAD. Here, CTA is primarily performed aiming to exclude CAD or coronary lesions ≥50 % stenosis [23]. Such a diagnostic approach has been demonstrated more recently not to increase the rate of other cardiovascular imaging modalities while being cost effective [24, 25].
Relation Between Morphologic Coronary Artery Lesion and Physiologic Myocardial Blood Flow
Despite the relatively high diagnostic accuracy of scintigraphic myocardial perfusion imaging in the detection of focal, flow-limiting CAD lesions, no simple relationship exists between the severity of epicardial lesions and stress-induced regional perfusion defects [5–8]. For example, stress-induced regional myocardial perfusion defects, when assessed with 201Thallium SPECT, were seen in 33 % of regions with 60 %–70 % stenosis, 54 % of regions with 70 %–80 % stenosis, and 86 % of regions with ≥80 % stenosis [26]. Although severity of stenosis was determined from non-invasive CTCA, which is certainly apt to some criticism [1, 27], this investigation emphasizes the complexity between focal CAD stenosis of >50 % and the corresponding myocardial perfusion during hyperemic flow increases [26]. Apart from an adaptive vasodilation of the coronary arteriolar vessels to balance increases in epicardial resistance due to significant epicardial narrowing of the arterial lumen [28•, 29••], other possible explanations for the observed discrepancy between CAD lesions and stress-induced myocardial perfusion defects include; (1) a potential overestimation of the severity of coronary artery stenosis by coronary angiography, and more so with non-invasive CTCA [27, 30], and (2) the absence of concurrent MBF quantification and determination of regional MFR with PET [10••, 18••]. Although with increasing severity of epicardial luminal narrowing, starting from ≥50 % diameter stenosis, a corresponding progressive decrease in MFR is observed, there is a high variability of MFR amongst individuals, in particular for epicardial lesions ranging between 50 %–80 % diameter stenosis [5, 7, 8]. The underlying mechanism of this variability in MFR may be seen in the individual capacity of the coronary arteriolar vessels to vasodilate in response to different vasomotor stimuli or cardiovascular risk factors related microvascular dysfunction [10••]. This also provides the rationale that a relatively preserved MFR may represent a compensatory response for CAD-induced epicardial resistance and, thereby, circumvent the manifestation of a stress-induced perfusion defect even in the presence of intermediate-to-high grade epicardial coronary artery stenosis [26]. Hence, medical therapy that improves coronary circulatory function (eg, HMG-CoA reductase or angiotensin-converting enzyme inhibitors) may improve or even prevent the manifestation of stress-induced myocardial ischemia [31, 32], as was observed in the sub-analysis of the recently performed COURAGE (Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation) trial [33•]. The induction of collaterals by hypoxic stimulus represents yet another compensatory factor that may contribute in the amelioration of stress-induced myocardial ischemia [34].
Quantification of Myocardial Blood Flow Reserve
Recent studies have shown that the quantification of regional MFRs with PET may lead to an improved characterization of the extent and severity of CAD burden beyond the visual and/or semiquantitative assessment of relative radiotracer uptake (Fig. 1) [10••, 35•]. MFR may identify “balanced” reduction of MBF in all vascular territories, including the so called “normal” reference region. In a previous clinical investigation where intracoronary Doppler ultrasound findings were compared with both 201Tl SPECT imaging and coronary angiography, Doppler-derived coronary flow reserve of <1.7 during adenosine hyperemia accurately predicted the presence of exercise-induced ischemia on stress 201Tl imaging in regions with intermediate coronary stenosis (40 % to 70 %) while the coronary angiogram alone did not [36]. This magnitude of a diminished coronary flow reserve during pharmacologic-induced hyperemic flows in territories of scintigraphic perfusion defects is similar to other investigations in this field [36–39]. These observations from invasive Doppler recordings during cineangiography in poststenotic and nonstenotic reference coronary arteries provide credence to the concept of noninvasive assessment of MFR with PET to characterize the downstream functional consequences of anatomically mild-to-intermediate CAD lesions by CTCA.
13N-ammonia PET in the evaluation of CAD. A, Myocardial perfusion study with 13N-ammonia PET/CT during dipyridamole stimulation and at rest in a 61-year-old patient with arterial hypertension and type 2 diabetes mellitus. On stress images, there is a moderately decreased perfusion defect involving the mid-to-distal anterior, anteroseptal, and apical regions of the left ventricle, which becomes reversible on the rest images. Uptake is preserved in the lateral and inferior regions. B, Regional myocardial blood flow quantification (MBF) and myocardial flow reserve (MFR) calculation with 13N-ammonia PET/CT and tracer kinetic modeling. The summarized quantitative data suggest a distinct impairment of the MFR not only in the (LAD) territory, but also in the RCA and LCX vascular territories (regional MFR <2.0). C, Invasive coronary angiography in this patient demonstrated a proximal occlusion of the LAD, 80 % stenosis in the proximal segments of the LCX (left panel), and sequential 50 % to 60 % lesions in the RCA (right panel). Corresponding regional MFRs are indicated for each vascular territory. (Reproduced with permission from Schindler TH, Schelbert HR, Quercioli A, et al. Cardiac PET imaging for the detection and monitoring of coronary artery disease and microvascular health. JACC Cardiovasc Imaging. 3:623–40) [10••] MBF, myocardial blood flow; MFR, myocardial flow reserve; LAD, left anterior descending artery; LCx, left circumflex artery; RCA, right coronary artery
Variability in Published Threshold Values for Absolute MBF and MFR
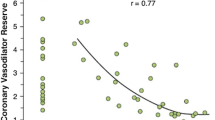
Several PET flow studies [40] have strived to define optimal threshold values of hyperemic MBFs or MFR for the identification of flow-limiting CAD lesions. The estimation of a flow threshold value of hyperemic MBFs or MFR with PET flow studies in concert with tracer kinetic modeling, however, is dependent on the methodological approach and radiotracer applied [10••, 18••]. For example, using 13N-ammonia PET, the diagnostic value of hyperemic MBF, MFR, and the relative radiotracer uptake for the detection of ≥70 % diameter coronary artery stenosis was highest with a hyperemic adenosine-stimulated MBF threshold of <1.85 ml/g/min, while most accurate with a MFR <2.0 [41]. Conversely, using 15O-water, a hyperemic MBF threshold of 2.5 ml/g/min was reported to be most accurate for the detection of ≥50 % diameter coronary artery stenosis [42]. In a recent study comparing SPECT with 15O-water PET myocardial perfusion studies [43], vascular territories subtended by >50 % diameter stenosis that exhibited normal radiotracer uptake on SPECT showed abnormally reduced MFR on PET with a mean MFR value of 2.2. Others have shown a nonlinear decrease in hyperemic 82Rubidium PET MBFs with increasing severity of CAD lesions [44]. Interestingly, it appeared that by applying MFR threshold levels of 1.0 and 1.7, epicardial lesions between 70 %–80 % could be differentiated from those with 50 %–69 % stenosis. Nevertheless, it is important to point out that coronary microvascular dysfunction in patients with or without focal flow-limiting epicardial coronary artery lesions and cardiovascular risk factors may also account for reductions in hyperemic MBFs [10••, 45, 46, 47•]. Another problem for the detection of flow-limiting lesions using the MFR may arise from a grey zone, which may not allow a definite identification of down-stream effects of focal epicardial lesions on hyperemic coronary flows [10••, 26]. This is particularly true for epicardial lesions ≥50 %–70 % and a PET-determined MFR between 2.0–2.5 [5, 7, 10••]. Consequently, an abnormally reduced MFR in myocardial territories subtended by >50 %–70 % diameters stenosis but without stress-induced scintigraphic myocardial perfusion defects may be related to downstream, fluid dynamic effects of the CAD lesion and/or a dysfunction of the coronary arteriolar vessels. The exact contributions of an epicardial lesions and coronary arteriolar dysfunction to the reduced MFR remains uncertain, while with increasing stenosis severity it can be assumed to be progressively shifted to the increase in epicardial resistance (Fig. 2) [40]. Overall, the additional non-invasive evaluation of regional MBFs and MFR to the conventional visual analysis of standard myocardial stress-rest PET radiotracer uptake images leads to an increase in the sensitivity for detecting hemodynamically significant multivessel disease, but at the expense of decreased specificity [10••].
Algorithm for the integration of PET perfusion images and MFR. Algorithm for the integration of PET myocardial perfusion imaging and absolute myocardial blood flow (MBF) and flow reserve (MFR) quantification in individuals with suspected or an intermediate risk for developing CAD for clinical decision making towards revascularization or preventive medical therapy is shown. (Reproduced with permission from Schindler TH, Schelbert HR, Quercioli A, et al. Cardiac PET imaging for the detection and monitoring of coronary artery disease and microvascular health. JACC Cardiovasc Imaging. 3:623–40) [10••]
Incremental Prognostic Value of Absolute MBF and MFR in Patients with CAD
Emerging data suggest incremental prognostic value of absolute MBF and MFR in patients with suspected CAD to relative myocardial perfusion assessment with PET (Fig. 3) [48••, 49••, 50, 51•, 52•]. Using 13N-ammonia PET, an abnormal global MFR <2.0 was found to be independently associated with a higher annual event rate for major adverse cardiac events and cardiac death over 3 years compared with normal MFR [49••]. In subjects with abnormal perfusion, MFR remained predictive throughout the 10-year follow-up. When both stress myocardial perfusion and MFR were normal, the “warranty” period of event-free survival was about 3 years. Subsequent papers from different institutions confirmed the incremental prognostic value of MFR to visual assessment of regional radiotracer uptake using 82Rubidium PET [49••, 50, 52•]. Globally reduced MFR was reported to be an independent predictor of 3-vessel CAD and, therefore, provided added value to relative myocardial perfusion imaging [35•, 49••]. Notably, a preserved MFR rendered the presence of 3-vessel CAD very unlikely, while a continuous reduction in MFR predicted an increasing probability of 3 vessel disease [35•]. Interestingly, in the latter study, 40 % of patients with 3 vessel CAD did not present high risk findings such as stress-induced ST-segment depression, transient ischemic dilation, and LVEF reserve <0 %. In these patients, the identification of 3-vessel CAD may certainly identify some flow-limiting effects of epicardial lesions on hyperemic flow, while these CAD lesions may not have yet induced a more severe ischemic state associated with myocardial stunning leading to transient ischemic dilation or a decrease in stress-induced left-ventricular function. Among diabetic patients without CAD, those with impaired MFR experienced higher cardiac deaths that were comparable with patients with prior CAD while among diabetic patients with preserved MFR cardiac mortality rates were comparable with non-diabetics [52•]. It is important to point out, however, that a marked reduction in PET/CT-determined MFR can also reflect a severe dysfunction of the coronary microcirculation in patients without significant epicardial lesions but with cardiovascular risk factors [10••, 46, 47•]. Reductions in regional MFRs, therefore, should always be interpreted with underlying coronary anatomy and/or cardiovascular risk profile (Fig. 2) [10••].
PET-Determined Coronary Endothelial Vasoreactivity and Prognosis. Kaplan-Meier analyses in patients with cardiovascular risk factors and normal coronary angiograms undergoing assessment of myocardial blood flow (MBF) response to cold pressor test (CPT) with positron emission tomography (PET). Impairment of PET-measured and endothelium-related MBF responses to sympathetic stimulation with cold pressor testing are associated with a higher risk for cardiac events (during long-term follow-up) as compared with those with normal flow increases; normal (%ΔMBF ≥40 %), impaired (%ΔMBF > 0 % and <40 %), and decreased (%ΔMBF ≤0 %). (Reproduced with permission from Schindler TH, Nitzsche EU, Schelbert HR, et al. Positron emission tomography-measured abnormal responses of myocardial blood flow to sympathetic stimulation are associated with the risk of developing cardiovascular events. J Am Coll Cardiol. 2005;45:1505–12) [51•]
In patients with advanced ischemic cardiomyopathy, who were not amenable to coronary revascularization, the incidence of future cardiovascular events increased with the severity of abnormally-reduced MFR [53]. These observations give rise to a new concept that, apart from restoration of coronary blood flow with PCI or CABG in patients with hemodynamically obstructive CAD lesions, an improvement or even normalization of coronary circulatory dysfunction with preventive medical care should be strived for in order to further improve the cardiovascular outcome in these patients.
The Potential Value of LVEF Reserve for the Detection of Multivessel CAD
Previous studies have reported that the additional evaluation of post-stress LV function on gated SPECT may improve the detection of balanced myocardial ischemia by up to 25 % [54]. Since pharmacologic vasodilation or dobutamine stimulation during gated PET affords the unique opportunity for the evaluation of LV function at peak hyperemic stress rather than during post-stress conditions, gated PET myocardial perfusions studies may markedly improve the detection of flow-limiting effects of severe left main coronary artery stenosis or advanced 3-vessel CAD by unmasking the effects of stress-induced global ischemia on global LV function. When vasodilator LVEF reserve was assessed among patients undergoing clinically indicated gated 82Rubidium PET/CT at rest and during pharmacologic vasodilation, LVEF reserve ≥5 % yielded a high negative predictive value of 97 % to exclude 3-vessel disease but a low positive predictive value of only 41 % [55]. Beyond the low positive predictive value, it is important to point out that only 68 of 510 patients had coronary angiography, and of whom only 17 had either severe left main or 3-vessel CAD. This represents a highly selected subset of patients who presumably were referred to invasive coronary angiography on the basis of an abnormal PET myocardial perfusion and/or CT angiography study. Moreover, the addition of an abnormal LVEF reserve to myocardial perfusion defects did not increase the overall sensitivity of perfusion imaging (92 % vs 94 %) [55]. Hence, it would be important to test whether the findings from this observational study holds true in a «prospectively» conducted study to determine the true incremental value of abnormal LVEF reserve to myocardial perfusion defects and MFR.
Identification of Subclinical CAD
PET quantification of MBF responses to various stimuli and at rest affords the non-invasive assessment and characterization of the coronary circulatory capacity, which carries important diagnostic and prognostic information in asymptomatic cardiovascular risk individuals [56–60]. Such abnormalities of coronary circulatory function have been appreciated as a functional precursor of the CAD process [61, 62], which may also exist in the absence of structural alterations of the arterial wall as evaluated by IVUS [63] or with CTCA [46]. PET-determined non-invasive probes for coronary circulatory function include measurements of flow responses to adenosine or dipyridamole, as predominantly endothelial-independent, or to cold pressor testing, as predominantly endothelial-dependent, stimulation of coronary circulatory function [64]. Although MBF changes to pharmacologic vasodilation of the coronary arteriolar vessels with adenosine or dipyridamole can be seen as predominantly vascular smooth muscle dependent, a flow-related or shear-sensitive and endothelium-mediated dilation of the up-stream vessels may exist [65, 66]. Indeed, inhibition of endogenous NO synthesis by NG-nitro-L-arginine methyl ester (L-NAME) has been shown to attenuate myocardial flow increases during adenosine-induced hyperemia by about 20 %–25%, indicating that coronary vasodilation by adenosine is indeed partly endothelium dependent [65, 66]. Because of this, hyperemic flow response to pharmacologic vasodilation may also signify the “integrated coronary circulatory function” [47•, 64]. For example, in asymptomatic overweight individuals with normal stress-rest myocardial perfusion PET images, concurrent MFR evaluation has unmasked a reduced endothelium-dependent coronary vasodilator capacity in response to sympathetic stimulation with cold pressor testing (CPT), while the integrated coronary vascular response to pharmacologic vasodilation was still preserved [67]. In obese individuals again, not only endothelium-mediated flow responses to CPT but also pharmacologically-induced hyperemic flows were abnormally reduced [67]. These observations emphasize the notion that initial stages of the vascular injury may affect predominantly the vascular endothelium, whereas more advanced stages of coronary risk factor states, such as increases in oxidative stress burden, may also cause alterations in smooth muscle cell vasodilator function [47•, 68]. Similarly, in individuals with increasing severity of insulin resistance and carbohydrate intolerance, a progressive worsening of coronary endothelium function may manifest, while the impairment of the integrated function of the coronary circulation or the total vasodilator capacity can be appreciated in type 2 diabetes mellitus [69]. This is also in agreement with previously reported inverse association between elevations in plasma glucose levels and PET-determined coronary vasodilator capacity in type 2 diabetic patients [70], emphasizing direct adverse effects of elevated plasma glucose levels on coronary circulatory function in diabetes mellitus. Such functional abnormalities of the coronary circulation as determined with PET, reflecting an index of the overall stress burden imposed by various cardiovascular risk factors on the arterial wall and the genetic susceptibility of the vascular endothelium, also provide important prognostic information as numerous invasive and non-invasive flow studies have demonstrated [48••, 49••, 51•]. Not surprisingly, specific assessment of coronary endothelial dysfunction in cardiovascular risk individuals but with normal angiogram demonstrated that the incidence of future cardiovascular events increased with the severity of abnormally-reduced and endothelium-related flow responses to CPT (Fig. 3) [51•].
Conclusions
Cardiac myocardial perfusion PET/CT affords the noninvasive evaluation and quantification of MBF and myocardial flow reserve (MFR), which extends the scope of conventional myocardial perfusion imaging from identification of advanced and flow-limiting epicardial coronary artery lesions to early functional stages of the atherosclerotic process. This approach enables an improved identification characterization of the extent and severity of end-stage CAD burden, the detection of “balanced” reduction of MFR due to main stem lesion, or multiple vessel disease, and the identification of subclinical CAD. Whether the application of PET/CT for an optimal assessment of coronary pathology, its downstream effects on myocardial perfusion and coronary circulatory function will in effect lead to changes in clinical decision-making process, investiture in preventive health care, and improved long-term outcome, awaits scientific verification.
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance, • Of importance
Leber AW, Knez A, von Ziegler F, Becker A, Nikolaou K, Wintersberger B, et al. Quantification of obstructive and nonobstructive coronary lesions by 64-slice computed tomography: a comparative study with quantitative coronary angiography and intravascular ultrasound. J Am Coll Cardiol. 2005;46:147–54.
Gould KL. Quantification of coronary artery stenosis in vivo. Circ Res. 1985;57:341–53.
Gould KL, Lipscomb K, Calvert C. Compensatory changes of the distal coronary vascular bed during progressive coronary constriction. Circulation. 1975;51:1085–94.
Gould KL. Assessment of coronary stenoses with myocardial perfusion imaging during pharmacologic coronary vasodilatation. IV. Limits of detection of stenosis with idealized experimental cross-sectional myocardial imaging. Am J Cardiol. 1978;42:761–8.
Uren NG, Melin JA, De Bruyne B, Wijns W, Baudhuin T, Camici PG. Relation between myocardial blood flow and the severity of coronary-artery stenosis. N Engl J Med. 1994;330:1782–8.
Krivokapich J, Czernin J, Schelbert HR. Dobutamine positron emission tomography: absolute quantitation of rest and dobutamine myocardial blood flow and correlation with cardiac work and percent diameter stenosis in patients with and without coronary artery disease. J Am Coll Cardiol. 1996;28:565–72.
Di Carli M, Czernin J, Hoh CK, Gerbaudo VH, Brunken RC, Huang SC, et al. Relation among stenosis severity, myocardial blood flow, and flow reserve in patients with coronary artery disease. Circulation. 1995;91:1944–51.
Demer LL, Gould KL, Goldstein RA, Kirkeeide RL, Mullani NA, Smalling RW, et al. Assessment of coronary artery disease severity by positron emission tomography. Comparison with quantitative arteriography in 193 patients. Circulation. 1989;79:825–35.
Schelbert HR DL. Evaluation of myocardial blood flow in cardiac disease: second edition. Philadelphia: W.B. Saunders; 1996.
•• Schindler TH, Schelbert HR, Quercioli A, Dilsizian V. Cardiac PET imaging for the detection and monitoring of coronary artery disease and microvascular health. JACC Cardiovasc Imaging. 2010;3:623–40. This review provides an excellent update on clinical and research possibilities of noninvasive MBF quantification with PET/CT.
Goldstein RA, Kirkeeide RL, Demer LL, Merhige M, Nishikawa A, Smalling RW, et al. Relation between geometric dimensions of coronary artery stenoses and myocardial perfusion reserve in man. J Clin Invest. 1987;79:1473–8.
Marcus ML, Skorton DJ, Johnson MR, Collins SM, Harrison DG, Kerber RE. Visual estimates of percent diameter coronary stenosis: “a battered gold standard”. JAm Coll Cardiol. 1988;11:882–5.
Vogel RA. Assessing stenosis significance by coronary arteriography: are the best variables good enough? J Am Coll Cardiol. 1988;12:692–3.
White CW, Wright CB, Doty DB, Hiratza LF, Eastham CL, Harrison DG, et al. Does visual interpretation of the coronary arteriogram predict the physiologic importance of a coronary stenosis? N Engl J Med. 1984;310:819–24.
Tamaki N, Yonekura Y, Senda M, Yamashita K, Koide H, Saji H, et al. Value and limitation of stress thallium-201 single photon emission computed tomography: comparison with nitrogen-13 ammonia positron tomography. J Nucl Med. 1988;29:1181–8.
Machac J, Bacharach SL, Bateman TM, Bax JJ, Beanlands R, Bengel F, et al. Positron emission tomography myocardial perfusion and glucose metabolism imaging. J Nucl Cardiol. 2006;13:e121–51.
Knuuti J. Hybrid SPECT-CT and PET-CT: current concepts and developments. Curr Cardiovasc Imaging Rep. 2011;3:468–75.
•• Bengel FM, Higuchi T, Javadi MS, Lautamäki R. Cardiac positron emission tomography. J Am Coll Cardiol. 2009;54:1–15. This review provides very valuable information on the clinical role of PET-determined myocardial perfusion and flow.
Sampson UK, Dorbala S, Limaye A, Kwong R, Di Carli MF. Diagnostic accuracy of rubidium-82 myocardial perfusion imaging with hybrid positron emission tomography/computed tomography in the detection of coronary artery disease. J Am Coll Cardiol. 2007;49:1052–8.
Hsiao EM, Ali B, Dorbala S. Clinical role of hybrid imaging. Curr Cardiovasc Imaging Rep. 2010;3:325–35.
Flotats A, Bravo PE, Fukushima K, Chaudhry MA, Merrill FM, Bengel FM. 82-Rb PET myocardial perfusion imaging is superior to (99m)Tc-labelled agent SPECT in patients with known or suspected coronary artery disease. Eur J Nucl Med Mol Imaging. 2012;39:1233–9.
Bateman TM, Heller GV, McGhie AI, Friedman JD, Case JA, Bryngelson JR, et al. Diagnostic accuracy of rest/stress ECG-gated Rb-82 myocardial perfusion PET: comparison with ECG-gated Tc-99m sestamibi SPECT. J Nucl Cardiol. 2006;13:24–33.
Achenbach S, Dilsizian V, Kramer CM, Zoghbi WA. The year in coronary artery disease. JACC Cardiovasc Imaging. 2009;2:774–86.
Fiechter M, Ghadri JR, Wolfrum M, Kuest SM, Pazhenkottil AP, Herzog BA, et al. Downstream resource utilization following hybrid cardiac imaging with an integrated cadmium-zinc-telluride/64-slice CT device. Eur J Nucl Med Mol Imaging. 2011;39:430–6.
Pazhenkottil AP, Nkoulou RN, Ghadri JR, Herzog BA, Kuest SM, Husmann L, et al. Impact of cardiac hybrid single-photon emission computed tomography/computed tomography imaging on choice of treatment strategy in coronary artery disease. Eur Heart J. 2011;32:2824–9.
Sato A, Hiroe M, Tamura M, Ohigashi H, Nozato T, Hikita H, et al. Quantitative measures of coronary stenosis severity by 64-Slice CT angiography and relation to physiologic significance of perfusion in nonobese patients: comparison with stress myocardial perfusion imaging. J Nucl Med. 2008;49:564–72.
Boogers MJ, Broersen A, van Velzen JE, de Graaf FR, El-Naggar HM, Dijkstra J, et al. Automated quantification of coronary plaque with computed tomography: comparison with intravascular ultrasound using a dedicated registration algorithm for fusion-based quantification. Eur Heart J. 2012;33:1007–16.
• Gould KL. Does coronary flow trump coronary anatomy. JACC Cardiovasc Imaging. 2009;2:1009–23. Excellent article which compares the clinical value of coronary flow vs coronary stenosis assessment.
•• Gould KL. Coronary flow reserve and pharmacologic stress perfusion imaging: beginnings and evolution. JACC Cardiovasc Imaging. 2009;2:664–9. This review article provides a very thorough update on the evaluation and interpretation of the coronary flow reserve.
Keane D, Haase J, Slager CJ, Montauban van Swijndregt E, Lehmann KG, Ozaki Y, et al. Comparative validation of quantitative coronary angiography systems. Results and implications from a multicenter study using a standardized approach. Circulation. 1995;91:2174–83.
Baller D, Notohamiprodjo G, Gleichmann U, Holzinger J, Weise R, Lehmann J. Improvement in coronary flow reserve determined by positron emission tomography after 6 months of cholesterol-lowering therapy in patients with early stages of coronary atherosclerosis. Circulation. 1999;99:2871–5.
Mancini GB, Henry GC, Macaya C, O’Neill BJ, Pucillo AL, Carere RG, et al. Angiotensin-converting enzyme inhibition with quinapril improves endothelial vasomotor dysfunction in patients with coronary artery disease. The TREND (Trial on Reversing ENdothelial Dysfunction) Study. Circulation. 1996;94:258–65.
• Shaw LJ, Berman DS, Maron DJ, Mancini GB, Hayes SW, Hartigan PM, et al. Optimal medical therapy with or without percutaneous coronary intervention to reduce ischemic burden: results from the Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation (COURAGE) trial nuclear substudy. Circulation. 2008;117:283–91. This substudy of the COURAGE trial emphasizes that reduction in ischemic burden via PCI or optimal medical treatment results in a substantial lower risk for death and myocardial infarction.
Berry C, Balachandran KP, L’Allier PL, Lespérance J, Bonan R, Oldroyd KG. Importance of collateral circulation in coronary heart disease. Eur Heart J. 2007;28:278–91.
• Ziadi MC, Dekemp RA, Williams K, Guo A, Renaud JM, Chow BJ, et al. Does quantification of myocardial flow reserve using rubidium-82 positron emission tomography facilitate detection of multivessel coronary artery disease. J Nucl Cardiol. 2012;19:670–80. These observations are first to describe the use of MFR to enhance the detection of multivessel disease in clinical routine.
Joye JD, Schulman DS, Lasorda D, Farah T, Donohue BC, Reichek N. Intracoronary Doppler guide wire vs stress single-photon emission computed tomographic thallium-201 imaging in assessment of intermediate coronary stenoses. J Am Coll Cardiol. 1994;24:940–7.
Miller DD, Donohue TJ, Younis LT, Bach RG, Aguirre FV, Wittry MD, et al. Correlation of pharmacological 99mTc-sestamibi myocardial perfusion imaging with poststenotic coronary flow reserve in patients with angiographically intermediate coronary artery stenoses. Circulation. 1994;89:2150–60.
Deychak YA, Segal J, Reiner JS, Nachnani S. Doppler guide wire-derived coronary flow reserve distal to intermediate stenoses used in clinical decision making regarding interventional therapy. Am Heart J. 1994;128:178–81.
Donohue TJ, Miller DD, Bach RG, Tron C, Wolford T, Caracciolo EA, et al. Correlation of poststenotic hyperemic coronary flow velocity and pressure with abnormal stress myocardial perfusion imaging in coronary artery disease. Am J Cardiol. 1996;77:948–54.
Kern MJ. Coronary physiology revisited: practical insights from the cardiac catheterization laboratory. Circulation. 2000;101:1344–51.
Hajjiri MM, Leavitt MB, Zheng H, Spooner AE, Fischman AJ, Gewirtz H. Comparison of positron emission tomography measurement of adenosine-stimulated absolute myocardial blood flow vs relative myocardial tracer content for physiological assessment of coronary artery stenosis severity and location. JACC Cardiovasc Imaging. 2009;2:751–8.
Nesterov SV, Han C, Maki M, Kajander S, Naum AG, Helenius H, et al. Myocardial perfusion quantitation with 15O-labelled water PET: high reproducibility of the new cardiac analysis software (Carimas). Eur J Nucl Med Mol Imaging. 2009;36:1594–602.
Yoshinaga K, Katoh C, Noriyasu K, Iwado Y, Furuyama H, Ito Y, et al. Reduction of coronary flow reserve in areas with and without ischemia on stress perfusion imaging in patients with coronary artery disease: a study using oxygen 15-labeled water PET. J Nucl Cardiol. 2003;10:275–83.
Anagnostopoulos C, Almonacid A, El Fakhri G, Curillova Z, Sitek A, Roughton M, et al. Quantitative relationship between coronary vasodilator reserve assessed by 82Rb PET imaging and coronary artery stenosis severity. Eur J Nucl Med Mol Imaging. 2008;35:1593–601.
Quercioli A, Pataky Z, Vincenti G, Makoundou V, Di Marzo V, Montecucco F, et al. Elevated endocannabinoid plasma levels are associated with coronary circulatory dysfunction in obesity. Eur Heart J. 2011;32:1369–78.
Valenta I, Quercioli A, Vincenti G, Nkoulou R, Dewarrat S, Rager O, et al. Structural epicardial disease and microvascular function are determinants of an abnormal longitudinal myocardial blood flow difference in cardiovascular risk individuals as determined with PET/CT. J Nucl Cardiol. 2010;17:1023–33.
• Valenta I, Dilsizian V, Quercioli A, Schelbert HR, Schindler TH. The influence of insulin resistance, obesity, and diabetes mellitus on vascular tone and myocardial blood flow. Curr Cardiol Rep. 2012;14:217–25. Excellent review article describing the potential of PET flow quantification in the assessment of coronary circulatory dysfunction in the pre-diabetic and diabetic state.
•• Herzog BA, Husmann L, Valenta I, Gaemperli O, Siegrist PT, Tay FM, et al. Long-term prognostic value of 13N-ammonia myocardial perfusion positron emission tomography added value of coronary flow reserve. J Am Coll Cardiol. 2009;54:150–6. This is a retrospectively performed study 725 in 245 patients with suspicion for CAD. In patients with normal PET perfusion imaging the evaluation of the MFR proved to be a strong outcome predictor, whereas a reduced MFR in those with stress-induced perfusion defects improved the prediction of an adverse outcome.
•• Ziadi MC, Dekemp RA, Williams KA, Guo A, Chow BJ, Renaud JM, et al. Impaired myocardial flow reserve on rubidium-82 positron emission tomography imaging predicts adverse outcomes in patients assessed for myocardial ischemia. J Am Coll Cardiol. 2011;58:740–8. This study enrolled prospectively 704 patients with suspected and known CAD who underwent 82Rb PET perfusion measurements and MFR calculation. The noninvasively calculated MFR predicted hard cardiac events and major adverse cardiac events independent of the presence of stress-induced myocardial perfusion defects and other parameters.
Fukushima K, Javadi MS, Higuchi T, Lautamäki R, Merrill J, Nekolla SG, et al. Prediction of short-termcardiovascular events using quantification of global myocardial flow reserve in patients referred for clinical 82Rb PET perfusion imaging. J Nucl Med. 2011;52:726–32.
• Schindler TH, Nitzsche EU, Schelbert HR, Olschewski M, Sayre J, Mix M, et al. Positron emission tomography-measured abnormal responses of myocardial blood flow to sympathetic stimulation are associated with the risk of developing cardiovascular events. J Am Coll Cardiol. 2005;45:1505–12. This study was the first to describe that impaired myocardial blood flow increases to sympathetic stimulation with cold pressor test as determined with 13N-ammonia PET was predictive of future cardiovascular events in cardiovascular risk patients with normal coronary angiograms.
• Murthy VL, Naya M, Foster CR, Gaber M, Hainer J, Klein J, et al. Association between coronary vascular dysfunction and cardiac mortality in patients with and without diabetes mellitus. Circulation. 2012;126(15):1858–68. Interesting retrospective analysis in 2783 consecutive patients undergoing PET perfusion and flow measurements. As it was observed, among diabetic patients without CAD, those with impaired CFR have event rates comparable with patients with prior CAD while those with preserved CFR have event rates comparable to non-diabetics.
Tio RA, Dabeshlim A, Siebelink HM, de Sutter J, Hillege HL, Zeebregts CJ, et al. Comparison between the prognostic value of left ventricular function and myocardial perfusion reserve in patients with ischemic heart disease. J Nucl Med. 2009;50:214–9.
Smith WH, Kastner RJ, Calnon DA, Segalla D, Beller GA, Watson DD. Quantitative gated single photon emission computed tomography imaging: a counts-based method for display and measurement of regional and global 768 ventricular systolic function. J Nucl Cardiol. 1997;4:451–63.
Dorbala S, Vangala D, Sampson U, Limaye A, Kwong R, Di Carli MF. Value of vasodilator left ventricular ejection fraction reserve in evaluating the magnitude of myocardium at risk and the extent of angiographic coronary artery disease: a 82Rb PET/CT study. J Nucl Med. 2007;48:349–58.
Zeiher AM, Drexler H, Wollschlager H, Just H. Modulation of coronary vasomotor tone in humans. Progressive endothelial dysfunction with different early stages of coronary atherosclerosis. Circulation. 1991;83:391–401.
Schindler TH, Nitzsche EU, Olschewski M, Brink I, Mix M, Facta A, et al. PET-Measured responses of MBF to cold pressor testing correlate with indices of coronary vasomotion on quantitative coronary angiography. J Nucl Med. 2004;45:419–28.
Dimmeler S, Fleming I, Fisslthaler B, Hermann C, Busse R, Zeiher AM. Activation of nitric oxide synthase in endothelial cells by Akt-dependent phosphorylation. Nature. 1999;399:601–5.
Campisi R, Czernin J, Schoder H, Sayre JW, Schelbert HR. L-Arginine normalizes coronary vasomotion in long-term smokers. Circulation. 1999;99:491–7.
Czernin J, Barnard RJ, Sun KT, Krivokapich J, Nitzsche E, Dorsey D, et al. Effect of short-term cardiovascular conditioning and low-fat diet on myocardial blood flow and flow reserve. Circulation. 1995;92:197–204.
Lerman A, Zeiher AM. Endothelial function: cardiac events. Circulation. 2005;111:363–8.
Rubinshtein R, Yang EH, Rihal CS, Prasad A, Lennon RJ, Best PJ, et al. Coronary microcirculatory vasodilator function in relation to risk factors among patients without obstructive coronary disease and low to intermediate Framingham score. Eur Heart J. 2010;31:936–42.
Reddy KG, Nair RN, Sheehan HM, Hodgson JM. Evidence that selective endothelial dysfunction may occur in the absence of angiographic or ultrasound atherosclerosis in patients with risk factors for atherosclerosis. J Am Coll Cardiol. 1994;23:833–43.
Schindler TH, Zhang XL, Vincenti G, Lerch R, Schelbert HR. Role of PET in the evaluation and understanding of coronary physiology. J Nucl Cardiol. 2007;14:589–603.
Buus NH, Bottcher M, Hermansen F, Sander M, Nielsen TT, Mulvany MJ. Influence of nitric oxide synthase and adrenergic inhibition on adenosine-induced myocardial hyperemia. Circulation. 2001;104:2305–10.
Tawakol A, Forgione MA, Stuehlinger M, Alpert NM, Cooke JP, Loscalzo J, et al. Homocysteine impairs coronary microvascular dilator function in humans. J Am Coll Cardiol. 2002;40:1051–8.
Schindler TH, Cardenas J, Prior JO, Facta AD, Kreissl MC, Zhang XL, et al. Relationship between increasing body weight, insulin resistance, inflammation, adipocytokine leptin, and coronary circulatory function. J Am Coll Cardiol. 2006;47:1188–95.
Munzel T, Daiber A, Ullrich V, Mülsch A. Vascular consequences of endothelial nitric oxide synthase uncoupling for the activity and expression of the soluble guanylyl cyclase and the cGMP- dependent protein kinase. Arterioscler Thromb Vasc Biol. 2005;25:551–7.
Prior JO, Quinones MJ, Hernandez-Pampaloni M, Facta AD, Schindler TH, Sayre JW, et al. Coronary circulatory dysfunction in insulin resistance, impaired glucose tolerance, and type 2 diabetes mellitus. Circulation. 2005;111:2291–8.
Di Carli MF, Janisse J, Grunberger G, Ager J. Role of chronic hyperglycemia in the pathogenesis of coronary microvascular dysfunction in diabetes. J Am Coll Cardiol. 2003;41:1387–93.
Acknowledgments
This work was supported by Research Grant 3200B0-122237 of the Swiss National Science Foundation (SNF), with contributions of the Clinical Research Center, University Hospital and Faculty of Medicine, Geneva, and the Louis-Jeantet Foundation, Gustave and Simone Prevot, and Swiss Heart Foundation.
Disclosure
Conflicts of interest: I. Valenta: none; V. Dilsizian: none; A. Quercioli: none; T.D. Ruddy: has received grant support from Nordion Inc. (unrestricted Investigator-initiated research grant); GE Healthcare (unrestricted Investigator-initiated research grant); and AstraZeneca (unrestricted Investigator-initiated research grant); T.H. Schindler: none.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Nuclear Cardiology
Rights and permissions
About this article
Cite this article
Valenta, I., Dilsizian, V., Quercioli, A. et al. Quantitative PET/CT Measures of Myocardial Flow Reserve and Atherosclerosis for Cardiac Risk Assessment and Predicting Adverse Patient Outcomes. Curr Cardiol Rep 15, 344 (2013). https://doi.org/10.1007/s11886-012-0344-0
Published:
DOI: https://doi.org/10.1007/s11886-012-0344-0