Abstract
Spinal dural arteriovenous fistulas (SDAVF) are a rare pathologic entity with a diverse and often misleading clinical presentation. While digital subtraction spinal angiography remains the gold standard, recent advances in noninvasive vascular imaging have improved the diagnosis of SDAVF. As this condition can result in permanent spinal cord injury, all patients require treatment, which consists of surgical or endovascular occlusion of the fistula. Failure to recognize and treat SDAVF in a timely fashion can result in irreversible neurologic disability, including myelopathy, lower extremity weakness and bowel, bladder and sexual dysfunction. This article reviews the clinical features, pathogenesis, radiographic features and current treatment strategies for these complex lesions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction and Clinical Manifestations
Even though spinal dural arteriovenous fistulas (SDAVFs) account for the majority of spinal vascular malformations (~70 %) [1], they remain relatively under-diagnosed [2]. In contrast to spinal cord arteriovenous malformations (AVM), SDAVFs are acquired lesions and are rarely seen in young patients. Possible etiologies/predisposing factors include thrombosis of the extradural spinal veins and trauma. SDAVFs have an overwhelmingly male predominance (80 %), with an age presentation in the fifth or sixth decade. Only 1 % of patients present younger then 30 years of age. Most SDAVFs are solitary lesions arising from the lower thoracic and upper lumbar vertebral segmental arteries between T6 and L2. The highest documented level in a consecutive case series was at C7 [3], and sacral lesions have been seen in 4 % of patients [5]. Several studies have documented a predominance of lesions on the left side [3, 4].
A constellation of nonspecific symptomatology mimicking more common etiologies (degenerative disc disease, spinal stenosis, peripheral neuropathy) makes the diagnosis of SDAVFs particularly challenging. In a retrospective review of 326 patients, Wang et al. found a misdiagnosis of spinal DAVF in 265 patients, the most common being degenerative disc disease in 120 patients, leading to mistreatment in 62 patients prior to the diagnosis of SDAVF [6•]. Presenting symptoms include a combination of lower extremity weakness commonly exacerbated by exercise, gait disturbances, sensory symptoms (mostly paresthesias, although pain, patchy sensory loss and hyperesthesia are also possible) and bowel/bladder disturbances. The upper extremities are not frequently affected. Radiculopathies and low back pain can also be seen. Sexual dysfunction, bowel and bladder incontinence and urinary retention are usually indicative of an advanced disease process. In a recent review of 78 patients, Kirsch et al. found that all patients presented with neurologic deficits secondary to progressive myelopathy. Gait disturbance was observed in 96 % of patients, lower extremity sensory deficits in 77 % and 59 % of patients exhibited some degree of urinary dysfunction [7••]. Symptoms are typically progressive with an insidious development of disability following initial symptoms over a period of 6 months to 2 years, although rapid deterioration has also been reported [10]. Spontaneous recovery is almost never encountered and an estimated 50 % of untreated patients will be severely disabled within 3 years of the onset of lower extremity weakness [9]. The time between the onset of symptoms and diagnosis has been reported to be between 12 and 44 months, with a mean duration of 22.9 months [11, 12], although recent advances in spinal magnetic resonance imaging (MRI)/ magnetic resonance angiogram (MRA) have been instrumental in expediting the time to diagnosis. The progressive, chronic clinical symptomatology is in marked contrast to the acute presentation of a spinal cord AVM that typically presents with hemorrhage [13].
Vascular Anatomy and Pathophysiology
The pathologic characteristics of a SDAVF consist of an acquired low-flow arteriovenous shunt between a dural artery and the (peri)medullary venous system. The shunt is predominantly located in the intervertebral foramen within the dura. The resulting venous hypertension and congestion accounts for the myelopathy predominantly seen in patients. Arterial supply is provided via a radiculomeningeal branch of the corresponding segmental radicular artery (thoracic intercostal, lumbar radicular, and branches of the vertebral, deep cervical and ascending cervical arteries) that supplies the dura at every level with venous drainage via a radiculomedullary vein (almost always dorsal to the cord) into the perimedullary venous system, which coalesces with normal spinal cord venous drainage in a retrograde fashion [14, 15]. Arterial supply is distinct from radiculomedullary arteries, which exist at inconsistent levels and supply the anterior and posterior spinal arteries that perfuse the spinal cord. Arterial steal is excluded as an associated mechanism, since the fistula is supplied by meningeal branches that do not perfuse the spinal cord. Slow and extensive venous drainage characterizes the SDAVF, with venous drainage extending up to the cervical spine and cranial fossa in an ascending fashion, and the veins of the cauda equina in a descending fashion [16]. There is often a paucity of radiculomedullary veins draining the spinal cord flowing to the epidural space, and SDAVF is often associated with thrombosis of radiculomedullary and epidural veins. This explains why the low-flow arteriovenous shunt induces venous hypertension in SDAVFs while in congenital high-flow AVM, venous hypertension has less of a role in the pathophysiologic mechanism. The pressure in the vein draining the SDAVF rises to 74 % of the mean arterial pressure with resultant venous hypertension and decreased venous drainage of the spinal cord parenchyma and venous congestion [17]. Venous hypertension is responsive to the mean arterial pressure, which explains worsening symptoms during exercise [14].
Angiographic demonstration of venous hypertension can be seen with selective angiography of the artery of Adamkiewicz, which demonstrates severe prolongation of the venous phase [1]. Secondary to the valveless venous system, increased perimedullary venous pressure is transmitted to the intrinsic veins of the spinal cord. This results in ‘arterialization’ of these veins consisting of hypertrophies, tortuous walls, a lower arteriovenous pressure gradient and resultant decreased spinal cord tissue perfusion, and increased edema, hypoxia and blood-cord barrier disruption [14, 18, 19]. These pathologic changes, when resulting in irreversible necrotizing myelopathy of the spinal cord, are referred to as the Foix-Alajouanine syndrome [20].
The slow-flow nature of these lesions makes hemorrhage exceedingly rare, although it has been reported in the thoracolumbar region [9]. Cervicomedullary DAVFs have a much higher reported incidence of hemorrhage as the presenting symptom [8, 21–24]. The hemorrhage seen with these higher lesions can be spinal or intracranial when there is sufficient intracranial venous reflux [25]. A meta-analysis of cervical SDAVF, including those with intracranial involvement, found the rate of subarachnoid hemorrhage to be as high as 45 % [8], while it is an extremely rare presentation of thoracolumbar SDAVFs [26].
Imaging
MRI remains the primary modality for the evaluation of patients with myelopathy and lower extremity sensorimotor deficits, and the neuroradiologist often first raises the possibility of SDAVF. Spinal cord edema, manifested as hyperintensity on T2-weighted MRI images, has been found to be nearly 100 % sensitive for SDAVF [27], but this nonspecific finding can have numerous etiologies including demyelination, trauma, and spinal canal stenosis secondary to degenerative disease of the spine [27, 28]. The presence of dilated intradural veins of the spinal cord is a more specific finding, and can be seen as flow voids on T2-weighted imaging or as serpentine enhancing structures on T1-wieghted contrast-enhanced images. The dilated veins are usually in the dorsal surface of the spinal cord, but can be located ventrally in cervical SADVF. The results of early studies suggest that 50 % of patients with SDAVF may not show abnormal spinal vasculature on conventional MRI [29•, 30]. Enhancement of the dilated perimedullary veins, the spinal cord, or both has been reported in as many as 88 % of patients. Spinal cord appearance can range from edematous and enlarged to gliotic and atrophic. This appearance is typically related to the stage of venous hypertension [31]. Multiple studies have been conflicted on the correlation between resolution of the abnormal intramedullary T2 signal following treatment and clinical improvement, with some studies reporting a positive correlation [32]. Muralidharan et al. reports no correlation between improvement in MRI findings and clinical improvement [29•]. These findings may support the theory of ischemia secondary to venous hypertension as the underlying pathophysiology of the symptoms in SDAVF patients. Alternatively, the lack of T2 signal hyperintensity on MRI suggests a high likelihood of a negative spinal angiogram. Intramedullary T2 signal change is considered a stronger predictor of an abnormal spinal angiogram when compared to dilated perimedullary vessels on CT myelography, because nearly all patients with a negative angiogram had evidence of dilated vessels on myelography [33]. It should be noted, however, that turbulent flow of cerebrospinal fluid on T2-weighted sequences may be mistaken for abnormal spinal vasculature. Wang et al. confirmed earlier reports that intramedullary T2 weighted signal cord hyperintensity and perimedullary dilated vessels were two major changes observed in 87 % and 77 % of patients with SDAVF, respectively. However, these imaging changes were often not correlated with the location of the fistula in approximately half of the cases [6•].
MR Angiography (MRA) has emerged as an extremely useful diagnostic adjunct for the patient with a possible SDAVF by aiding in the visualization of the abnormally enlarged perimedullary veins resulting from the fistula over many spinal levels. Furthermore, it provides specific information regarding the vertebral level of the fistula and course of medullary venous drainage, including size, number and tortuosity. Sensitivity and specificity of MRA in the diagnosis of SDAVF was 91 % and 78 %, respectively, in a series of 31 patients by Bowen et al. [34]. Noninvasive localization of the fistula has also been shown to decrease the iodinated contrast load during the subsequent spinal angiogram by greater than 50 % when compared to the amount of contrast utilized in MRA negative cases [35]. MRA has also proven useful in preoperative planning for endovascular or surgical procedures and for following patients post-treatment [34, 36]. A recent case report of four patients with SDAVF confirmed with angiography demonstrated T2 prolongation on conventional MRI, without evidence of abnormal perimedullary veins. Dynamic gadolinium enhanced MRA, however, was successful at showing abnormal spinal vasculature in all four patients [28]. This supports the role of MRA in patients with abnormal spinal cord signal and symptoms suggestive of SDAVF, despite the lack of other typical MRI findings. Recent advances in multi-detector computed tomography angiography (MDCTA) have also led to the detection of abnormal perimedullary veins and an accurate identification of the fistula level in 73 % of patients. Interestingly, in this same study, CTA and MRA were also useful for detecting a second fistula in two patients, that otherwise may have been missed initially [6•, 37–39]. It is important for the practitioner to consider the diagnosis of SDAVF in the presence of spinal cord edema on MRI in the appropriate clinical context. In this situation, CTA and MRA can be useful for SDAVF diagnosis and may provide compelling evidence to proceed to invasive angiography.
Spinal Angiography
Catheter spinal angiography remains the gold standard for the diagnosis and classification of spinal arteriovenous lesions [40–42]. Spinal angiography can be performed with moderate sedation and local anesthetic, or under general anesthesia. The latter allows for better quality images, since the patient remains still and the use of intermittent apnea can help to eliminate motion artifact. Angiography is performed via a 5-French sheath placed in the common femoral artery and selective catheterization of each individual thoracic and lumbar segmental artery. In addition, catheterization of the bilateral vertebral, deep cervical and ascending cervical arteries must be evaluated for cervical cord involvement and the internal iliac and iliolumbar arteries should be catheterized to fully evaluate the lumbosacral region. Risks involved with catheter angiography include the invasive nature of the exam and the use of iodinated contrast with the potential for nephrotoxicity, but these are generally minimal with experienced interventionalists. Evaluation for SDAVF can be difficult, long and tedious, and therefore should be reserved for experienced practitioners. Spinal angiography must be performed at every level between the skull base and sacrum if the lesion is not identified initially. As in most spinal angiography involving the thoracolumbar region, localization of the artery of Adamkiewicz is recommended, with a thorough evaluation of venous drainage following injection in cases of possible SDAVF. Severe venous hypertension and myelopathy often result in delayed or absent venous drainage. Finally, when evaluating a patient with evidence of venous hypertension, the underlying etiology—usually a SDAVF—must be identified. In cases involving venous hypertension of the cervical cord, a negative complete spinal angiogram warrants a diagnostic cerebral angiogram, given the risk for an intracranial SDAVF draining into the veins of the brainstem and the spinal cord, also known as type V intracranial dural arteriovenous fistula [43–45].
Spinal Dural Arteriovenous Fistula Management
Following diagnosis, expedient treatment and an interdisciplinary approach is required. Neurologic status on initial presentation significantly influences clinical outcomes with SDAVF, but the time to diagnosis has not been shown to have any direct correlation in many studies [28, 46]. Still, clinical status is known to gradually worsen from the onset of symptoms so expedient diagnosis and treatment is essential [3]. The modality of treatment can include either microsurgery or endovascular embolization. The ultimate treatment goal of either modality is the permanent occlusion/obliteration of the arterialized draining vein at the fistulous connection in an attempt to halt and hopefully reverse the damaging effects of venous hypertension. Failure to obtain adequate occlusion of the drainage vein results in a high likelihood of recanalization of the fistula, even if the arterial inflow is occluded [14].
Endovascular treatment involves placement of a microcatheter in the distal radiculomeningeal artery, as close as possible to the fistulous connection between the artery and draining vein; followed by the injection of embolic material. Endovascular treatment has the advantage of being less invasive, can be attempted in the same setting as angiography, and has been shown to have outcomes that are comparable to surgical treatment in recent years [28]. Historically, two problems have plagued endovascular treatment, including limitations due to vascular anatomy and the risk of recanalization. Safe embolization is severely limited in the presence of a common origin of the radiculomedullary artery of Adamkiewicz from the same segmental artery as the radiculomeningeal artery feeding the SDAVF (Fig. 1) [47]. These patients face the risk of inadvertent embolization of the anterior spinal artery and development of anterior spinal artery syndrome, and therefore surgical treatment is the preferred modality. Endovascular treatment is also limited by the ability to properly maneuver the microcatheter due to vessel tortuosity or stenosis, impairing the delivery of embolic material, but advances in the design of microcatheters and micro-guidewires have improved the ability to navigate despite difficult anatomy.
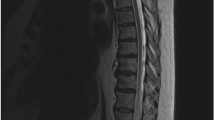
70-year-old female with spinal dural arteriovenous fistula arising from the right L1 radiculomeningeal artery. The early draining vein (arrow) and dilated, tortuous, perimedullary veins (curved arrows) are well visualized. A common origin of the feeding radiculomeningeal artery with the anterior spinal artery (arrowhead) made this patient a more appropriate candidate for surgical occlusion
Endovascular treatment had historically been associated with higher rates of recanalization when compared to surgery. Early reports of endovascular management with polyvinyl alcohol particles showed high recanalization rates [48], but this embolization technique is rarely used today. With the relatively recent use of liquid embolization material (N-butyl cyanoacrylate [NBCA] or Onyx) and constantly improving techniques, the success rate of endovascular treatment now ranges between 70 and 89 % [12, 49]. Kirsch et al. recently studied 78 patients treated endovascularly with NBCA and showed a 77 % success rate defined as persistent obliteration of the fistula [7••]. Notably, the successfully endovascularly treated patients had similar short-term post treatment outcomes when compared to surgically treated patients, and the patients who failed endovascular treatment all subsequently underwent successful surgical treatment. The relatively noninvasive nature of endovascular treatment and comparable outcomes when compared to surgery, suggest that embolization may be recommended as a primary treatment for those with suitable vascular anatomy (Fig. 2). Surgery can be reserved for refractory cases or patients who are not amenable to embolization due to vessel anatomy [29•].
a 73-year-old male with progressive weakness and decreased sensation of the lower extremities bilaterally. Catheter angiography shows a DAVF supplied by the left L2 segmental artery (circle). The venous drainage is via a radiculomedullary vein that is enlarged and filling in a retrograde fashion (arrows), draining into the anterior spinal vein (arrowheads). b The artery of Adamkiewicz (arrow) originates by the left T10 segmental artery and supplies the anterior spinal artery (arrowheads). c The DAVF was successfully embolized with NBCA without any residual arteriovenous shunt. Post-embolization image shows the radiopaque glue cast (circle) within the fistula and the proximal draining vein
Microsurgical treatment of SDAVF entails interrupting the arterialized vein in the intradural compartment. This requires laminectomy and correlation of preoperative angiography and direct intraoperative findings to identify the level of dural penetration of the fistula. The draining spinal vein is identified and either coagulated with bipolar forceps and cut, or the vein is occluded with an aneurysm clip. Early meta-analysis by Steinmetz et al. there was a 98 % initial success rate for obliteration of SDAVFs [50], which has subsequently been confirmed by more recent studies by Wakao et al. and Repper et al. [51••, 52]. In addition the invasive nature of surgical occlusion, the surgical treatment is less able to identify abnormal shunting when compared to endovascular treatment, which is performed using fluoroscopy.
Both treatment options are safe and have equivalent efficacy in improving neurological deterioration when the fistula and draining vein remain occluded [51••, 53]. As with all inherent disease processes of the spinal cord, postoperative function is intimately related to the patient’s preoperative presentation, regardless of the treatment modality. Clinical status prior to surgery or embolization has been shown to be the most important predictor in patient outcome [7••, 51••]. While partial results can be obtained in patients suffering from severe neurologic impairment, early treatment before advanced disease is essential to maximizing postoperative functional results.
Following successful surgical or endovascular treatment of SDAVFs, approximately 90 % of patients have stabilization or improvement of their symptoms [29•, 50]. Generally, motor dysfunction is more likely to improve following treatment, when compared to sensory and bladder function disturbances. Approximately one half to two-thirds of patients have motor function improvement, whereas one-third of patients report improvement in sensory disturbances [14]. Recent studies have shown that 70–81 % of patients have postoperative improvement in gait function, whereas micturition and sphincter dysfunction are less likely to improve [7••, 54]. Impotence rarely improves and pain may also persist following treatment [55]. Many patients who experience immediate postoperative worsening do so because of the worsening of bladder function. There are multiple possible etiologies including the effect of anesthesia, worsening cord edema, or preexisting prostate hypertrophy that can result in the transient worsening of bladder functioning. A recent study published by Muralidharan et al. involved long-term follow-up of 153 patients following surgical treatment. This study showed that worsening bladder functioning was responsible for 61 % of the patients who showed postoperative deterioration. Muralidharan et al. also showed that patients suffering from exertional claudication preoperatively, absent pinprick level, and early clinical recovery following surgery were associated with better outcomes, and may have greater chances of long-term post-surgical improvement. Notably, the presence of a sensory level was associated with a worse postoperative outcome in this study, possibly suggesting that a sensory level in SDAVF patients may be a marker of irreversible cord damage [29•].
Imaging findings, while necessary to establish a diagnosis of SADVF, have not been useful for prognostication. Pre-treatment and post-treatment MRI findings have not been consistent with functional outcomes [56]. Duration of time from initial symptom onset, location of the fistula, and age of the patient were also not associated with postoperative outcome. Importantly, even advanced neurologic deterioration does not preclude improvement since treatment for SDAVF can be beneficial for patients suffering from severe neurologic symptoms, including paraplegia [57]. Aghakhani et al. reported significant improvement for six patients with paraplegia caused by SDAVF. Six months after surgical treatment, two out of five patients had improvement in preoperative sphincter disturbances, and all six patients had regained their ability to walk [33].
Conclusion
This article represents an overview of spinal dural arteriovenous fistulas (SDAVF) with reference to the clinical manifestations, pathophysiology, imaging characteristics and surgical and endovascular treatment modalities. The clinical presentation includes lower extremity sensory deficits, myelopathy or bowel and bladder dysfunction secondary to spinal venous hypertension. Early recognition and expedient treatment of patients with SDAVF should be emphasized as the natural history of these lesions follows a course of clinical deterioration with the potential for irreversible spinal cord injury. An interdisciplinary approach to the management of SDAVFs with endovascular and surgical specialists is mandatory to achieve the best possible clinical outcomes and the endpoint for effective treatment, either endovascular or surgical, is the obliteration and permanent occlusion of the arterialized vein that drains the fistula.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Merland JJ, Richie MC, Chiras J. Intraspinal extramedullary arteriovenous fistulae drinaing into the medullary veins. J Neuroradiol. 1980;7:271–320.
Schaat TJ, Salzman KL, Stevens EA. Sacral origin of a dural arteriovenous fistula: case report and review. Spine. 2002;27:893–7.
Van Dijk JM, TerBrugge KG, Willinsky RA, Farb RI, Wallace MC. Multidisciplinary management of spinal dural arteriovenous fistulas: clinical presentation and long-term follow-up in 49 patients. Stroke. 2002;33(6):1578–83.
Gobin YP, Houdart E, Casasco A, et al. Endovascular therapy for arteriovenous malformations and fistulae in the spinal cord. Semin Interv Radiol. 1993;10:227–42.
Aminoff MJ, Logue V. The prognosis of patients with spinal vascular malformations. Brain. 1974;97:211–8.
• Wang D, Yang N, Zhang P, Xu S, Li X, Zhao P, Huang B, Li X. The diagnosis of spinal dural arteriovenous fistulas. Spine (Phila Pa 1976). 2013 Feb 1. [Epub ahead of print] This article provides insight into radiologic signs for SDAVF and current advances in imaging techniques for diagnosis.
•• Kirsch M, Berg-Dammer E, Musahl C, Bazner H, Kuhne D, Henkes H. Endovascular management of spinal dural arteriovenous fistulas in 78 patients. Neuroradiology 2013 Jan 19. This article provides evidence of equal efficacy of endovascular treatment when compared to surgical treatment, a relatively recent development.
Aviv RI, Shad A, Tomlinson G, et al. Cervical dural arteriovenous fistulae manifesting as subarachnoid hemorrhage: report of two cases and literature review. AJNR Am J Neuroradiol. 2004;25:854–8.
Koch C, Gottschalk S, Giese A. Dural arteriovenous fistula of the lumbar spine presenting with subarachnoid hemorrhage. Case report and review of the literature. J Neurosurg. 2004;100(4 Suppl Spine):385–91.
Willinsky R, TerBrugge K, Lasjaunias P, et al. The variable presentations of craniocervical and cervical dural arteriovenous malformations. Surg Neurol. 1990;34:118–23.
Jellema K, Tijssen CC, van Gijn J. Spinal dural arteriorvenous fistulae: clinical features and long-term results. Neurosurgery. 2008;62:159–66.
Narvid J, Hetts SW, Larsen D, Neuhaus J, Singh TP, McSwain H, et al. Spinal dural arteriovenous fistulae: clinical features and long-term results. Neurosurgery. 2008;62(1):159–66.
Gobin YP. Classification and endovascular treatment of spinal cord arteriovenous malformations and fistulas. J Stroke Cerebrovasc Dis. 1997;6:282–6.
Patsalides A, Santillan A, Knopman J, Tsiouris AJ, Riina HA, Gobin YP. Endovascular management of spinal dural arteriovenous fistulas. J NeuroIntervent Surg. 2011;3:80–4.
Krings T. Vascular malformations of the spine and spinal cord: anatomy, classification, treatment. Klin Neuroradiol. 2010;20:5–24.
Kai Y, Hamada J, Morioka M, et al. Arteriovenous fistulas at the cervicomedullary junction presenting with subarachnoid hemorrhage: six case reports with special reference to the angiographic pattern of venous drainage. AJNR Am J Neuroradiol. 2005;26:1949–54.
Hassler W, Thron A, Grote EH. Hemodynamics of spinal dural arteriovenous fistulas. An intraoperative study. J Neurosurg. 1989;70:360–70.
Hurst RW, Kenyon LC, Lavi E, et al. Spinal dural arteriovenous fistula: the pathology of venous hypertensive myelopathy. Neurology. 1995;45:1309–13.
Jellema K, Tijssen CC, van Gijn J. Spinal dural arteriovenous fistulas: a congestive myelopathy that initially mimics a peripheral nerve disorder. Brain. 2006;129:3150–64.
Foix C, Alajouanine T. La myélite nécrotique subaguie. Rev Neurol. 1926;2:1–42.
Do HM, Jensen ME, Cloft HJ, et al. Dural arteriovenous fistula of the cervical spine presenting with subarachnoid hemorrhage. AJNR Am J Neuroradiol. 1999;20:348–50.
Fassett DR, Rammos SK, Patel P, et al. Intracranial subarachnoid hemorrhage resulting from cervical spine dural arteriovenous fistulas: literature review and case presentation. Neurosurg Focus. 2009;26:E4.
Guo LM, Zhou HY, Xu JW, et al. Dural arteriovenous fistula at the foramen magnum presenting with subarachnoid hemorrhage: case reports and literature review. Eur J Neurol. 2010;17:684–91.
Takahashi K, Hayashi S, Ootani T, et al. Dural arteriovenous fistula manifesting as subarachnoid hemorrhage at the craniocervical junction. A case report. No Shinkei Geka. 2008;36:901–6.
Houdart E, Gobin YP, Casasco A, et al. A proposed angiographic classification of intracranial arteriovenous fistulae and malformations. Neuroradiology. 1993;35:381–5.
Mascalchi M, Mangiafico S, Marin E. Hematomyelia complicating a spinal dural arteriovenous fistula. Report of a case. J Neuroradiol. 1998;25:140–3.
Gilbertson JR, Miller GM, Goldman MS, et al. Spinal dural arteriovenous fistulas: MR and myelographic findings. AJNR Am J Neuroradiol. 1995;16:2049–57.
Da Costa L, Dehdashti AR, TerBrugge KG. Spinal cord vascular shunts: spinal cord vascular malformations and dural arteriovenous fistulas. Neurosurg Focus. 2009;26(1):E6.
• Muralidharan R, Saladino A, Lanzino G, Atkinson JL, Rabinstein AA. The clinical and radiological presentation of spinal dural arteriovenous fistula. Spine (Phila Pa 1976). 2011;36(25):E1641–7. This article provides useful and current information on post-treatment outcomes and identifies possible useful prognostic factors, including absence of sensory level and presence of exertional claudication.
Miller TR, Eskey CJ, Mamourian AC. Absence of abnormal vessels in the subarachnoid space on conventional magnetic resonance imaging in patients with spinal dural arteriovenous fistulas. Neurosurg Focus. 2012;32(5):E15.
Niimi Y, Berenstein A, Setton A, et al. Embolization of spinal dural arteriovenous fistulae: results and follow-up. Neurosurgery. 1997;40:675–82. discussion 682–3.
Song JK, Gobin YP, Duckwiler GR, et al. N-butyl 2-cyanoacrylate embolization of spinal dural arteriovenous fistulae. AJNR Am J Neuroradiol. 2001;22:40–7.
Aghakhani N, Parker F, David P, et al. Curable cause of paraplegia: spinal dural arteriovenous fistulae. Stroke. 2008;39:2756–9.
Bowen BC, Fraser K, Kochan JP, et al. Spinal dural arteriovenous fistulas: evaluation with MR angiography. AJNR Am J Neuroradiol. 1995;16:2029–43.
Luetmer PH, Lane JI, Gilbertson JR, et al. Preangiographic evaluation of spinal dural arteriovenous fistulas with elliptic centric contrast-enhanced MR angiography and effect on radiation dose and volume of iodinated contrast material. AJNR Am J Neuroradiol. 2005;26:711–8.
Binkert CA, Kollias SS, Valavanis A. Spinal cord vascular disease: characterization with fast three-dimensional contrast-enhanced MR angiography. AJNR Am J Neuroradiol. 1999;20:1785–93.
Yamaguchi S, Nagayama T, Eguchi K, Takeda M, Arita K, Kurisu K. Accuracy and pitfalls of multidetector-row computed tomography in detecting spinal dural arteriovenous fistulas. J Neurosurg Spine. 2010;12(3):243–8.
Osanai T, Hida K, Asano T, et al. The Multi-detector-row CT angiography for diagnosis of spinal dural arteriovenous fistula. No Shinkei Geka. 2012;40:695–703.
Boeckh-Behrens T, Bitterling H, Schichor C, Bruckmann H, Seelos K. Improved localization of spinal AV fistulas using contrast-enhanced MR angiography at 3T. Rofo. 2010;182(1):53–7.
Krings T, Geibprasert S. Spinal dural arteriovenous fistulas. AJNR Am J Neuroradiol. 2009;30:639–48.
Krings T, Lasjaunias PL, Hans FJ, et al. Imaging in spinal vascular disease. Neuroimaging Clin N Am. 2007;17:57–72.
Jellema K, Tijssen CC, Sluzewski M, et al. Spinal dural arteriovenous fistulas—an underdiagnosed disease. A review of patients admitted to the spinal unit of a rehabilitation center. J Neurol. 2006;253:159–62.
Brunereau L, Gobin YP, Meder JF, et al. Intracranial dural arteriovenous fistulas with spinal venous drainage: relation between clinical presentation and angiographic findings. AJNR Am J Neuroradiol. 1996;17:1549–54.
Gobin YP, Rogopoulos A, Aymard A, et al. Endovascular treatment of intracranial dural arteriovenous fistulas with spinal perimedullary venous drainage. J Neurosurg. 1992;77:718–23.
Patsalides A, Tzatha E, Stubgen JP, et al. Intracranial dural arteriovenous fistula presenting as an enhancing lesion of the medulla. J Neurointerv Srg. 2010;2(4):390–3.
Cenzato M, Debernadi A, Stefini R, D’Aliberti G, Piparo M, Talamonti G, et al. Spinal dural arteriovenous fistulas: outcome and prognostic factors. Neurosurg Focus. 2012;32(5):E11.
Ruiz-Juretschke F, Perez-Calvo JM, Castro E, Garcia-Leal R, Mateo-Sierra O, Fortea F, et al. A single-center, long-term study of spinal dural arteriovenous fistulas with multidisciplinary treatment. J Clin Neurosci. 2011;18(12):1662–8.
Hall WA, Oldfield EH, Doppman JL. Recanalization of spinal arteriovenous malformations following embolization. J Neurosurg. 1989;70(5):714–20.
Andres RH, Barth A, Guzman R, Remonda L, El-Koussy M, Seiler RW, et al. Endovascular and surgical treatment of spinal dural arteriovenous fistulas. Neuroradiology. 2008;50(10):869–76.
Steinmetz MP, Chow MM, Krishnaney AA, Andrews-Hinders D, Benzel EC, Masaryk TJ, et al. Outcome after the treatment of spinal dural arteriovenous fistulae: a contemporary single-institution series and meta-analysis. Neurosurgery. 2004;55:77–88.
•• Wakao N, Imagama S, Ito Z, Ando K, Hirano K, Tauchi R, et al. Clinical outcome of treatments for spinal dural arteriovenous fistulas: results of multivariate analysis and review of the literature. Spine. 2012;37(6):482–8. This meta-analysis provides evidence of equal efficacy of endovascular treatment when compared to surgical treatment, a relatively recent development.
Ropper AE, Gross BA, Du R. Surgical treatment of Type I spinal dural arteriovenous fistulas. Neurosurg Focus. 2012;32(5):E3.
Sivakumar W, Zada G, Yashar P, Giannotta SL, Teitelbaum G, Larsen DW. Endovascular management of spinal dural arteriovenous fistulas. A review. Neurosurg Focus. 2009;26(5):E15.
Sherif C, Gruber A, Bavinzski G, Standhardt H, Widhalm G, Gibson D, et al. Long-term outcome of a multidisciplinary concept of spinal dural arteriovenous fistulae treatment. Neuroradiology. 2008;50(1):67–74.
Shinoyama M, Endo T, Takahash T, Shimizu H, Takahashi A, Suzuki M, et al. Long-term outcome of cervical and thoracolumbar dural arteriovenous fistulas with emphasis on sensory disturbance and neuropathic pain. World Neurosurg. 2010;73(4):401–8.
Kaufmann TJ, Morris JM, Saladino A, Mandrekar JN, Lanzino G. Magnetic resonance imaging findings in treated spinal dural arteriovenous fistulas: lack of correlation with clinical outcomes. J Neurosurg Spine. 2011;14(4):548–54.
Prieto R, Pascual JM, Gutierrez R, Santos E. Recovery from paraplegia after the treatment of spinal dural arteriovenous fistula: a case report and review of the literature. Acta Neurochir (Wien). 2009;151(11):1385–97.
Conflicts of Interest
Joshua Marcus declares no conflicts of interest.
Justin Schwarz declares that he has no conflicts of interest.
I. Paul Singh declares that he has no conflicts of interest.
Dimitri Sigounas declares that he has no conflicts of interest.
Jared Knopman declares that he has no conflicts of interest.
Y. Pierre Gobin declares that he has no conflicts of interest.
Athos Patsalides declares that he has no conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Cardiovascular Disease and Stroke
Rights and permissions
About this article
Cite this article
Marcus, J., Schwarz, J., Singh, I.P. et al. Spinal Dural Arteriovenous Fistulas: A Review. Curr Atheroscler Rep 15, 335 (2013). https://doi.org/10.1007/s11883-013-0335-7
Published:
DOI: https://doi.org/10.1007/s11883-013-0335-7