Abstract
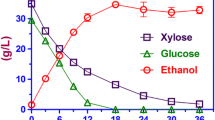
The present investigation was aimed towards pretreatment optimization of corncob to maximize cellulose and hemicellulose recovery, followed by substrate selection for holocellulase production using psychrotolerant Aspergillus niger SH3. Dilute alkali pretreatment (1.5% NaOH) resulted in higher recovery of cellulose (59.66%) and hemicellulose (28.34%) from corncob, while corn stover proved to be the best substrate for holocellulase production. Further, saccharification was optimized by Box-Behnken design to select the suitable conditions for maximum sugar release from pretreated corncob. The optimum conditions for maximum sugar release were 8% (w/v) substrate loading, 11 FPU/gds enzyme loading at temperature 38 °C and pH 3.0 which resulted in 114.5% higher sugar yield (912 mg/gds of pretreated biomass) as compared with un-optimized conditions (425.35 mg/gds). Theoretical yield of 48.8% ethanol was achieved through simultaneous saccharification and fermentation (SSF) using pretreated corncob. This study illustrates the potential of different corn residues as a promising substrate for bioethanol production.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
A. Ahmad, N. M. Yasin, C. Derek and J. Lim, Renew. Sust. Energy Rev., 15, 584 (2011).
E. Warner, Y. Zhang, D. Inman and G. Heath, Biofuels Bioprod. Bioref., 8, 114 (2014).
N. Ravindranath, C. S. Lakshmi, R. Manuvie and P. Balachandra, Energy Policy, 39, 5737 (2011).
S. Mohanram, D. Amat, J. Choudhary, A. Arora and L. Nain, Sustain. Chem. Process, 1, 15 (2013).
K. Byjesh, S. N. Kumar and P. K. Aggarwal, Mitigation and Adaptation Strategies for Global Change, 15, 413 (2010).
India maize summit (2015), [http://ficci.in/events/22310/ISP/Background-paper-India-maize-Summit.pdf].
S. H. Mood, A. H. Golfeshan, M. Tabatabaei, G. S. Jouzani, G.H. Najafi, M. Gholami and M. Ardjmand, Renew. Sust. Energy Rev., 27, 77 (2013).
R. Masran, Z. Zanirun, E.K. Bahrin, M.F. Ibrahim, P.L. Yee and S. Abd-Aziz, Appl. Microbiol. Biot., 100, 5231 (2016).
D. Deswal, R. Gupta, P. Nandal and R. C. Kuhad, Carbohydr. Polym., 99, 264 (2014).
F.B. Chaabane and R. Marchal, Oil. Gas Sci. Technol., 68, 663 (2013).
E. Johnson, Biofuels Bioprod. Bioref., 10, 164 (2016).
P.V. Harris, F. Xu, N. E. Kreel, C. Kang and S. Fukuyama, Curr. Opin. Chem. Biol., 19, 162 (2014).
A.A. Modenbach and S.E. Nokes, Biomass Bioenergy, 56, 526 (2013).
J. Larsen, M.O. Petersen, L. Thirup, H.W. Li and F. K. Iversen, Chem. Eng. Technol., 31, 265 (2008).
D.B. Hodge, M.N. Karim, D. J. Schell and J.D. McMillan, Appl. Biochem. Biotechnol., 152, 88 (2009).
L. Paulova, P. Patakova, M. Rychtera and K. Melzoch, Fuel, 122, 294 (2014).
R. Tiwari, P. K. Nain, S. Singh, A. Adak, M. Saritha, S. Rana, A. Sharma and L. Nain, J. Taiwan Inst. Chem. Eng., 56, 57 (2015).
D.M. Updegraff, Anal. Biochem., 32, 420 (1969).
TAPPI, Technical association of pulp and paper industry, Atlanta, Georgia (1997).
G. S. Rautela and E. B. Cowling, Appl. Microbiol., 14, 892 (1966).
T. Ghose, Pure Appl. Chem., 59, 257 (1987).
T. Ghose and V. S. Bisaria, Pure Appl. Chem., 59, 1739 (1987).
T.M. Wood and K.M. Bhat, Methods Enzymol., 160, 87 (1982).
G. L. Miller, Anal. Chem., 31, 426 (1959).
P. Sahare, R. Singh, R. S. Laxman and M. Rao, Appl. Biochem. Biotechnol., 168, 1806 (2012).
E. van der Pol, R. Bakker, A. van Zeeland, D. S. Garcia, A. Punt and G. Eggink, Bioresour. Technol., 181, 114 (2015).
K.Y. Won, B. H. Um, S.W. Kim and K. K. Oh, Korean J. Chem. Eng., 29, 614 (2012).
A.K. Kumar and B. S. Parikh, Bioresour. Bioprocesss, 2, 1 (2015).
A.U. Buranov and G. Mazza, Ind. Crop. Prod., 28, 237 (2008).
Q. Luo, H. Peng, M. Zhou, D. Lin, R. Ruan, Y. Wan, J. Zhang and Y. Liu, Bioresources, 7, 5817 (2012).
S.L. Sun, J.L. Wen, M.G. Ma and R.C. Sun, Carbohyd Res., 92, 2224 (2013).
A. Teleman, J. Lundqvist, F. Tjerneld, H. Stalbrand and O. Dahlman, Carbohyd. Res., 329, 807 (2000).
T. Juhasz, Z. Szengyel, K. Reczey, M. Siika-Aho and L. Viikari, Process Biochem., 40, 3519 (2005).
R. P. de Vries and J. Visser, Microbiol. Mol. Biol. R., 65, 497 (2001).
D. P. Maurya, S. Vats, S. Rai and S. Negi, Indian J. Exp. Biol., 51, 992 (2013).
J.K. Saini, R. K. Anurag, A. Arya, B. Kumbhar and L. Tewari, Ind. Crops Prod., 44, 211 (2013).
C. Cara, M. Moya, I. Ballesteros, M. J. Negro, A. González and E. Ruiz, Process Biochem., 42, 1003 (2007).
K.D. Ramachandriya, M. Wilkins, H. K. Atiyeh, N.T. Dunford and S. Hiziroglu, Bioresour. Technol., 147, 168 (2013).
P. Li, D. Cai, Z. Luo, P. Qin, C. Chen, Y. Wang, C. Zhang, Z. Wang and T. Tan, Bioresour. Technol., 206, 86 (2016).
S. Priya, R. Tiwari, S. Rana, M. Saritha, S. Singh, A. Arora and L. Nain, Energy Ecol. Environ. (2016), (In press), DOI:10.1007/s40974-016-0021-zM.
M. Chen, Y. Qin, Z. Liu, K. Liu, F. Wang and Y. Qu, Enzyme Microb. Technol., 46, 444 (2010).
S. Singh, K. Pranaw, B. Singh, R. Tiwari and L. Nain, J. Taiwan Inst. Chem. Eng., 45, 2379 (2014).
S. Suwannarangsee and L. Eurwilaichitr, J. Microbiol. Biotechnol., 24, 1427 (2014).
C. Li, B. Knierim, C. Manisseri, R. Arora, H.V. Scheller, M. Auer, K. P. Vogel, B. A. Simmons and S. Singh, Bioresour. Technol., 101, 4900 (2010).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
11814_2016_334_MOESM1_ESM.pdf
Simultaneous saccharification and fermentation of alkali-pretreated corncob under optimized conditions using cold-tolerant indigenous holocellulase
Rights and permissions
About this article
Cite this article
Sharma, A., Nain, V., Tiwari, R. et al. Simultaneous saccharification and fermentation of alkali-pretreated corncob under optimized conditions using cold-tolerant indigenous holocellulase. Korean J. Chem. Eng. 34, 773–780 (2017). https://doi.org/10.1007/s11814-016-0334-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-016-0334-9