Abstract
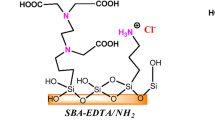
Organofunctionalized nanostructured silica SBA-15 with tri(2-aminoethyl)amine tetradentate-amine ligand was synthesized and applied as adsorbent for the removal of Cu2+, Pb2+, and Cd2+ from both synthetic wastewater and real paper mill and electroplating industrial effluents. The prepared materials were characterized by XRD, N2 adsorption-desorption, TGA, and FT-IR analysis. The Tren-SBA-15 was found to be a fast adsorbent for heavy metal ions from single solution with affinity for Cu2+, Pb2+, than for Cd2+ due to the complicated impacts of metal ion electronegativity. The kinetic rate constant decreased with increasing metal ion concentration due to increasing of ion repulsion force. The equilibrium batch experimental data is well described by the Langmuir isotherm. The maximum adsorption capacity was 1.85 mmol g−1 for Cu2+, 1.34 mmol g−1 for Pb2+, and 1.08 mmol g−1 for Cd2+ at the optimized adsorption conditions (pH=4, T=323 K, t=2 h, C0=3 mmol L−1, and adsorbent dose=1 g L−1). All Gibbs energy was negative as expected for spontaneous interactions, and the positive entropic values from 103.7 to 138.7 J mol−1 K−1 also reinforced this favorable adsorption process in heterogeneous system. Experiment with real wastewaters showed that approximately a half fraction of the total amount of studied metal ions was removed within the first cycle of adsorption. Hence, desorption experiments were performed by 0.3M HCl eluent, and Tren-SBA-15 successfully reused for four adsorption/desorption cycles to complete removal of metal ions from real effluents. The regenerated Tren-SBA-15 displayed almost similar adsorption capacity of Cu2+, Pb2+, and Cd2+ even after four recycles. The results suggest that Tren-SBA-15 is a good candidate as an adsorbent in the removal of Cu2+, Pb2+, and Cd2+ from aqueous solutions.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
F. Fu and Q. Wang, J. Environ. Manage., 92, 407 (2011).
E. Da’na, N. De Silva and A. Sayari, Chem. Eng. J., 166, 454 (2011).
D. Perez-Quintanilla, I. del Hierro, M. Fajardo and I. Sierra, J. Mater. Chem., 16, 1757 (2006).
J. Aguado, J. M. Arsuaga, A. Arencibia, M. Lindo and V. Gascón, J. Hazard. Mater., 163, 213 (2009).
A. Benhamou, M. Baudu, Z. Derriche and J. P. Basly, J. Hazard. Mater., 171, 1001 (2009).
E. Da’na and A. Sayari, Desalination, 285, 62 (2012).
D. Zhao, J. Feng, Q. Huo, N. Melosh, G. H. Fredrickson, B. F. Chmelka and G. D. Stucky, Science, 279, 548 (1998).
A. Walcarius and C. Delacôte, Anal. Chim. Acta, 547, 3 (2005).
E. Da’na, N. De Silva and A. Sayari, Chem. Eng. J., 166, 454 (2011).
M. C. Bruzzoniti, A. Prelle, C. Sarzanini, B. Onida, S. Fiorilli and E. Garrone, J. Sep. Sci., 30, 2414 (2007).
A.M. Burke, J. P. Hanrahan, D. A. Healy, J. R. Sodeau, J. D. Holmes and M. A. Morris, J. Hazard. Mater., 164, 229 (2009).
A. Shahbazi, H. Younesi and A. Badiei, Chem. Eng. J., 168, 505 (2011).
A. Shahbazi, H. Younesi and A. Badiei, Canadian J. Chem. Eng., 91(4), 739 (2013).
A. Badiei, H. Goldooz and G. M. Ziarani, Appl. Surf. Sci., 257, 4912 (2011).
D. Zhao, J. Feng, Q. Huo, N. Melosh, G. H. Fredrickson, B. F. Chmelka and G. D. Stucky, Science, 279, 548 (1998).
A. Badiei, H. Goldooz, G. M. Ziarani and A. Abbasi, J. Colloid Interface Sci., 357, 63 (2011).
D. A. House, Ammonia & N-Donor Ligands, in: Encyclopedia of Inorganic Chemistry, Wiley, New York (2006).
C. H. Giles, D. Smith and A. Huitson, J. Colloid Interface Sci., 47, 755 (1974).
G. Limousin, J. P. Gaudet, L. Charlet, S. Szenknect, V. Barthès and M. Krimissa, Appl. Geochem., 22, 249 (2007).
W. Yantasee, Y. Lin, G. E. Fryxell, K. L. Alford, B. J. Busche and C. D. Johnson, Ind. Eng. Chem. Res., 43, 2759 (2004).
E. R. Nightingale, J. Phys. Chem., 63, 1381 (1959).
R. G. Pearson, Inorg. Chem., 27, 734 (1988).
I. Langmuir, J. Am. Chem. Soc., 40, 1361 (1918).
B. P. Bering, M. M. Dubinin and V.V. Serpinsky, J. Colloid Interface Sci., 38, 185 (1972).
X. Xue and F. Li, Micropor. Mesopor. Mater., 116, 116 (2008).
A. Heidari, H. Younesi and Z. Mehraban, Chem. Eng. J., 153, 70 (2009).
Y. Kim, C. Kim, I. Choi, S. Rengaraj and J. Yi, Environ. Sci. Technol., 38, 924 (2003).
G. Li, Z. Zhao, J. Liu and G. Jiang, J. Hazard. Mater., 192, 277 (2011).
M. R. Awual, I. M. Rahman, T. Yaita, M. A. Khaleque and M. Ferdows, Chem. Eng. J., 236, 100 (2014).
M. E. Argun, S. Dursun, C. Ozdemir and M. Karatas, J. Hazard. Mater., 141, 77 (2007).
Y. Khambhaty, K. Mody, S. Basha and B. Jha, Chem. Eng. J., 145, 489 (2009).
E. Guibal, M. Jansson-Charrier, I. Saucedo and P. L. Cloirec, Langmuir, 11, 591 (1995).
A. Kilislioglu and B. Bilgin, Appl. Radiat. Isot., 58, 155 (2003).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shahbazi, A., Younesi, H. & Badiei, A. Functionalized nanostructured silica by tetradentate-amine chelating ligand as efficient heavy metals adsorbent : Applications to industrial effluent treatment. Korean J. Chem. Eng. 31, 1598–1607 (2014). https://doi.org/10.1007/s11814-014-0089-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-014-0089-0