Abstract
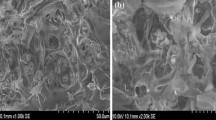
The biosorption of chromium from an aqueous solution onto Erythrina Variegata Orientalis leaf powder was investigated in batch operations. The equilibrium agitation time was 180 min. The extent of chromium biosorption increased from 74.2% to 86.4% with decrease in biosorbent size from 150 to 45 μm for a dosage of 30 g/L. The biosorption decreased from 99.1 (0.45 mg/g) to 45.5% (1.64 mg/g) with an increase in chromium initial concentration (C o ) from 22.5 to 180 mg/L. The extent of biosorption was maximum at pH=3. The experimental data were well explained by Langmuir and Redlich-Peterson isotherm models. The biosorption data followed second-order kinetics with a rate constant of 0.078 g/mg-min for 50 g/L of 45 μm size biosorbent. The biosorption was exothermic and feasible. The biosorption was tending towards irreversibility with increasing temperature.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
B.C. Son, K.M. Park, S. H. Song and Y. J. Yoo, Korean J. Chem. Eng., 21(6), 1168 (2004).
K. Srinivasa Rao, Shashi Anand and P. Venkateswarlu, Korean J. Chem. Eng., 27(5), 1547 (2010).
Web site of Central Pollution Control Board: www.cpcb.nic.in.
E. A. Oliveira, S. F. Montanher, A. D. Andrade, J. A. Nobrega and M. C. Rollemberg, Process Biochem., 40, 3485 (2005).
A. Baran, E. B cak, S. H. Baysal and S. Onal, Bioresour. Technol., 98, 661 (2006).
H. Barrera, F. Urena-Nunez, B. Bilyeu and C. Barrera-Diaz, J. Hazard. Mater., B136, 846 (2006).
S. Ilhan, M. Nurbas, S. Nourbakhsh, Kilicarslan and H. Ozdag, Turkish Electron. J. Biotechnol., 2, 50 (2004).
M. F. Sawalha, J. L. Gardea-Torredey, J.G. Parsons, G. Saupe and J. R. Peralta-Videa, Microchem. J., 81, 122 (2005).
G. Ozdemir, N. Ceyhan, T. Ozturk, F. Akirmak and T. Cosar, Chem. Eng. J., 102, 249 (2004).
P. Suksabye, P. Thiravetyan, W. Nakbanpote and S. Chayabutra, J. Hazard. Mater., 141(3), 637 (2007).
F.N. Acar and E. Malkoc, Bioresour. Technol., 94, 13 (2004).
G. Donmez and Z. Aksu, Process Biochem., 38, 751 (2002).
S. Gupta and B.V. Babu, Adsorption of chromium (VI) by a lowcost adsorbent prepared from tamarind seeds, Paper presented at CHEMCON-2006, India.
M. X. Loukidou, A. I. Zouboulis, T. D. Karapantsios and K. A. Matis, Colloids and Surfaces, A: Physicochem. Eng. Aspects, 242, 93 (2004).
P. Rohinikumar, M. Venkateswara Rao, N. Chittibabu, P.V. Ravikumar and P. Venkateswarlu, Ind. J. Chem. Technol., 16, 308 (2009).
V. K. Garg, R. K. Gupta and R. K. Gupta, Bioresour. Technol., 92, 79 (2004).
V. Sarin and K. K. Pant, Bioresour. Technol., 97, 5 (2006).
E. Malkoc, Y. Nuhoglu and M. Dundar, J. Hazard. Mater., B138, 142 (2006).
S. S. Barala, S. N. Das, P. Rath and G. R. Choudary, Biochem. Eng. J., 34, 69 (2007).
S. Tunali, I. Kiran and T. Akar, Miner. Eng., 18, 681 (2005).
K.G. Bhattacharya and A. Sharma, J. Hazard. Mater., B113, 97 (2004).
G. Xing, S. Zhang, B. Ju and J. Yang, Carbohydr. Polym., 66, 246 (2006).
N. K. Lazaridis and Ch. Charalambous, Water Res., 39, 4385 (2005).
H. Freundlich, Z. Phys. Chem., 57, 387 (1906).
L. Langmuir, J. Am. Chem. Soc., 40, 1361 (1918).
O. Redlich and D. L. Peterson, J. Phys. Chem., 63, 1024 (1959).
S. Lagergren, K. Sven. Vetenskapsakad. Handl, 24, 1 (1898).
B. Kiran, A. Kaushik and C. P. Kaushik, J. Hazard. Mater., 141(3), 662 (2006).
A. Sharma and G. Bhattacharya, J. Hazard. Mater., B125, 102 (2005).
M. Erdem, H. S. Altundogan and F. Taumen, Miner. Eng., 17, 1045 (2004).
E. Sabah, M. Turan and M. S. Celik, Sep. Sci. Technol., 37, 3081 (2005).
M. Kara, H. Yuzer, E. Sabah and M. S. Celik, Water Res., 37, 224 (2003).
T. Karthikeyan, S. Rajagopal and L. R. Mireanda, J. Hazard. Mater., 124(1–3), 192 (2005).
S. S. Baral, S. N. Das and P. Rath, Biochem. Eng. J., 34, 69 (2007).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aditya, G.V.V., Pujitha, B.P., Babu, N.C. et al. Biosorption of chromium onto Erythrina Variegata Orientalis leaf powder. Korean J. Chem. Eng. 29, 64–71 (2012). https://doi.org/10.1007/s11814-011-0139-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-011-0139-9