Abstract
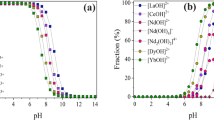
Although much research has been conducted on the separation of single species of heavy metal, the selective adsorption of two or more heavy metals in mixture is relatively little known. In this study, polysaccharide beads were prepared to selectively remove the targeted heavy metal ion from mixture. Among the biomasses, polysaccharide was examined due to its low cost and easy accessibility. In a single metal ion system, chitosan, λ-carrageenan, and alginic acid showed high affinity to mercury, copper, and lead, respectively. In the ion mixture, the same trend shown in the single metal ion solution was observed. The optimum electrolyte concentraion was investigated to adsorb the metal ion selectively, and it was possible to remove the targeted metal ion selectively with chitosan, alginic acid and λ-carrageenan at 1 mmol concentration of electrolyte. In order to demonstrate the feasibility of selective biosorption, two packed-bed reactors in series containing chitosan and alginic acid beads in each were studied and selective adsorption to Hg2+ and Pb2+, respectively, was observed.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Asai, S., Konishi, Y., Tomisaki, H. and Nakanishi, M., “Separation of Mercury from Aqueous Mercuric Chloride Solutions by OnionSkins,”Sep. Sci. Technol.,21, 809 (1986).
Aspinall, O. G., “The Polysaccharides” Academic Press, New York, Vol. 3 (1985).
Bailiey, S. E. and Trudy, J. O., “A Review of Potenially Low-cost Sorbents for Heavy Metals”,Wat. Res.,33, 2469 (1999).
Bedell, G. W. andDarnall,D. W., in “Biosorption of Heavy Metals” (Volesky, B., ed.), CRC Press, pp. 313 (1990).
BrÆk, G S., Malnes, A. and Jensen, A., “Heavy Metal Tolerance of Marine Phytoplankton. IV. Combined Effect of Zinc and Cadmium od Growth and Uptake in Some Marine Diatoms,”J. Exp. Mar. Biol. Ecol.,42, 39 (1980).
Brierly, C. L., “Bioremediation of Metal Contaminated Surface and Groundwaters,”Geomicrobiol. J.,8, 201 (1990).
Chen, J. and Yiacoumi, S., “Biosorption of Metal Ions from Aqueous Solutions”,Sep Sci Technol.,32, 51 (1997).
Gimenez, M. D., Arino, C. and Esteban, M., “Voltammetry of Pb(II), Cd(II) and Zn(II) Ions in the Presence of the Sulphated Polysaccharide λ-Carrageenan,”Analytica Chimica Acta,310, 12 (1995).
Jang, L. K., Nguyen, D. and Geesy, G. G., “An Equilibrium Model for Absorption of Multiple Divalent Metals by Alginate Gel under Acidic Conditions”,Wat. Res.,33(12), 2826 (1999).
Jeon, C., Park, J. Y. and Yoo, Y. J., “Removal of Heavy Metals in Plating Wastewater Using Carboxylated Alginic Acid,”Korean J. Chem. Eng.,18, 955 (2001).
Jeon, C. and Höll, W. H., “Application of the Surface Complexation Model to Heavy Metal Sorption Equilibria onto Aminated Chitosan,”Hydrometallurgy,71, 421 (2004).
Joseph, D. E., “Industrial Wastewater Treatment (A Guide Book)” Lewis Publishers, CRC Press Inc, CRC Press, Inc., New York (1995).
Lovely, D. R. and Covas, J. D., “Bioremediation of Metal Contamination,”Curr. Opin. In Biotechnol.,8, 285 (1997).
Oyrton, A. C., Monteiro, Jr. and Claudio, A., “Some Thermodynamic Data on Copper-Chitin and Copper-Chitosan Biopolymer Interactions,”J. of Colloid Interface Sci.,212, 212 (1999).
Peniche-Covas, C., Alvarez, L. W. and Argüelle-Monal, W., “The Adsorption of Mercuric Ions by Chitosan”,J. ofAppl. Pol. Sci.,46, 1147 (1992).
Singh, S., Pradhan, S. and Rai, L. C., “Comparative Assessment of Fe(III) and Cu(II) Biosorption by Field and Laboratory-grownMicrocystis,”Process Biochem.,33, 495 (1998).
Terry, P. A. and Stone, W., “Biosorption of Cadmium and Copper Contaminated Water byScenedesmus abundans”,Chemosphere,47, 249 (2002).
Ting, Y. P., “Uptake of Cadmium and Zinc by the AlgaChlorella vulgaris: II. Multi-Ion Situation,”Biotechnol. and Bioeng.,37, 445 (1991).
Udaybhaskar, P., Iyengar, L. and Abhakara Rao, A. V. S., “Hexavalent Chromium Interaction with Chitosan,”J. Appl. Polymer Sci.,39, 739 (1990).
Volesky, B., “Biosorbent Materials,”Biotechnol. Bioeng. Symp.,16, 121 (1986).
Volesky, B., (ed.) “Biosorption of Heavy Metals,” CRC Press, Boca Raton, FL, pp. 318 (1990).
Yoshinari, B., Koichi, M. and Yoshinobu, K., “Synthesis of a Chitosan Derivative Recognizing Planar Metal Ion and Its Selective Adsorption Equilibria of Copper(II) over Iron(III),”Reactive Func. Pol.,36, 167 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Son, B.C., Park, K., Song, S.H. et al. Selective biosorption of mixed heavy metal ions using polysaccharides. Korean J. Chem. Eng. 21, 1168–1172 (2004). https://doi.org/10.1007/BF02719489
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02719489