Abstract
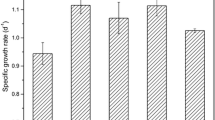
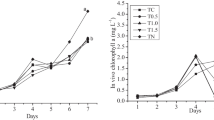
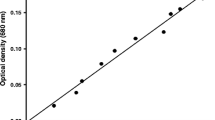
The effects of several nutritional factors on the growth and eicosapentaenoic acid (EPA) production of diatom Nitzschia laevis were studied. 4 LDM (quadrupled concentration of the nutrient salt) was the optimal concentration of nutrient salt for the growth and EPA production of N. laevis. The growth of N. laevis was inhibited when the glucose concentration was either lower than 10 gL−1 or higher than 15 gL−1. Both sodium nitrate and urea were good nitrogen sources for the growth and EPA production, while ammonium chloride seriously decreased the dry cell weight (DW) and the EPA content. Silicate seriously influenced the growth of N. laevis. The maximum DW of 2.34 gL−1 was obtained in the presence of 150 mgL−1 Na2SiO3·9H2O. The EPA content remained almost the same when the silicate concentration was lower than 150 mgL−1; however, higher silicate concentrations resulted in a steady decrease of EPA content. Low medium salinity (⩽29) did not seem to influence the DW of N. laevis, and high salinity resulted in a decrease of DW. The highest EPA content (4.08%) and yield (110 mgL−1) were observed at the salinity of 36 and 29, respectively.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Barclay, W. R., K. M. Meager, and J. R. Abril, 1994. Heterotrophic production of long chain omega-3 fatty acids utilizing algae and algae-like microorganisms. J. Appl. Phycol., 6: 123–129.
Bligh, E, G., and W. J. Dyer, 1959. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol., 37: 911–917.
Feng, F. Y., W. Yang, G. Z. Jiang, Y. N. Xu, and T. Y. Kuang, 2005. Enhancement of fatty acid production of Chlorella sp. (Chlorophyceae) by addition of glucose and sodium thiosulphate to culture medium. Process Biochem., 40: 1315–1318.
Kessell, R. H. J., 1968. Fatty acids of Rhodotorula gracilis: fat production in submerged culture and the particular effect of pH value. J. Appl. Bacteriol., 31: 220–231.
Lebeau, T., and J. M. Robert, 2003a. Diatom cultivation and biotechnology relevant products: Part II. Current and putativeproducts. Appl. Microbiol. Biotechnol., 60: 624–632.
Molina, G. E., E. H. Belarbi, F. F. G. Acién, M. A. Robles, and Y. Chisti, 2003. Recovery of microalgal biomass and metabolites: process options and economics. Biotechnol. Adv., 20: 491–515.
Nettleton, J. A., 1993. Are n-3 fatty acids essential nutrients for fetal and infant development? J. Am. Diet. Assoc., 93(1): 58–64.
Otero, A., D. García, E. D. Morales, J. Arán, and J. Fábregas, 1997. Manipulation of the biochemical composition of the eicosapentaenoic acid-rich microalga Isochrysis galbana in semicontinuous culture. Biotechnol. Appl. Biochem., 26: 171–177.
Ratledge, C., 2001. Microorganisms as sources of polyunsaturated fatty acids. In: Structured and Modified Lipids. Gunstone, F. D., ed., Marcel Dekker, New York, 351–399.
Ratledge, C., and C. T. Evans, 1989. Lipids and their metabolism. In: The Yeasts. 2nd edition. Rose, A. H., and Harrison, J. S., eds., Academic Press, London, 367–455.
Seto, A., H. L. Wang, and C. W. Hesseltine, 1984. Culture conditions affect eicosapentaenoic acid content of Chlorella minutissima. J. Am. Oil Chem. Soc., 61: 892–894.
Starr, R. C., and J. A. Zeikus, 1993. The culture collection of algae at the University of Texas at Austin. J. Phycol. (Suppl.), 29: 90–95.
Tan, C. K., and M. R. Johns, 1996. Screening of diatoms for heterotrophic eicosapentaenoic acid production. J. Appl. Phycol., 8: 59–64.
Wen, Z. Y., and F. Chen, 2000a. Production potential of eicosapentaenoic acid by the diatom Nitzschia laevis. Biotechnol. Lett., 22: 727–733.
Wen, Z. Y., and F. Chen, 2000b. Heterotrophic production of eicosapentaenoic acid by the diatom Nitzschia laevis: effects of silicate and glucose. J. Ind. Microbiol. Biotechnol., 25: 218–224.
Wen, Z. Y., and F. Chen, 2001a. Optimization of nitrogen sources for heterotrophic production of eicosapentaenoic acid by the diatom Nitzschia laevis. Enzyme Microb. Technol., 29: 341–347.
Wen, Z. Y., and F. Chen, 2003. Heterotrophic production of eicosapentaenoic acid by microalgae. Biotechnol. Adv., 21: 273–294.
Werner, D., 1977. The Biology of Diatoms. University of California Press, Berkeley, 250–283.
Xu, X. Q., and J. Beardall, 1997. Effect of salinity on the fatty acid composition of a green microalga from an Antarctic hypersaline lake. Phytochemistry, 45: 655–658.
Yamamura, R., and Y. Shimomura, 1997. Industrial high-performance liquid chromatography purification of docosahexaenoic acid ethyl ester and docosapentaenoic acid ethyl ester from single-cell oil. J. Am. Oil Chem. Soc., 74: 1435–1440.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cao, X., Li, S., Wang, C. et al. Effects of nutritional factors on the growth and heterotrophic eicosapentaenoic acid production of diatom Nitzschia laevis . J. Ocean Univ. China 7, 333–338 (2008). https://doi.org/10.1007/s11802-008-0333-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11802-008-0333-5