Abstract
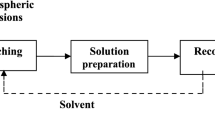
The dissolution kinetics and mechanisms of reaction of Batagbon Kaolin in sulphuric and fluosilicic acids were studied. Leaching temperature, acid concentration, particle size, solid-to-liquid ratio, and stirring speed were selected as process parameters. It is observed that the dissolution rate increases with decreasing particle size and solid-to-liquid ratio, and increases with stirring speed, acid concentration, and leaching temperature. The experimental results indicate that the dissolution rate is of mixed control via hydrogen ion [H+] action, with reaction order of 0.813 and the reaction kinetics can be expressed as K m t=[1-(1-x)1/3+y/6[(1-x)1/3+ 1-2(1-x)2/3]. The activation energy of the process is determined to be 21.6 kJ/mol. The level of the product quality is also evaluated.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
CONSIDINE G D. Van NOSTRAND’S Encyclopedia of Chemistry [M]. London: John Wiley and Sons, Inc., 2005: 251–255.
Industrial Project Report Alum. Aluminium sulphate [EB/OL] [2011–11-28]. http// www.scribd.com/doc.
ISMAIL A K. Energy saving during alum production from kaolin with coproduction of alumina-silica composites from process silica wastes [C]// Proceedings of the XI International Seminar on Mineral Processing Technology. Jamshedpur, India, 2010: 781–787.
FORD K J R. Sulphuric acid and ammonium sulphate leaching of calcined clay [J]. Hydrometallurgy, 1992, 29: 109–130.
PHILIPS C V, WILLS K J. Acid dissolution of alumina from waste aluminium [J]. Hydrometallurgy, 1982, 9: 15–28.
REGINA O A, OKECHUKWU D O. Application of the shrinking core model to the analysis of alumina leaching from ukpor clay using nitric acid [J]. International Journal of Engineering Research & Technology (IJERT), 2012, 3(1): 1–13.
GIRIGISU S. Production of alum from some local kaolin samples: leaching kinetics and process optimization [D]. Department of Chemistry, University of Ilorin, Ilorin, Nigeria. 2013.
FAN A, BROWN R C, WHEELOCK T D, COOKER T A, MOMURA M, ZHUANG Y. Production of a complex coagulant from fly ash [J]. Chemical Engineering Journal, 2005, 106: 269–277.
ADEREMI B O. Kinetics study on aluminium alumination spent acid recovery process [J]. Nigeria Journal of Engineering, 2002, 10(10): 27–34.
BABA A A, MASOBALAJE M A, IBRAHIM A S, GIRIGISU S, ELETTA O A A, ALUKO F I, ADEKOLA F A. Bleaching of a Nigerian kaolin by oxalic acid leaching [J]. Journal of Chemical Technology and Metallurgy, 2015, 50(5): 623–630.
BABA A A, ASALA O A, BALOGUN A F, AYINLA K I, BALE R B, ADEKOLA F A, ALABI A G F. Treatment of a Nigerian kaolin ore for improved industrial applications [J]. Moroccan Journal of Chemistry, 2016, 4(1): 119–127.
BABA A A, ADEKOLA F A, ARODOLA O A, IBRAHIM L, GHOSH M K, SHEIK A R. Simultaneous recovery of total iron and titanium from ilmenite ore by hydrometallurgical processing [J]. Metall Mater Eng, 2012, 18(1): 67–78.
BABA A A, IBRAHIM A S, BALE R B. Purification of a Nigerian talc ore by acid leaching [J]. Applied Clay, 2015, 114: 476–483.
TUMAK S, CIFTILIK A, AKUIL. Factorial experiments for iron removal from kaolin by using single acid two-step leaching with sulphuric acid [J]. Hydrometallurgy, 2013, 134–135: 80–86
SULTANA U K, GULSHAN A S W, KURN Y. Kinetics of leaching of iron oxide in clay in oxalic acid and in hydrochloric acid solutions [J]. Materials Science and Metallurgy Engineering, 2014, 2(1): 5–10.
LAWRENCE C, EDOMWONYI O, BENJAMIN O A. Comparative study of alum production from Nigeria kaolin clay [J]. Materials Science and Technology Conference and Exhibition, 2011: 16–20.
AL-ZAHRAN A A, ABDUL-MAJID M H. Extraction of alumina from clays by hydrochloric acid process [J]. JKAU: Eng Sci, 2009, 20(2): 29–41.
DEGER U, MUSTAFA. Dissolution kinetics of iron and aluminium from red mud in sulphuric acid solution [J]. Indian Journal of Chemical Technology, 2007, 14(3): 263–268.
LAPIDUS G, de LOURDES M M. The effect of product solubility on the leaching kinetics of non-porous minerals [J]. Hydrometallurgy, 1988, 20: 49–64.
AYDOGAN S, ARAS A, CANBAZOGLU M. Dissolution kinetics of sphalerite in acidic Ferric chloride leaching [J]. Chemical Engineering, 2005, 114: 67–72.
HABASHI F. Principles of Extractive Metallurgy [M]. New York: Gordon & Breach, 1980: 107–153.
RAY H S. Kinetics of Metallurgical Reactions [M]. New Delhi: Oxford and IBH Publishing, 1993: 23–41.
GÖKHAN U. Kinetics of sphalerite dissolution by sodium chlorate in hydrochloric acid [J]. Hydrometallurgy, 2005, 95: 39–43.
BABA A A, ADEKOLA A F, BALE R B. Development of a combined pyro-and hydro-metallurgical route to treat spent zinc-carbon batteries [J]. Journal of Hazardous Materials, 2009, 171: 838–84.
BABA A A, ADEKOLA F A. Hydrometallurgical processing of a Nigerian sphalerite in hydrochloric acid: Characterization and dissolution kinetics [J]. Hydrometallurgy, 2010, 101: 69–75.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Adekola, F.A., Baba, A.A. & Girigisu, S. Dissolution kinetics of kaolin mineral in acidic media for predicting optimal condition for alum production. J. Cent. South Univ. 24, 318–324 (2017). https://doi.org/10.1007/s11771-017-3433-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11771-017-3433-9