Abstract
Purpose
Physical activity is a well-established strategy to alleviate breast cancer-related adverse outcomes. To optimise health benefits, behaviour change theories provide frameworks to support women in improving their physical activity. This review aimed to evaluate (i) the effects of behaviour change theory-based physical activity interventions for women with breast cancer and (ii) the application of these theories.
Methods
Seven online databases were searched. Trials were included if randomised and controlled, involved physical activity interventions ≥ 12 weeks duration, used a behaviour change theory, and participants were < 3 years post-cancer treatment. Risk of bias and theory use were assessed. Data were synthesised narratively and meta-analysed.
Results
Forty articles describing 19 trials were included. Overall risk of bias was moderately high. Post-intervention pooled effect estimates were medium for self-reported (SMD = 0.57) and objectively measured physical activity (SMD = 0.52). Most trials cited the social cognitive theory (n = 10) and transtheoretical model (n = 9). Trials rarely applied theories in their entirety, expounded on behavioural mechanisms, or tailored interventions according to behavioural constructs. The most commonly used types of behavioural techniques were goals and planning (n = 18), shaping of knowledge (n = 18), feedback and monitoring (n = 17), and comparisons of outcomes (n = 17).
Conclusions
The included trials were effective for increasing physical activity in women with breast cancer. Theories were applied using a wide range of approaches and levels of rigour, although shared the use of common behavioural techniques.
Implications for Cancer Survivors
Future research may benefit breast cancer survivors by more comprehensively applying behaviour change theories, emphasising individual patient needs and goals.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Physical activity is a well-researched intervention to alleviate health problems associated with breast cancer and reduce the risk of future comorbidities [1]. Improvements can be made in quality of life, fatigue, and physical function, counteracting many commonly experienced symptoms and also lowering the overall risk of all-cause mortality [2, 3]. In the context of physical activity trials which provide professional instruction and/or facilities, it is standard to endeavour to offset inconveniences that come with participation, for example, by assisting with travel or employing experienced study personnel. Accordingly, the key predictors of adherence to exercise interventions for people with cancer include convenience of participation and travel, the presence of professional supervision, and a high frequency of contact with research staff [4, 5]. A recent systematic review of interventions following adjuvant therapy for breast cancer showed that programs typically lasted 8 to 12 weeks, and many did not follow-up and reassess whether participants continued to be physically active after the conclusion of the intervention [6]. As adaptations due to physical activity require ongoing commitment to be maintained, the long-term effects of such trials, once the provided guidance and facilities are removed, are unclear.
Behaviour change theories have been used in physical activity trials for a wide variety of populations to promote self-management and the continuing adoption of health behaviours. These theories outline numerous interconnected processes and factors that are present when changing behaviour, potentially creating a better intervention through identification of factors that may be of particular relevance [7]. Frequently cited behaviour change theories used in health research include the social cognitive theory [8], transtheoretical model [9], and theory of planned behaviour [10]. These theories share some key behavioural constructs, including: self-efficacy i.e. one’s self-belief in their ability to achieve the behaviour change, barriers and enablers, education around the “how” and “why” of the behaviour change, and external social or environmental influences. Underpinned by these theories are behaviour change techniques, which are practical actions or strategies that facilitate change such as cognitive reframing, consideration of health outcomes, or monitoring and goal setting. Behaviour change theories are particularly relevant to programs for breast cancer survivors, due to both the physical and psychological burden of the disease presenting challenges when trying to lead a healthy lifestyle [11, 12].
Physical activity interventions that use behaviour change theories have been explored in women with breast cancer using a wide range of intervention settings and protocols. A previous systematic review, reporting on physical activity behaviour interventions up to 2013, has provided some insight into how the extent of theory use enhances intervention efficacy, while also highlighting the need for further exploration [13]. Hence, this systematic review sought to investigate the benefits of using theory-based physical activity interventions for women with breast cancer by synthesising and evaluating (i) the effects on physical activity and related health outcomes and (ii) the application of behaviour change theories.
Methods
Inclusion criteria and database search
This review was registered with the PROSPERO Register of Systematic Reviews (CRD42019121782). Trials were included if they (1) were a peer-reviewed journal article in English; (2) used a randomised or quasi-randomised study design with a control group; (3) involved women with breast cancer; (4) enrolled women in whom the majority were within three years of their last active treatment (i.e. not including hormone therapy); (5) reported an intervention targeting physical activity, with a minimum duration of 12 weeks; and (5) reported using a behaviour change theory. The criteria for types of interventions were broad and could range from unsupervised lifestyle activity programs to centre-based structured exercise. Multimodal interventions involving health behaviours in addition to physical activity were not included. For studies that reported multiple intervention arms, only arms that met the review criteria were included.
Database search
A search was conducted for published articles in MEDLINE, EMBASE, PsycINFO, the Cochrane Library, CINAHL, Web of Science, and Scopus online databases. Search strategies (Supplementary Material 1) encompassing the inclusion criteria were formulated with the assistance of an academic librarian and cross-referenced with a previous Cochrane review relating to physical activity for women with breast cancer [6]. As records may describe a behaviour change theory in its full text but not in the study title, abstract, or keywords, the search strategies did not include this criterion; instead, use of theory was screened for during the review of full texts. There were no restrictions on publication date. References of the final included papers were also searched. The initial search was undertaken in October 2018, with follow-up searches completed in November 2020 and July 2021.
Selected outcomes
The primary outcome for this review was change in physical activity, measured with questionnaires or objective activity trackers. Secondary outcomes included quality of life, fatigue, and behaviour change variables (e.g., self-efficacy or social support) measured using questionnaires, and physical function, aerobic fitness, and muscular strength.
Trial screening and data extraction
All authors were involved in screening, full text review and data extraction, with each step completed independently by two authors and disagreements resolved by a third author. For publications in which the title and abstract appeared to meet the review criteria, the full text was then reviewed. Study characteristics extracted included aims, eligibility criteria, recruitment methods, intervention and control group details, adherence, and outcome measurement methods. Immediate post-intervention and follow-up (> 12 weeks post-intervention) values were extracted for the selected outcomes, along with information about intervention adherence, such as session attendance. For trials with multiple publications, we collated information across all articles and used the primary article (i.e. with the most complete, relevant outcomes for the review) as the reference article in this review.
Assessment of risk of bias
The revised Cochrane Risk of Bias 2 (RoB 2) was used to assess study risk of bias [14]. The tool covers five domains in which bias may exist: (1) randomisation process, (2) deviations from the intended intervention, (3) missing outcome data (> 5% missing for continuous outcomes), (4) the measurement of outcomes (physical activity), and (5) the selection of the reported results. The tool provides decision tree algorithms that suggest a judgement of either “low risk,” “some concerns,” or “high risk” for each of the 5 domains, and for the overall risk of bias. Risk of bias was assessed independently then discussed by two authors (ML, SK or JY).
Assessment of theory use
To evaluate the extent of behaviour change theory application, items 1–11 of Michie and Prestwich’s Theory Coding Scheme were used [15]. The remaining items were not assessed as they address methodological issues rather than the application of a behaviour change theory. Items 1–6 assess whether a specific behaviour change theory was mentioned, if the intervention was based on a single theory, and if a theory construct was used to inform inclusion criteria (e.g. recruiting low-motivation participants assessed using a theory-based questionnaire), intervention development, and/or the tailoring of the intervention to participants. Items 7–11 assess the link between intervention techniques and theory constructs or other behavioural predictors. The Theory Coding Scheme was scored by two authors (ML and JY).
In addition, the Behaviour Change Technique Taxonomy [16] was used as a means to systematically identify and categorise behaviour change-related intervention components.
Statistical analysis
All extracted continuous outcomes assessed in at least two studies were considered for meta-analyses. Analyses were performed on post-intervention and follow-up means and standard deviations (SD), with the most distal follow-up values selected to reflect the lasting effects of trials. For trials that did not report means or SDs, effect or variability values were imputed from the available data when feasible [17]. Cochrane’s guidelines [18] were followed when deciding what outcome data was appropriate for meta-analysis.
All analyses were carried out using Review Manager (v5.4). Outcomes were combined using the inverse variance random-effects method because, in addition to the possibility of random error, differences were expected from the variation in intervention protocols and population characteristics between the trials (e.g. cancer stage and treatments) [19]. Pooled effect estimates and the corresponding 95% confidence intervals (CI) for intervention versus control arms were calculated for each outcome. Effect size estimates were calculated as standardised mean differences (SMD) to allow synthesis of different measurement methods assessing one outcome (e.g. different self-reported physical activity questionnaires).
To examine the effect of different theories, subgroup analysis was conducted to compare trials according to the theory used. Sensitivity analyses were also performed to discern whether high risk of bias trials or outcomes that required data imputation significantly changed the pooled effect estimates or statistical heterogeneity [20].
Results
Search results
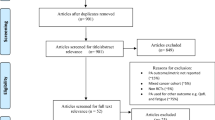
A total of 13,288 records were identified across the database searches (Fig. 1). After removing duplicates, 6593 titles and abstracts were screened, with 485 full texts proceeding to further review. We requested additional data via email from the corresponding authors of four studies; two authors responded, one confirming eligibility and one confirming ineligibility. From the evaluation of the full texts, 40 articles including protocols were identified as being suitable for inclusion in this review, reporting on 19 unique trials.
Participant characteristics
Details on participant characteristics across the included trials are presented in Table 1. Sixteen trials reported average age of participants with an overall mean age of 52.7 years, with one study reporting no age information [21]. Six trials (32%) recruited women with breast cancer across all stages [22,23,24,25,26,27]. The other 13 trials (68%) recruited women with early-stage or non-metastatic cancer only. Fourteen trials reported cancer stage: stage 0 = 4%, stage I = 38%, stage II = 43%, stage III = 15%, and stage IV = 1%. Eight trials (47%) were conducted while participants were undergoing active treatment [23, 26, 28,29,30,31,32,33], with the remaining trials conducted after completion of active treatment (not including hormone therapy). One trial specifically recruited women who were receiving hormone therapy [34]. Twelve trials (63%) limited inclusion into the trial based on participants’ baseline physical activity level with the allowable level varying widely, ranging from < 5 days a week of exercise to reporting no current physical activity.
Intervention and control group characteristics
Details on the interventions are presented in Table 1. Intervention length ranged from 12 weeks to 8 months (mean = 16.5 ± 7.1 weeks). All trials included instruction on home-based physical activity, with six trials also including a centre-based exercise component [28, 30, 32,33,34,35]. Nine trials involved phone calls [23,24,25, 27, 29, 31, 36,37,38] and five trials involved group discussions [22, 30, 32, 33, 35]. Other approaches included a combination of individual counselling sessions and group discussions (n = 2) [28, 34], online/email delivery (n = 2) [21, 27], written material alone (n = 2) [39], and training nurses to engage with participants during treatment visits (n = 1) [26]. All trials that involved regular contact between participants and study personnel reported information about the personnel delivering the intervention: nine trials reported using personnel that had expertise in delivering behaviour change interventions or were trained specifically for the trial [21, 23, 24, 26, 27, 33, 36,37,38], with the remaining trials reporting non-specific information such as research staff titles or allied health profession.
Control groups either received usual care were asked not to change their physical activity level, or were provided with limited breast cancer and physical activity information. Four trials used a contact control group in which control group participants received a comparable frequency of contact but without the primary intervention components (e.g. to administer a symptom questionnaire) [25, 31, 37, 38].
Group allocation and retention
In total, 1966 participants (mean n per study = 103, range = 22–204) were allocated either to an intervention (n = 1054, mean = 55, range = 12–106) or a control group (n = 912, mean per study = 48, range = 10–102). Fifteen trials allocated participants equally between groups, while four trials allocated more participants to the intervention group due to time, financial, and/or recruitment constraints [22, 24, 25, 36]. Post-intervention, 1756 participants were retained, with similar levels of dropout from the intervention (n = 121, 12%) and control arms (n = 89, 10%). In 10 trials (53%), participants were reassessed at follow-up timepoints after the intervention ended, with the final timepoints ranging from 24 weeks to 5 years after baseline (mean = 18 months) (n = 923; intervention groups n = 471, control groups n = 452).
Adherence
Trial adherence was reported in ten trials (53%) and presented in Table 1. Adherence to scheduled sessions was reported in five of the six centre-based trials, with all ≥ 70% [28, 30, 32, 34, 35]. Adherence to home-based physical activity was reported in different formats: three trials reported adherence as the achievement of an activity level goal (ranging from 90 [31, 32] to 150 min a week [26, 29]), and three trials reported the percentage of prescribed sessions completed [24, 31, 36].
Risk of bias
Risk of bias across the included trials is presented in Fig. 2. Notably, 10 trials (53%) were classified as high risk of bias due to insufficient evidence that missing outcome data from participant dropout would not bias the results (Domain 3, > 5% missing for continuous outcomes). Eighteen trials had at least some concern for risk of bias due to having no participant blinding (Domain 4), of which 11 trials (29%) were at high risk due to the nature of the outcomes being likely influenced by knowing the group allocation. Minimal issues were identified relating to randomisation (Domain 1) and deviations from the intended intervention (Domain 2). All trials had at least some concern for risk of bias due to reporting of results, as a-priori analysis plans were not available or sufficiently detailed (Domain 5).
The overall risk of bias judgement decisions were modified from the original Cochrane RoB 2 Tool guidance to account for unavoidable characteristics with physical activity trials, such as lack of blinding and the patient-reported nature of outcome measures. Accordingly, eight trials were graded as having overall low risk of bias and one trial was graded as having some concerns for risk of bias because of its insufficient reporting on the randomisation process. The remaining 10 trials were considered as having high risk of bias due to missing > 5% of participants’ outcome data.
Behaviour change theory use
Coding of the Theory Coding Scheme is shown in Table 2. Most trials specified the behaviour change theories used; the exceptions were two trials that indirectly referenced a theory [33, 39], and one trial that reported being theory-based but with no reference or further information [28]. The theories used were the social cognitive theory (n = 10), transtheoretical model (n = 9), and theory of planned behaviour (n = 2). Four trials referred to more than one theory [25, 27, 29, 38], and one referenced a wide assortment of both behavioural and psychological theories [35]. No trials screened or selected participants based on theory-related constructs. Twelve (63%) trials reported using theory to develop the intervention, and eight trials (42%) reported using theory to tailor the intervention to participants. Items 7, 8, 9, and 11 were scored as one item due to generally ambiguous reporting around theoretical constructs or predictors of physical activity; only four trials did not link intervention techniques to theoretical constructs or predictors of physical activity [24, 26, 28, 32].
Coding of interventions using the Behaviour Change Technique Taxonomy is summarised by category in Table 3. The most frequently applied techniques included those associated with goals and planning (n = 18), shaping of knowledge (i.e. education and instruction) (n = 18), feedback and monitoring (n = 17), comparisons of outcomes (e.g. pros and cons) (n = 17), repetition and substitution (n = 14), social support (n = 11), and associations (e.g. prompts and cues) (n = 10).
Synthesis of intervention effects
Outcome measures and corresponding timepoints included in analyses are presented in Supplementary Material 2. Pooled post-intervention effect estimates showed medium improvements in self-reported physical activity (SMD = 0.57, CI = 0.33, 0.80) (Fig. 3), objectively measured physical activity (SMD = 0.52, CI = 0.14, 0.89) (Fig. 4) and physical function (SMD = 0.66, CI = 0.33, 0.98), and small improvements in quality of life (SMD = 0.18, CI = 0.08, 0.29), fatigue (SMD = 0.26, CI = 0.14, 0.37), aerobic fitness (0.30, CI = 0.10, 0.50), and muscular strength (SMD = 0.20, CI = − 0.01, 0.41) (Supplementary Material 3). At follow-up assessments, small improvements were made for self-reported physical activity, quality of life, fatigue, and physical function. Forest plots for the meta-analysis of secondary outcomes are presented in Supplementary Material 3. Statistical heterogeneity (assessed using I2, where values > 50% may represent substantial heterogeneity) may be substantial for self-reported (I2 = 67%) and objectively measured physical activity (I2 = 75%).
Subgroup analyses comparing the effectiveness of trials according to each specific theory were performed but were not significant and produced wide and overlapping subgroup CIs. Sensitivity analysis for trials at high risk of bias revealed that for objectively measured physical activity and quality of life, the effect estimate for low-risk trials alone was not significant, but when high-risk trials were included, the overall effect estimate became significant. Sensitivity analysis for trials that required imputation of means or SD values showed negligible differences.
Discussion
Nineteen physical activity interventions for women with breast cancer based on behaviour change theories were identified, their results synthesised, and application of behaviour change theories appraised. Intervention groups had small to medium improvements in physical activity and related health outcomes when compared to control groups. Risk of bias was present in all trials to various extents, with missing data from participant dropout being the primary source. However, the overall risk of bias judgements were made with consideration of unavoidable factors in physical activity studies, as is the inability to blind participants allocated to the intervention. The most referenced behaviour change theories were the Social Cognitive Theory and the Transtheoretical Model. Although different theories were used with varying degrees of application, trials reported sharing the use of some common behaviour change techniques, for example goal setting and planning, and the shaping of knowledge. This corresponds with recent research that proposes shifting the focus away from established behaviour change theories, to instead analysing the effectiveness and psychosocial mechanisms of action for the behaviour change techniques themselves [40].
Included trials mostly evaluated interventions against control groups that involved minimal to no instructions to participants. It was therefore unsurprising that between-group comparisons usually favoured the intervention arm, and were consistent with findings from a broader meta-analytic review for general physical activity trials in breast cancer [6] (which included this review’s trials). Only two trial designs used control groups that resembled the intervention arm but without the theory-based components [25, 39]; although the findings favoured the theory-based arm, their results did not vary distinctively from the other included trials.
The development and application of theoretical practices for physical activity behaviour are intended to promote both long-term and autonomous change, through in-depth deliberation of individuals’ intrinsic and extrinsic context, such as personal goals, preferences, and underlying attitudes towards the behaviour [7]. As outlined in the Theory Coding Scheme, trials using behaviour change theories should aim to incorporate them in their entirety, and also effectively assess any changes in the behaviour. The results from this review indicate that theory application, although varied, was not incorporated in its entirety. Several trials mentioned a theoretical construct as a predictor of the behaviour, used theory to select or develop intervention techniques, and used theory to tailor the intervention to participants, but descriptions were typically concise and the use of theory appeared to be secondary to other intervention components, namely the instructed exercise parameters. In some cases, the prescribed intervention will serve participants’ preferences and needs, but not all personal and unique contexts will be accommodated if the support they require is not offered within the program [41]. Although no included trials screened participants based on theory-relevant outcomes, 59% of trials selectively recruited participants who were not regularly physically active. Such inclusion criteria should be of priority, akin to how behaviour change interventions for other lifestyle health behaviours would naturally concentrate on relevant participants, for example smoking cessation interventions. In many cases, the reporting around theory application was clear in the introduction but diminished in the methods and results, as demonstrated by how few trials linked theory constructs to the intervention, such that links between the theory constructs, intervention components and interpretation of the results were ambiguous or lacking. This is aligned with the continuing discourse on the delineation between interventions that are merely “theory-informed” versus truly “theory-based” [42]; a more comprehensive implementation of theory has been suggested to enhance the effectiveness of an intervention in producing behaviour change [13]. The Theory Coding Scheme ratings from this review were similar to behaviour change trials in other populations [43], indicating that health behaviour studies generally do not fulfil all the items and hence are not maximising their potential.
Behaviour change techniques known to be pertinent to physical activity behaviour, namely the monitoring of the behaviour, goal setting and goal revision, and receiving feedback [44], were used in the majority of included trials. Despite being an important component of behaviour change for people with cancer [45], the use of social support, both practically (e.g. accompanying during physical activity) or emotionally (e.g. encouragement), was less commonly reported. In particular, none of the trials that cited the Transtheoretical Model reported the involvement of social support, despite it being a discrete concept within the model. Variability of behaviour change techniques across trials may be due to trials placing differing levels of priority on behaviour change within the intervention, ranging from using the theory as a core component for the study aims [22] to a supplementary means to enhance participant adherence to the overall intervention [26].
More generally, this review also reaffirms known challenges when delivering interventions in physical activity and behavioural research. The potential for discrepancies between a trial protocol and how it is delivered in practice is somewhat unavoidable, which has particular implications for any attempts to identify causal or correlational associations between use of theory, the intervention content, and its effects [46]. Use of evaluative tools like the Theory Coding Scheme relies on the information reported in manuscripts, and it is possible that interventions were extensively theory-based but details were not reported in the published article [47]. In addition, where intervention staff are trained behavioural coaches, behaviour change techniques may also be inadvertently implemented, when not part of the intended protocol. Similarly, contamination of the control, where control group participants independently increase physical activity despite being requested at baseline to not make changes, is acknowledged to potentially be a common occurrence and difficult to account for when making between-group comparisons [48]. Fidelity between the reporting of protocols and practice thus should be considered when evaluating intervention delivery and drawing conclusions on a trial’s findings.
The findings of this review are constrained by some limitations. First, the year of publication for the included trials extended to as early as 2005, and as a body of research, behaviour change for physical activity has been continually evolving and recommendations around the use and reporting of behaviour change theories are still being established. Therefore, the appraisal of earlier trials in comparison to more recent ones may not be equitable. Second, the most used measures of physical activity level in the included trials were self-reported, for which validity is debated [49]. However, this is less important when evaluating between-group differences as opposed to pre/post-intervention values alone. Third, there was marked heterogeneity between the behaviour change variables which were meta-analysed, along with lacking information about the development, validity, or reliability of the outcome measures, which together reduces the validity of the analysis. However, a meta-analysis was conducted to provide an approximate effect estimate.
Conclusions
The current research around the use of behaviour change theories in physical activity interventions for women with breast cancer was appraised. Although effective in increasing physical activity, the application and reporting of theories in the included trials was mixed, with many studies lacking in detail. To better understand the value of adopting behaviour change theories to increase physical activity, evaluation of the effectiveness of defined behavioural constructs and techniques is required.
Data availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Sweegers MG, Altenburg TM, Chinapaw MJ, Kalter J, Verdonck-De Leeuw IM, Courneya KS, et al. Which exercise prescriptions improve quality of life and physical function in patients with cancer during and following treatment? A systematic review and meta-analysis of randomised controlled trials. Br J Sports Med. 2018;52(8):505–13. https://doi.org/10.1136/bjsports-2017-097891.
Beasley JM, Kwan ML, Chen WY, Weltzien EK, Kroenke CH, Lu W, et al. Meeting the physical activity guidelines and survival after breast cancer: findings from the after breast cancer pooling project. Breast Cancer Res Treat. 2012;131(2):637–43. https://doi.org/10.1007/s10549-011-1770-1.
Friedenreich CM, Stone CR, Cheung WY, Hayes SC. Physical activity and mortality in cancer survivors: a systematic review and meta-analysis. JNCI cancer spectr. 2019;4(1). https://doi.org/10.1093/jncics/pkz080.
Courneya KS, Segal RJ, Gelmon K, Reid RD, Mackey JR, Friedenreich CM, et al. Predictors of supervised exercise adherence during breast cancer chemotherapy. Med Sci Sports Exerc. 2008;40(6):1180–7. https://doi.org/10.1249/MSS.0b013e318168da45.
McGuire R, Waltman N, Zimmerman L. Intervention components promoting adherence to strength training exercise in breast cancer survivors with bone loss. West J Nurs Res. 2011;33(5):671–89. https://doi.org/10.1177/0193945910379004.
Lahart IM, Metsios GS, Nevill AM, Carmichael AR. Physical activity for women with breast cancer after adjuvant therapy. Cochrane Database Syst Rev. 2018(1). https://doi.org/10.1002/14651858.CD011292.pub2.
Buchan DS, Ollis S, Thomas NE, Baker JS. Physical activity behaviour: an overview of current and emergent theoretical practices. J Obes. 2012;2012:11. https://doi.org/10.1155/2012/546459.
Bandura A. Social cognitive theory: an agentic perspective. Annual Review of Psychology2001. p. 1–26.
Prochaska JO, Velicer WF. The transtheoretical model of health behavior change. Am J Health Promot. 1997;12(1):38–48. https://doi.org/10.4278/0890-1171-12.1.38.
Ajzen I. The theory of planned behavior. Organ Behav Hum Decis Process. 1991;50(2):179–211. https://doi.org/10.1016/0749-5978(91)90020-T.
Phillips SM, McAuley E. Social cognitive influences on physical activity participation in long-term breast cancer survivors. Psychooncology. 2013;22(4):783–91. https://doi.org/10.1002/pon.3074.
Loprinzi PD, Cardinal BJ, Si Q, Bennett JA, Winters-Stone KM. Theory-based predictors of follow-up exercise behavior after a supervised exercise intervention in older breast cancer survivors. Support Care Cancer. 2012;20(10):2511–21. https://doi.org/10.1007/s00520-011-1360-0.
Bluethmann SM, Bartholomew LK, Murphy CC, Vernon SW. Use of theory in behavior change interventions: an analysis of programs to increase physical activity in posttreatment breast cancer survivors. Health Educ Behav. 2017;44(2):245–53. https://doi.org/10.1177/1090198116647712.
Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. Br Med J. 2019;366. https://doi.org/10.1136/bmj.l4898.
Michie S, Prestwich A. Are interventions theory-based? Development of a theory coding scheme. Health Psychol. 2010;29(1):1–8. https://doi.org/10.1037/a0016939.
Michie S, Richardson M, Johnston M, Abraham C, Francis J, Hardeman W, et al. The behavior change technique taxonomy (v1) of 93 hierarchically clustered techniques: building an international consensus for the reporting of behavior change interventions. Ann Behav Med. 2013;46(1):81–95. https://doi.org/10.1007/s12160-013-9486-6.
Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ et al. Cochrane Handbook for Systematic Reviews of Interventions. John Wiley & Sons; 2019.
López-López JA, Page MJ, Lipsey MW, Higgins JPT. Dealing with effect size multiplicity in systematic reviews and meta-analyses. Research Synthesis Methods. 2018;9(3):336–51. https://doi.org/10.1002/jrsm.1310.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7(3):177–88. https://doi.org/10.1016/0197-2456(86)90046-2.
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. Br Med J. 2003;327(7414):557–60. https://doi.org/10.1136/bmj.327.7414.557.
Hatchett A, Hallam JS, Ford MA. Evaluation of a social cognitive theory-based email intervention designed to influence the physical activity of survivors of breast cancer. Psychooncology. 2013;22(4):829–36. https://doi.org/10.1002/pon.3082.
Basen-Engquist K, Taylor CLC, Rosenblum C, Smith MA, Shinn EH, Greisinger A, et al. Randomized pilot test of a lifestyle physical activity intervention for breast cancer survivors. Patient Educ Couns. 2006;64(1–3):225–34. https://doi.org/10.1016/j.pec.2006.02.006.
Eakin EG, Lawler SP, Winkler EA, Hayes SC. A randomized trial of a telephone-delivered exercise intervention for non-urban dwelling women newly diagnosed with breast cancer: exercise for health. Ann Behav Med. 2012;43(2):229–38. https://doi.org/10.1007/s12160-011-9324-7.
Matthews CE, Wilcox S, Hanby CL, Der Ananian C, Heiney SP, Gebretsadik T, et al. Evaluation of a 12-week home-based walking intervention for breast cancer survivors. Support Care Cancer. 2007;15(2):203–11. https://doi.org/10.1007/s00520-006-0122-x.
Pinto BM, Papandonatos GD, Goldstein MG. A Randomized trial to promote physical activity among breast cancer patients. Health Psychol. 2013;32(6):616–26. https://doi.org/10.1037/a0029886.
van Waart H, Stuiver MM, van Harten WH, Geleijn E, Kieffer JM, Buffart LM, et al. Effect of low-intensity physical activity and moderate- to high-intensity physical exercise during adjuvant chemotherapy on physical fitness, fatigue, and chemotherapy completion rates: results of the PACES randomized clinical trial. J Clin Oncol. 2015;33(17):1918–27. https://doi.org/10.1200/jco.2014.59.1081.
Hartman SJ, Nelson SH, Myers E, Natarajan L, Sears DD, Palmer BW, et al. Randomized controlled trial of increasing physical activity on objectively measured and self-reported cognitive functioning among breast cancer survivors: The memory & motion study. Cancer. 2018;124(1):192–202. https://doi.org/10.1002/cncr.30987.
Anderson RT, Kimmick GG, McCoy TP, Hopkins J, Levine E, Miller G, et al. A randomized trial of exercise on well-being and function following breast cancer surgery: The RESTORE trial. J Cancer Surviv. 2012;6(2):172–81. https://doi.org/10.1007/s11764-011-0208-4.
Cadmus LA, Salovey P, Yu H, Chung G, Kasl S, Irwin ML. Exercise and quality of life during and after treatment for breast cancer: results of two randomized controlled trials. Psychooncology. 2009;18(4):343–52. https://doi.org/10.1002/pon.1525.
Campbell A, Mutrie N, White F, McGuire F, Kearney N. A pilot study of a supervised group exercise programme as a rehabilitation treatment for women with breast cancer receiving adjuvant treatment. Eur J Oncol Nurs. 2005;9(1):56–63. https://doi.org/10.1016/j.ejon.2004.03.007.
Demark-Wahnefried W, Case LD, Blackwell K, Marcom PK, Kraus W, Aziz N, et al. Results of a diet/exercise feasibility trial to prevent adverse body composition change in breast cancer patients on adjuvant chemotherapy. Clin Breast Cancer. 2008;8(1):70–9. https://doi.org/10.3816/CBC.2008.n.005.
Travier N, Velthuis MJ, Steins Bisschop CN, van den Buijs B, Monninkhof EM, Backx F et al. Effects of an 18-week exercise programme started early during breast cancer treatment: a randomised controlled trial. BMC Med. 2015;13(1). https://doi.org/10.1186/s12916-015-0362-z.
Mutrie N, Campbell AM, Whyte F, McConnachie A, Emslie C, Lee L, et al. Benefits of supervised group exercise programme for women being treated for early stage breast cancer: Pragmatic randomised controlled trial. Br Med J. 2007;334(7592):517–20. https://doi.org/10.1136/bmj.39094.648553.AE.
Rogers LQ, Hopkins-Price P, Vicari S, Pamenter R, Courneya KS, Markwell S, et al. A randomized trial to increase physical activity in breast cancer survivors. Med Sci Sports Exerc. 2009;41(4):935–46. https://doi.org/10.1249/MSS.0b013e31818e0e1b.
Kim S, Ko YH, Song Y, Kang MJ, Lee H, Kim SH, et al. Pre-post analysis of a social capital-based exercise adherence intervention for breast cancer survivors with moderate fatigue: a randomized controlled trial. Support Care Cancer. 2020;28(11):5281–9. https://doi.org/10.1007/s00520-020-05363-7.
Baruth M, Wilcox S, Der Ananian C, Heiney S. Effects of home-based walking on quality of life and fatigue outcomes in early stage breast cancer survivors: a 12-week pilot study. J Phys Act Health. 2015;12:S110–8. https://doi.org/10.1123/jpah.2012-0339.
Pinto BM, Frierson GM, Rabin C, Trunzo JJ, Marcus BH. Home-based physical activity intervention for breast cancer patients. J Clin Oncol. 2005;23(15):3577–87. https://doi.org/10.1200/JCO.2005.03.080.
Pinto BM, Stein K, Dunsiger S. Peers promoting physical activity among breast cancer survivors: a randomized controlled trial. Health Psychol. 2015;34(5):463–72. https://doi.org/10.1037/hea0000120.
Vallance JKH, Courneya KS, Plotnikoff RC, Yasui Y, Mackey JR. Randomized controlled trial of the effects of print materials and step pedometers on physical activity and quality of life in breast cancer survivors. J Clin Oncol. 2007;25(17):2352–9. https://doi.org/10.1200/JCO.2006.07.9988.
Michie S, Carey RN, Johnston M, Rothman AJ, De Bruin M, Kelly MP, et al. From theory-inspired to theory-based interventions: a protocol for developing and testing a methodology for linking behaviour change techniques to theoretical mechanisms of action. Ann Behav Med. 2018;52(6):501–12. https://doi.org/10.1007/s12160-016-9816-6.
An K-Y, Kang D-W, Morielli AR, Friedenreich CM, Reid RD, McKenzie DC, et al. Patterns and predictors of exercise behavior during 24 months of follow-up after a supervised exercise program during breast cancer chemotherapy. Int J Behav Nutr Phys Act. 2020;17(1):23. https://doi.org/10.1186/s12966-020-00924-9.
Michie S, Abraham C. Interventions to change health behaviours: evidence-based or evidence-inspired? Psychol Health. 2004;19(1):29–49. https://doi.org/10.1080/0887044031000141199.
Prestwich A, Sniehotta FF, Whittington C, Dombrowski SU, Rogers L, Michie S. Does theory influence the effectiveness of health behavior interventions? Meta-analysis Health Psychol. 2014;33(5):465–74. https://doi.org/10.1037/a0032853.
Michie S, Abraham C, Whittington C, McAteer J, Gupta S. Effective techniques in healthy eating and physical activity interventions: a meta-regression. Health Psychol. 2009;28(6):690–701. https://doi.org/10.1037/a0016136.
Barber FD. Social support and physical activity engagement by cancer survivors. Clin J Oncol Nurs. 2012;16(3):292. https://doi.org/10.1188/12.CJON.E84-E98.
Borrelli B. The assessment, monitoring, and enhancement of treatment fidelity in public health clinical trials. J Public Health Dent. 2011;71(s1):S52–63. https://doi.org/10.1111/j.1752-7325.2011.00233.x.
Meneses-Echavez JF, Rodriguez-Prieto I, Elkins M, Martínez-Torres J, Nguyen L, Bidonde J. Analysis of reporting completeness in exercise cancer trials: a systematic review. BMC Med Res Methodol. 2019;19(1):220. https://doi.org/10.1186/s12874-019-0871-0.
Courneya KS, Friedenreich CM, Sela RA, Quinney HA, Rhodes RE. Correlates of adherence and contamination in a randomized controlled trial of exercise in cancer survivors: an application of the theory of planned behavior and the five factor model of personality. Ann Behav Med. 2002;24(4):257–68. https://doi.org/10.1207/S15324796ABM2404_02.
Prince SA, Adamo KB, Hamel ME, Hardt J, Connor Gorber S, Tremblay M. A comparison of direct versus self-report measures for assessing physical activity in adults: a systematic review. International Journal of Behavioral Nutrition and Physical Activity. 2008;5. https://doi.org/10.1186/1479-5868-5-56.
Scruggs S, Mama SK, Carmack CL, Douglas T, Diamond P, Basen-Engquist K. Randomized trial of a lifestyle physical activity intervention for breast cancer survivors: effects on transtheoretical model variables. Health Promot Pract. 2018;19(1):134–44. https://doi.org/10.1177/1524839917709781.
Hayes S, Rye S, Battistutta D, Yates P, Pyke C, Bashford J, et al. Design and implementation of the Exercise for Health trial — a pragmatic exercise intervention for women with breast cancer. Contemp Clin Trials. 2011;32(4):577–85. https://doi.org/10.1016/j.cct.2011.03.015.
Hartman SJ, Weiner LS, Nelson SH, Natarajan L, Patterson RE, Palmer BW, et al. Mediators of a physical activity intervention on cognition in breast cancer survivors: evidence from a randomized controlled trial. JMIR Cancer. 2019;5(2): e13150. https://doi.org/10.2196/13150.
Hartman SJ, Natarajan L, Palmer BW, Parker B, Patterson RE, Sears DD. Impact of increasing physical activity on cognitive functioning in breast cancer survivors: Rationale and study design of Memory & Motion. Contemp Clin Trials. 2015;45:371–6. https://doi.org/10.1016/j.cct.2015.09.021.
Kim SH, Song YK, Han J, Ko YH, Lee H, Kang MJ, et al. Pro-inflammatory cytokine levels and cancer-related fatigue in breast cancer survivors: effects of an exercise adherence program. J Breast Cancer. 2020;23(2):205–17. https://doi.org/10.4048/jbc.2020.23.e22.
Kim S, Ko YH, Song Y, Kang MJ, Lee H, Kim SH, et al. Development of an exercise adherence program for breast cancer survivors with cancer-related fatigue—an intervention mapping approach. Support Care Cancer. 2019;27(12):4745–52. https://doi.org/10.1007/s00520-019-04785-2.
Mutrie N, Campbell A, Barry S, Hefferon K, McConnachie A, Ritchie D et al. Five-year follow-up of participants in a randomised controlled trial showing benefits from exercise for breast cancer survivors during adjuvant treatment. Are there lasting effects? J Cancer Surviv. 2012;6(4):420–30. https://doi.org/10.1007/s11764-012-0233-y.
Trinh L, Mutrie N, Campbell AM, Crawford JJ, Courneya KS. Effects of supervised exercise on motivational outcomes in breast cancer survivors at 5-year follow-up. Eur J Oncol Nurs. 2014;18(6):557–63. https://doi.org/10.1016/j.ejon.2014.07.004.
Pinto BM, Rabin C, Papandonatos GD, Frierson GM, Trunzo JJ, Marcus BH. Maintenance of effects of a home-based physical activity program among breast cancer survivors. Support Care Cancer. 2008;16(11):1279–89. https://doi.org/10.1007/s00520-008-0434-0.
Rabin C, Pinto BM, Frierson GM. Mediators of a randomized controlled physical activity intervention for breast cancer survivors. J Sport Exerc Psychol. 2006;28(3):269–84. https://doi.org/10.1123/jsep.28.3.269.
Pinto BM, Dunsiger S, Waldemore M. Physical activity and psychosocial benefits among breast cancer patients. Psychooncology. 2013;22(10):2193–9. https://doi.org/10.1002/pon.3272.
Pinto B, Stein K, Dunsiger S. Peer mentorship to promote physical activity among cancer survivors: Effects on quality of life. Psychooncology. 2015;24(10):1295–302. https://doi.org/10.1002/pon.3884.
Rogers LQ, Hopkins-Price P, Vicari S, Markwell S, Pamenter R, Courneya KS, et al. Physical activity and health outcomes three months after completing a physical activity behavior change intervention: Persistent and delayed effects. Cancer Epidemiol Biomark Prev. 2009;18(5):1410–8. https://doi.org/10.1158/1055-9965.EPI-08-1045.
Rogers LQ, Markwell S, Hopkins-Price P, Vicari S, Courneya KS, Hoelzer K, et al. Reduced barriers mediated physical activity maintenance among breast cancer survivors. J Sport Exerc Psychol. 2011;33(2):235–54. https://doi.org/10.1123/jsep.33.2.235.
Witlox L, Hiensch AE, Velthuis MJ, Steins Bisschop CN, Los M, Erdkamp FLG et al. Four-year effects of exercise on fatigue and physical activity in patients with cancer. BMC Med. 2018;16(1). doi:https://doi.org/10.1186/s12916-018-1075-x.
Velthuis MJ, May AM, Koppejan-Rensenbrink RAG, Gijsen BCM, van Breda E, de Wit GA et al. Physical Activity during Cancer Treatment (PACT) Study: design of a randomised clinical trial. BMC Cancer. 2010;10. doi:https://doi.org/10.1186/1471-2407-10-272.
Vallance JK, Courneya KS, Plotnikoff RC, Dinu I, MacKey JR. Maintenance of physical activity in breast cancer survivors after a randomized trial. Med Sci Sports Exerc. 2008;40(1):173–80. https://doi.org/10.1249/mss.0b013e3181586b41.
Vallance JKH, Courneya KS, Plotnikoff RC, Mackey JR. Analyzing theoretical mechanisms of physical activity behavior change in breast cancer survivors: results from the Activity Promotion (ACTION) Trial. Ann Behav Med. 2008;35(2):150–8. https://doi.org/10.1007/s12160-008-9019-x.
Vallance J, Plotnikoff RC, Karvinen KH, MacKey JR, Courneya KS. Understanding physical activity maintenance in breast cancer survivors. Am J Health Behav. 2010;34(1):225–36.
van Waart H, Stuiver MM, van Harten WH, Sonke GS, Aaronson NK. Design of the Physical exercise during Adjuvant Chemotherapy Effectiveness Study (PACES): a randomized controlled trial to evaluate effectiveness and cost-effectiveness of physical exercise in improving physical fitness and reducing fatigue. BMC Cancer. 2010;10. doi:https://doi.org/10.1186/1471-2407-10-673.
Acknowledgements
The authors would like to thank Gemma Louise Tierney for her assistance with data extraction for this review.
Funding
Mark Liu was supported by the Sally Crossing Memorial Scholarship.
Author information
Authors and Affiliations
Contributions
All authors collaborated in conceiving the review idea, aims, and design. ML leads the data extraction process with contributions from all other authors. ML was responsible for data analysis and the writing of the initial draft, with key input from SK and JY to shape the direction and conclusions of the review. All authors assisted with editing the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, M.G., Davis, G.M., Kilbreath, S.L. et al. Physical activity interventions using behaviour change theories for women with breast cancer: a systematic review and meta-analysis. J Cancer Surviv 16, 1127–1148 (2022). https://doi.org/10.1007/s11764-021-01104-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11764-021-01104-9