Abstract
Background
Chemotherapy-induced peripheral neuropathy (CIPN) is a significant and often lasting side effect of cancer treatment, with increasing CIPN severity associated with increasing deficits in balance, gait, and mobility. The 6-min walk test (6MWT) is a widely validated and utilized measure of general physical functioning and mobility, although its utility in a CIPN context is unclear. This study aimed to determine the utility of the 6MWT as an assessment of mobility deficits in a CIPN cohort and utilize the 6MWT to compare mobility data from CIPN patients to those of healthy and clinical populations.
Methods
Cancer survivors exposed to neurotoxic chemotherapies (N = 100; mean 17 ± 13 months post-treatment; mean age 59 ± 13 years) completed a single cross-sectional assessment of patient-reported and objective CIPN, mobility (6MWT), and disability.
Results
CIPN symptoms were reported in the majority of the cohort (87%). Increasing age, patient-reported and objective CIPN symptoms, and disability were associated with decreasing 6MWT distance (.48 ≤ R ≤ .63; p < .001) in bivariate models. Multiple regression models of 6MWT distance included age, sex, and patient-reported or objective CIPN severity as significant independent correlates (.62 ≤ R ≤ .64; p < .03). 6MWT distances in patients with CIPN symptom severity above the cohort mean were consistent with mean values reported in diabetic neuropathy and clinical populations.
Conclusions
Increased CIPN symptoms are associated with increased mobility deficits. The 6MWT demonstrates promising utility as a mobility assessment in a CIPN cohort.
Implications for Cancer Survivors
The impact of the progression of CIPN on mobility deficits in survivors emphasizes the need for effective interventions to treat and prevent CIPN.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chemotherapy-induced peripheral neuropathy (CIPN) is a significant side effect of cancer treatment, producing numbness and tingling in the hands and feet and resulting in deficits in fine motor function and long-term disability [1, 2]. Up to 40% of survivors treated with neurotoxic chemotherapies including taxanes, vinca alkaloids, and platinum compounds are estimated to be affected by CIPN [3]. The resolution of CIPN symptoms varies significantly between patients and between different neurotoxic cancer therapies. CIPN symptoms may improve in the 6–12 months following the cessation of neurotoxic therapy, although symptoms may also persist following treatment in a proportion of patients; the percentage of patients affected by long-term CIPN is still unclear and varies widely across prior studies [2], but is likely to exceed 30% [1]. Functionally, CIPN is associated with an increased fall incidence and deficits in balance, gait (i.e., gait speed, step length [4, 5]), and mobility [4,5,6,7]. Gait, balance, and mobility deficits have been shown to be influenced by CIPN severity, with increasing CIPN symptom severity associated with increasing balance, mobility, and gait dysfunction [4, 7, 8].
To facilitate high-quality clinical trials in CIPN, a recent consensus statement has advocated for the inclusion of relevant functional measures into trial designs [9]. To this end, the 6-min walk test (6MWT), an integrated measure of general physical functioning and mobility [10, 11], could serve as a valuable summary outcome measure. Increased neuropathy severity has been associated with reduced 6MWT distance in prior studies of childhood cancer survivors [7]. The 6MWT has been demonstrated to be a valid and reliable measure across a range of cohorts including cancer patients and survivors [12,13,14,15], has established normative values from other populations available for comparison [16,17,18,19], and is a low-cost assessment requiring no specialized equipment. However, the ability of the 6MWT to quantify increasing functional deficits related to increasing CIPN symptoms has yet to be investigated in adult cancer survivors. Additionally, 6MWT data has yet to be utilized to compare mobility in CIPN with other cohorts.
The primary aim of this study was to determine the utility of the 6MWT as an assessment of functional mobility deficits appearing with increasing CIPN symptoms. The secondary aim of the study was to use the 6MWT to compare mobility and function in cancer survivors exposed to neurotoxic cancer treatments to data from healthy and clinical populations.
Methods
Patients
Patients underwent comprehensive neurological testing including a 6MWT as part of ongoing exercise or observational studies from June 2016 to September 2018 (IN FOCUS study; www.infocusstudy.org.au). Patients were included if they were 3 months to 5 years post-treatment with known neurotoxic chemotherapies (oxaliplatin, cisplatin, docetaxel, paclitaxel, nab-paclitaxel, thalidomide, vincristine, vinblastine, vinorelbine, bortezomib, lenalidomide [2]) and had valid 6MWT data. Eligible patients were identified from oncology clinic lists and approached by research staff following approval from their treating clinician. The mean time since treatment completion was 17 months (Table 1). Patients who agreed to participate were scheduled for testing at the earliest suitable date. Written informed consent was given by all patients prior to study participation, and the study was conducted according to the Declaration of Helsinki. The study was approved by the Human Research Ethics Committee of South Eastern Sydney Local Health District and delivered at hospitals in the greater Sydney and Brisbane areas.
6MWT
Mobility was evaluated using the 6MWT [20]. Patients were instructed to walk between two markers set 15 m apart as many times as possible over 6 min. Scripted verbal encouragement was given at 1-min intervals. A greater distance (meters) covered over 6 min indicates greater mobility and general functioning.
Assessment of CIPN severity
A combination of objective and patient-reported assessments was utilized as per recommendations from prior study demonstrating that assessment of both domains is required for comprehensive evaluation of CIPN [21]. All patients were assessed using both the Total Neuropathy Score, clinical (TNSc [22, 23]; objective) and EORTC CIPN-20 ( [24] patient-reported) instruments, as described below.
Total Neuropathy Score, clinical (TNSc)–objective CIPN assessment
The TNSc includes clinical assessments of muscle weakness and numbness and tingling in the hands and feet, as well as pinprick sensibility, vibration sensibility, tendon reflexes, and strength assessments. Each item of the TNSc is scored from 0 to 4, with the total score ranging from 0 to 24 points; a higher score indicates greater neuropathy severity.
EORTC CIPN-20
The EORTC CIPN-20 questionnaire was used to assess patient-reported neuropathy across sensory, motor, and autonomic domains. Each of the 20 items is scored from 1 (not at all) to 4 (very much), with and the total questionnaire converted to a scale from 0 to 100; a higher score indicates increased symptom burden. Additionally, to measure the extent of sensory symptoms in the feet and hands, a “CIPN symptom index” consisting of the first four CIPN-20 items was utilized—items 1 and 2 concern tingling in the hands and feet, and items 3 and 4 address numbness. CIPN symptom index scores were classified in the following manner (4–16 possible points): no symptoms = 4 points, mild symptoms = 5–8 points, moderate symptoms = 9–12 points, and severe symptoms = 13–16 points.
Assessment of disability
CIPN Rasch-Built Overall Disability Scale
The CIPN Rasch-Built Overall Disability Scale (CIPN-R-ODS) is a 28-item questionnaire designed to address general disability, with an emphasis on domains relevant to CIPN [25]. Patients were asked to indicate whether each of the 28 specified activities was “possible with no difficulty” (2 points), “possible, with some difficulty” (1 point), or “not possible to perform” (0 points). Scores were converted to a 0–100 scale using previously established methodology [25]; a lower score indicates greater disability.
Data analysis
Bivariate linear regression analyses were conducted to confirm previously reported relations between decreasing 6MWT distance and increasing age [16, 17] in the study cohort. Similarly, an independent-samples t test was conducted to confirm the relationship between increased 6MWT distance and male sex [16, 17]. Sex was coded as “1” for male and “0” for female in all analyses. Bivariate linear regression analyses were also conducted to determine the relations between 6MWT distance and patient-reported CIPN (CIPN-20 score), objective CIPN (TNSc), and overall disability (CIPN-R-ODS). Following confirmation of significant linear relations or differences with respect to age, sex, and CIPN symptoms, a stepwise multiple linear regression model was used to determine the interactions between 6MWT distance and age, sex, and CIPN severity (patient reported or objective).
6MWT distances for each individual patient were compared with normative values generated based on models from a large (N = 300) study of healthy adults [16]:
Men
Women
Average height and weight statistics for Australia were used to calculate normative values [26]. Normative values were compared to individual 6MWT distances from the study cohort and also to values generated from multiple linear regression models. Significance was set at α = 0.05 for all analyses. The strength of correlations was interpreted as per Hinkle et al. [27]. All statistical analyses were conducted in SPSS Statistics 24.0 (IBM; Armonk, NY).
Results
Patient characteristics
Data were available for 100 patients treated with neurotoxic chemotherapies. Eighty-seven patients reported numbness and tingling at the time of testing. The majority of patients were treated for breast cancer (39%), colorectal cancer (18%), or lymphoma (12%). Other cancer types included appendix, cervical, endometrial, hypopharyngeal, liver, lung, myeloma, pancreatic, prostate, testicular, tongue, and urothelial cancers. Taxanes were the most commonly received class of neurotoxic chemotherapies (46%), followed by platinum-based agents (34%). See Table 1 for full patient demographic details.
All patients completed the 6MWT without incident. Median 6MWT distance in the cohort was 425.5 m (mean = 416.4 ± 85.5 m), with distances ranging from 214 to 681 m. Patient 6MWT distances were 41.2% to 109.0% (median 78.6%) of age- and sex-matched normative values for healthy individuals.
Effect of CIPN severity and patient demographics on 6MWT distance
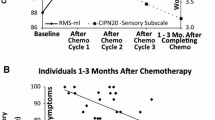
Increasing CIPN symptom severity, both patient reported and objectively assessed, was significantly associated with decreasing 6MWT distance (.47 < R < .49; p < .001) (Fig. 1). Increasing age was also associated with decreasing 6MWT distance (R = .49; p < .001; Fig. 1). 6MWT distance was significantly greater in males compared to females (males 447.4 ± 78.3 m, females 396.8 ± 87.4; p < .01) (Fig. 1). Decreasing 6MWT distance was also moderately to strongly correlated with increasing patient-reported disability (R = .62; p < .001; Fig. 1).
Multiple regression models including age, sex, and patient-reported or objectively assessed CIPN symptom severity explained 39–41% of the variance in 6MWT distance, with all independent variables significantly contributing (p < .03) (Table 2 and Fig. 2):
Based on these models, patients with more severe CIPN symptoms (CIPN-20 and TNSc scores at or above the mean) (Table 3) displayed 6MWT distances consistent with mean values reported in diabetic neuropathy [28], chronic stroke [29], and mild to moderate chronic obstructive pulmonary disease [30].
Discussion
This study demonstrates that CIPN is significantly associated with reduced 6MWT distance in cancer survivors exposed to neurotoxic chemotherapies, with greater symptom severity associated with greater mobility deficits. In multivariate models, increasing age and CIPN severity and female sex were all independent correlates of decreasing 6MWT distance. Reductions in 6MWT distance were moderately to strongly associated with increases in overall patient-reported disability. Accordingly, this study indicates that the 6MWT demonstrates promising utility as a means of quantifying functional mobility deficits appearing with increasing CIPN severity.
Comparisons to 6MWT values from healthy cohorts [16, 17] provide further evidence of functional impairments in the general survivorship population [31, 32], with even asymptomatic patients displaying reduced mobility compared to age- and sex-matched reference values [16]. In comparison to previously reported 6MWT distances in cancer patients and survivors, the mean distance from the present study (416.4 m) is near the bottom of the established range (403–594 m) [15, 33,34,35,36]. Given the significant impact of CIPN on 6MWT distance, it is unsurprising that 6MWT distance was reduced in our predominantly affected cohort (87% presenting CIPN symptoms). Similar associations between neuropathy and mobility impairment have been reported in diabetic, chronic inflammatory demyelinating polyradiculoneuropathy, and hereditary transthyretin amyloidosis cohorts [37,38,39,40]. Emphasizing the impact of the functional decline associated with CIPN symptoms, mobility impairments have been associated with increased mortality in both healthy and clinical populations [41, 42].
The strength of the models presented in the present study, with age, sex, and CIPN severity explaining greater than 39% of the variance in 6MWT distance, is consistent with models from large 6MWT studies of healthy populations [16, 17]. Additionally, the significant association between increasing CIPN severity and decreasing 6MWT distance in our cohort of adult cancer survivors was also reported in a cohort of young adult survivors of childhood cancers [7]. Both provide a degree of face validity to the models presented, although it should be noted that our models are primarily indicative of the impact of CIPN severity on mobility and function and the utility of the 6MWT in a CIPN setting. They are less likely to be a basis for prediction of 6MWT distance and mobility deficits in cancer survivors exposed to neurotoxic chemotherapies given limitations described below.
Determination of the independent impact of CIPN on 6MWT distance and mobility and functioning is ultimately limited by the absence of height and weight data obtained at the time of assessment. Height and weight have consistently been demonstrated to have a significant impact on 6MWT distance in healthy patients [16,17,18], with a higher body mass index (BMI = weight/(height)2) also associated with increased CIPN incidence [4, 43, 44]. However, a multivariate model of 6MWT distances in young adult survivors of childhood cancers included both BMI and CIPN severity as significant correlates [7], suggesting that the effects of BMI and CIPN severity on mobility are additive rather than redundant. Further research is required to confirm the independent influences of CIPN severity and BMI on mobility. The impact of other known effectors of 6MWT distance such as leg strength and medication usage also require investigation [10, 43].
Further study is also required to determine the sensitivity and responsiveness of the 6MWT in cancer survivors exposed to neurotoxic cancer treatments to inform prospective use in CIPN trials. The results of a recent exercise trial suggest good responsiveness of the 6MWT in CIPN [45], with an increase in 6MWT distance mirroring improvements in clinical and patient-reported CIPN following an 8-week exercise intervention. The development of exercise interventions to simultaneously alleviate both CIPN symptoms and related balance and mobility deficits is promising [45,46,47], although investigations have only been conducted in small cohorts of survivors to date. A complete understanding of the effects of the only recommended pharmaceutical intervention for CIPN symptom relief (duloxetine [48]) is presently lacking, as studies have not included objective functional and neurophysiologic outcomes. Accordingly, additional research including appropriate symptom, neurophysiologic and functional outcomes is needed to assess whether interventions deliver maximum value to patients by comprehensively addressing both CIPN symptom burden and functional deficits.
In conclusion, the 6MWT demonstrates promising utility as an assessment of increasing deficits in mobility and general disability related to increasing CIPN symptom severity. Comparisons to 6MWT data from other populations highlight the functional impact of CIPN; 6MWT distances were reduced regardless of CIPN severity compared to healthy populations and similar to diabetic neuropathy and other clinical populations in patients with more severe CIPN. These data further emphasize the need for effective interventions to treat CIPN symptoms and related functional deficits, with the 6MWT a potentially valuable endpoint for studies of such interventions.
References
Seretny M, Currie GL, Sena ES, Ramnarine S, Grant R, MacLeod MR, et al. Incidence, prevalence, and predictors of chemotherapy-induced peripheral neuropathy: a systematic review and meta-analysis. PAIN. 2014;155(12):2461–70.
Park SB, Goldstein D, Krishnan AV, Lin CSY, Friedlander ML, Cassidy J, et al. Chemotherapy-induced peripheral neurotoxicity: a critical analysis. CA Cancer J Clin. 2013;63(6):419–37.
Wolf S, Barton D, Kottschade L, Grothey A, Loprinzi C. Chemotherapy-induced peripheral neuropathy: prevention and treatment strategies. Eur J Cancer. 2008;44(11):1507–15.
Winters-Stone KM, Horak F, Jacobs PG, Trubowitz P, Dieckmann NF, Stoyles S, et al. Falls, functioning, and disability among women with persistent symptoms of chemotherapy-induced peripheral neuropathy. J Clin Oncol. 2017;35:2604–12 JCO 2016.71. 3552.
Hile ES, Fitzgerald GK, Studenski SA. Persistent mobility disability after neurotoxic chemotherapy. Phys Ther. 2010;90(11):1649–57.
Gewandter J, Fan L, Magnuson A, Mustian K, Peppone L, Heckler C, et al. Falls and functional impairments in cancer survivors with chemotherapy-induced peripheral neuropathy (CIPN): a University of Rochester CCOP study. Support Care Cancer. 2013;21(7):2059–66.
Varedi M, Lu L, Howell CR, Partin RE, Hudson MM, Pui C-H, et al. Peripheral neuropathy, sensory processing, and balance in survivors of acute lymphoblastic leukemia. J Clin Oncol. 2018;36:2315–22 JCO2017.76. 7871.
Monfort SM, Pan X, Patrick R, Ramaswamy B, Wesolowski R, Naughton MJ, et al. Gait, balance, and patient-reported outcomes during taxane-based chemotherapy in early-stage breast cancer patients. Breast Cancer Res Treat. 2017;161(1):69–77.
Gewandter JS, Brell J, Cavaletti G, Dougherty PM, Evans S, Howie L, et al. Trial designs for chemotherapy-induced peripheral neuropathy prevention: ACTTION recommendations. Neurology. 2018;91(9):403–13.
Lord SR, Menz HB. Physiologic, psychologic, and health predictors of 6-minute walk performance in older people. Arch Phys Med Rehabil. 2002;83(7):907–11.
Harada ND, Chiu V, Stewart AL. Mobility-related function in older adults: assessment with a 6-minute walk test. Arch Phys Med Rehabil. 1999;80(7):837–41.
Kervio G, Carre F, Ville NS. Reliability and intensity of the six-minute walk test in healthy elderly subjects. Med Sci Sports Exerc. 2003;35(1):169–74.
Ries JD, Echternach JL, Nof L, Gagnon Blodgett M. Test-retest reliability and minimal detectable change scores for the timed “up & go” test, the six-minute walk test, and gait speed in people with Alzheimer disease. Phys Ther. 2009;89(6):569–79.
Hamilton DM, Haennel R. Validity and reliability of the 6-minute walk test in a cardiac rehabilitation population. J Cardiopulm Rehabil Prev. 2000;20(3):156–64.
Schmidt K, Vogt L, Thiel C, Jäger E, Banzer W. Validity of the six-minute walk test in cancer patients. Int J Sports Med. 2013;34(07):631–6.
Enright PL, Sherrill DL. Reference equations for the six-minute walk in healthy adults. Am J Respir Crit Care Med. 1998;158(5):1384–7.
Casanova C, Celli B, Barria P, Casas A, Cote C, De Torres J, et al. The 6-min walk distance in healthy subjects: reference standards from seven countries. Eur Respir J. 2011;37(1):150–6.
Troosters T, Gosselink R, Decramer M. Six minute walking distance in healthy elderly subjects. Eur Respir J. 1999;14(2):270–4.
Bittner V, Weiner DH, Yusuf S, Rogers WJ, Mcintyre KM, Bangdiwala SI, et al. Prediction of mortality and morbidity with a 6-minute walk test in patients with left ventricular dysfunction. Jama. 1993;270(14):1702–7.
Enright PL. The six-minute walk test. Respir Care. 2003;48(8):783–5.
Alberti P, Rossi E, Cornblath D, Merkies I, Postma T, Frigeni B, et al. Physician-assessed and patient-reported outcome measures in chemotherapy-induced sensory peripheral neurotoxicity: two sides of the same coin. Ann Oncol. 2013;25(1):257–64.
Cornblath D, Chaudhry V, Carter K, Lee D, Seysedadr M, Miernicki M, et al. Total neuropathy score validation and reliability study. Neurology. 1999;53(8):1660–4.
Cavaletti G, Frigeni B, Lanzani F, Piatti M, Rota S, Briani C, et al. The Total Neuropathy Score as an assessment tool for grading the course of chemotherapy-induced peripheral neurotoxicity: comparison with the National Cancer Institute-Common Toxicity Scale. J Peripher Nerv Syst. 2007;12(3):210–5.
Postma T, Aaronson N, Heimans J, Muller M, Hildebrand J, Delattre J-Y, et al. The development of an EORTC quality of life questionnaire to assess chemotherapy-induced peripheral neuropathy: the QLQ-CIPN20. Eur J Cancer. 2005;41(8):1135–9.
Binda D, Vanhoutte E, Cavaletti G, Cornblath D, Postma T, Frigeni B, et al. Rasch-built Overall Disability Scale for patients with chemotherapy-induced peripheral neuropathy (CIPN-R-ODS). Eur J Cancer. 2013;49(13):2910–8.
Australian Bureau of Statistics. Profiles of health, Australia, 2011-13. Canberra 2012.
Hinkle DE, Wiersma W, Jurs SG. Applied statistics for the behavioral sciences. Boston: Houghton Mifflin; 1988.
Mueller MJ, Tuttle LJ, LeMaster JW, Strube MJ, McGill JB, Hastings MK, et al. Weight-bearing versus nonweight-bearing exercise for persons with diabetes and peripheral neuropathy: a randomized controlled trial. Arch Phys Med Rehabil. 2013;94(5):829–38.
Wevers LE, Kwakkel G, van de Port IG. Is outdoor use of the six-minute walk test with a global positioning system in stroke patients' own neighbourhoods reproducible and valid? J Rehabil Med. 2011;43(11):1027–31.
Cote CG, Casanova C, Marin J, Lopez M, Pinto-Plata V, De Oca M, et al. Validation and comparison of reference equations for the 6-min walk distance test. Eur Respir J. 2008;31(3):571–8.
Peters E, Schulz LM, Reuss-Borst M. Quality of life after cancer—how the extent of impairment is influenced by patient characteristics. BMC Cancer. 2016;16(1):787.
Huang MH, Blackwood J, Godoshian M, Pfalzer L. Prevalence of self-reported falls, balance or walking problems in older cancer survivors from surveillance, epidemiology and end results—Medicare Health Outcomes Survey. J Geriatr Oncol. 2017;8(4):255–61.
Kaltsatou A, Mameletzi D, Douka S. Physical and psychological benefits of a 24-week traditional dance program in breast cancer survivors. J Bodyw Mov Ther. 2011;15(2):162–7.
LaStayo PC, Marcus RL, Dibble LE, Smith SB, Beck SL. Eccentric exercise versus usual-care with older cancer survivors: the impact on muscle and mobility-an exploratory pilot study. BMC Geriatr. 2011;11(1):5.
Basen-Engquist K, Taylor CLC, Rosenblum C, Smith MA, Shinn EH, Greisinger A, et al. Randomized pilot test of a lifestyle physical activity intervention for breast cancer survivors. Patient Educ Couns. 2006;64(1–3):225–34.
Yuen HK, Sword D. Home-based exercise to alleviate fatigue and improve functional capacity among breast cancer survivors. J Allied Health. 2007;36(4):257E–75E.
Bruce DG, Davis WA, Davis TM. Longitudinal predictors of reduced mobility and physical disability in patients with type 2 diabetes: the Fremantle Diabetes Study. Diabetes Care. 2005;28(10):2441–7.
van Sloten TT, Savelberg HH, Duimel-Peeters IG, Meijer K, Henry RM, Stehouwer CD, et al. Peripheral neuropathy, decreased muscle strength and obesity are strongly associated with walking in persons with type 2 diabetes without manifest mobility limitations. Diabetes Res Clin Pract. 2011;91(1):32–9.
Vita GL, Stancanelli C, Gentile L, Barcellona C, Russo M, Di Bella G, et al. 6MWT performance correlates with peripheral neuropathy but not with cardiac involvement in patients with hereditary transthyretin amyloidosis (hATTR). Neuromuscul Disord. 2018;29(3):213–20.
Spina E, Topa A, Iodice R, Tozza S, Ruggiero L, Dubbioso R, et al. Six-minute walk test is reliable and sensitive in detecting response to therapy in CIDP. J Neurol. 2019;266(4):860–65.
Hirvensalo M, Rantanen T, Heikkinen E. Mobility difficulties and physical activity as predictors of mortality and loss of independence in the community-living older population. J Am Geriatr Soc. 2000;48(5):493–8.
Passantino A, Lagioia R, Mastropasqua F, Scrutinio D. Short-term change in distance walked in 6 min is an indicator of outcome in patients with chronic heart failure in clinical practice. J Am Coll Cardiol. 2006;48(1):99–105.
Ottaiano A, Nappi A, Tafuto S, Nasti G, De Divitiis C, Romano C, et al. Diabetes and body mass index are associated with neuropathy and prognosis in colon cancer patients treated with capecitabine and oxaliplatin adjuvant chemotherapy. Oncology. 2016;90(1):36–42.
Bao T, Basal C, Seluzicki C, Li SQ, Seidman AD, Mao JJ. Long-term chemotherapy-induced peripheral neuropathy among breast cancer survivors: prevalence, risk factors, and fall risk. Breast Cancer Res Treat. 2016;159(2):327–33.
McCrary JM, Goldstein D, Sandler CX, Barry BK, Marthick M, Timmins HC, et al. Exercise-based rehabilitation for cancer survivors with chemotherapy-induced peripheral neuropathy. Support Care Cancer. 2019. https://doi.org/10.1007/s00520-019-04680-w.
Zimmer P, Trebing S, Timmers-Trebing U, Schenk A, Paust R, Bloch W, et al. Eight-week, multimodal exercise counteracts a progress of chemotherapy-induced peripheral neuropathy and improves balance and strength in metastasized colorectal cancer patients: a randomized controlled trial. Support Care Cancer. 2018;26(2):615–24.
Streckmann F, Lehmann H, Balke M, Schenk A, Oberste M, Heller A, et al. Sensorimotor training and whole-body vibration training have the potential to reduce motor and sensory symptoms of chemotherapy-induced peripheral neuropathy—a randomized controlled pilot trial. Support Care Cancer. 2019;27(7):2471–78.
Hershman DL, Lacchetti C, Dworkin RH, Lavoie Smith EM, Bleeker J, Cavaletti G, et al. Prevention and management of chemotherapy-induced peripheral neuropathy in survivors of adult cancers: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2014;32(18):1941–67.
Acknowledgments
This study was supported by a Cancer Institute NSW Program Grant (14/TPG/1-05) and a National Health and Medical Research Council of Australia (NHMRC) Project Grant (#1080521). SBP is supported by a NHMRC Career Development Fellowship (#1148595).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in the studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
McCrary, J.M., Goldstein, D., Wyld, D. et al. Mobility in survivors with chemotherapy-induced peripheral neuropathy and utility of the 6-min walk test. J Cancer Surviv 13, 495–502 (2019). https://doi.org/10.1007/s11764-019-00769-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11764-019-00769-7