Abstract
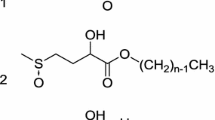
The esterification reaction kinetics of long-chain fatty acids and fatty alcohols catalyzed with a surfactant-coated lipase in a microaqueous n-hexane system were studied. The biocatalytic complex, surfactant-lipase adduct, showed 40 times the activity after a reaction time of 5 h compared to the unmodified lipase in the same reaction system. Various factors that may affect the activity of the modified lipase were studied, such as the influence of substrate fatty acid chainlength, water content, and temperature. By varying the concentration of each of the two substrates while keeping that of the other substrate constant, it was found that the esterification reaction follows Michaelis-Menten kinetics. The surfactant-enzyme complex kinetic parameters were determined with respect to both substrates. It was suggested that the kinetics of the lipase-catalyzed esterification reaction model follow a Ping-Pong Bi Bi mechanism with no substrate or product inhibition.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Linko, Y.-Y., M. Lamsa, A. Huhtala, and O. Rantanen, Lipase Biocatalysis in the Production of Esters, J. Am. Oil Chem. Soc. 72:1293–1299 (1995).
Bloomer S., P. Adlercreutz, and B. Mattiasson, Facile Synthesis of Fatty Acid Esters in High Yields, Enzyme Microb. Technol. 14:546–552 (1992).
Habulin, M., V. Krmelj, and Z. Knez, Synthesis of Oleic Acid Esters Catalyzed by Immobilized Lipase, J. Agric. Food Chem. 44:338–342 (1996).
Isono, Y., H. Nabetani, and M. Nakajima, Wax Ester Synthesis in a Membrane Reactor with Lipase-Surfactant Complex in Hexane, J. Am. Oil Chem. Soc. 72:887–890 (1995).
Senatalar, A.E., E. Erencek, A.T. Erciyes, Mechanism and Kinetics of the CoCl2-Catalyzed Esterification Reaction of Castor Oil with Oleic Acid, Ibid.:891–894 (1995).
Valivety, R.H., P.J. Halling, A.D. Peilow, and A.R. Macrae, Lipases from Different Sources Vary Widely in Dependence of Catalytic Activity on Water Activity, Biochim. Biophys. Acta 1122:143–146 (1992).
Balcao, V.M., A.L. Pavia, and F.X. Malcata, Bioreactors with Immobilized Lipases: State of the Art, Enzyme Microb. Technol. 18:392–416 (1996).
Boyer, J.L., B. Gilot, and R. Guiraud, Heterogeneous Enzymatic Esterification: Analysis of the Effect of Water, Appl. Microbiol. Biotechnol. 33:372–376 (1990).
Sonnet, P.E., G.M. McNeill, and W. Jun, Lipase of Geotrichum candidum Immobilized on Silica Gel, J. Am. Oil Chem. Soc. 71:1421–1423 (1994).
Luisi, P.L., Enzymes Hosted in Reverse Micelles in Hydrocarbon Solutions, Angew. Chem. Int. Ed. Engl. 24:439–450 (1985).
Inada, Y., M. Furukawa, H. Sasaki, Y. Kodera, M. Hiroto, H. Nishimura, and A. Matsushima, Biomedical and Biotechnological Application of PEG- and PM-Modified Proteins, TIBTECH. 13:86–91 (1995).
Zaidi, A., J.L. Gainer, and G. Carta, Fatty Acid Esterification Using Nylon-Immobilized Lipase, Biotechnol. Bioeng. 48:601–605 (1995).
Basri, M., K. Ampon, W.M.Z. Yunus, C.N.A. Razak, and A.B. Salleh, Amidation of Lipase with Hydrophobic Imidoesters, J. Am. Oil Chem. Soc. 69:579–583 (1992).
Basri, M., K. Ampon, W.M.Z.W. Yunus, C.N.A. Razak, and A.B. Salleh, Enzymic Synthesis of Fatty Esters by Hydrophobic Lipase Derivatives Immobilized on Organic Polymer Beads, Ibid.:407–411 (1995).
Marty, A., W. Chulalaksananukul, R.M. Willemot, and J.S. Condoret, Kinetics of Lipase-Catalyzed Esterification in Supercritical CO2, Ibid.:273–280 (1992).
Basheer, S., M. Nakajima, and U. Cogan, Sugar Ester-Modified Lipase for the Esterification of Fatty Acids and Long-Chain Alcohols, Ibid.:1475–1479 (1996).
Basheer, S., K. Mogi, and M. Nakajima, Surfactant-Coated Lipase for the Catalysis of Interesterification of Triglycerides and Fatty Acids, Biotechnol. Bioeng. 45:187–195 (1995).
Ito, Y., H. Fuji, and Y. Imanishi, Modification of Lipase with Various Synthetic Polymers and Their Catalytic Activities in Organic Solvents, Biotechnol. Prog. 10:398–402 (1994).
Goto, M., N. Kamiya, M. Miyata, and F. Nakashio, Enzymatic Esterification by Surfactant-Coated Lipase in Organic Media, Ibid.:263–268 (1994).
Kamiya, N., M. Goto, and F. Nakashio, Surfactant-Coated Lipase Suitable for the Enzymatic Resolution of Menthol as a Biocatalyst in Organic Media, Ibid.:270–275 (1995).
Kwon, S.J., K.M. Song, W.H. Hong, and J.S. Rhee, Removal of Water Produced from Lipase-Catalyzed Esterification in Organic Solvent by Pervaporation, Biotechnol. Bioeng. 46:393–395 (1995).
Valivety, R.H., P.J. Halling, and A.R. Macrae, Reaction Rate with Suspended Lipase Catalyst Shows Similar Dependence on Water Activity in Different Organic Solvents, Biochim. Biophys. Acta 1118:218–222 (1992).
Stamatis, H., A. Xenakis, U. Menge, and F.N. Kolisis, Kinetic Study of Lipase Catalyzed Esterification Reactions in Water-in-Oil Microemulsions, Biotechnol. Bioeng. 42:931–937 (1993).
Ramamurthi, S., and A.R. McCurdy, Lipase-Catalyzed Esterification of Oleic Acid and Methanol in Hexane—A Kinetic Study, J. Am. Oil Chem. Soc. 71:927–930 (1994).
Basheer, S., K. Mogi, and M. Nakajima, Interesterification Kinetics of Triglycerides and Fatty Acids with Modified Lipase in n-Hexane, Ibid.:511–518 (1995).
Okahata, Y., and K. Ijiro, A Surfactant-Coated Lipase as a New Catalyst for Triglycerides Synthesis in Organic Solvents, J. Chem. Soc. Chem. Commun.:1392 (1988).
Segel, I.H. (ed.), Enzyme Kinetics, Wiley Interscience, New
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Basheer, S., Cogan, U. & Nakajima, M. Esterification kinetics of long-chain fatty acids and fatty alcohols with a surfactant-coated lipase in n-hexane. J Amer Oil Chem Soc 75, 1785–1790 (1998). https://doi.org/10.1007/s11746-998-0332-0
Issue Date:
DOI: https://doi.org/10.1007/s11746-998-0332-0