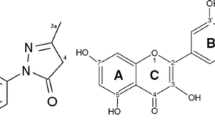
Abstract
Ab initio and density functional theory molecular orbital calculations were carried out at both the HF/6-31+G(d) and B3LYP/6-31+G(d) levels for the four antioxidants, p-hydroxycinnamic acid derivatives, namely, the p-coumaric, caffeic, ferulic, and sinapinic acid and the corresponding radicals, in an attempt to explain the structural dependency of the antioxidant activity of these compounds. Optimized resulting geometries, vibrational frequencies, absolute infrared intensities, and electron-donating ability are discussed. Both the high degree of conjugation and the extended spin delocalization in the phenoxyl radicals offer explanation for the scavenging activity of the four acids. In structurally related compounds, the calculated heat of formation value in radical formation appears as a meaningful molecular descriptor of antioxidant activity in accordance with experimental data. This becomes more clear at the B3LYP level.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- ARP:
-
antiradical power
- B3LYP:
-
Becke's three parameter hybrid functional using the Lee-Yang-Parr correlation functional
- DFT:
-
density functional theory
- DPPH:
-
2,2-diphenyl-1-pierylhydrazyl radical
- EC50 :
-
efficient concentration
- HF:
-
Hartree-Fock
- HOMO:
-
highest occupied molecular orbital
- IP:
-
induction period
- PF:
-
protection factor
References
Zhang, H.Y. (1998) Selection of Theoretical Parameter Characterizing Scavenging Activity of Antioxidants on Free Radicals, J. Am. Oil Chem. Soc. 75, 1705–1709.
Zhang, H.Y. (1999) Theoretical Methods Used in Elucidating Activity Differences of Phenolic Antioxidants, J. Am. Oil Chem. Soc. 76, 745–748.
van Acker, S.A.B.E., Koymans, L.M.H., and Bast, A. (1993) Molecular Pharmacology of Vitamin E: Structural Aspects of Antioxidant Activity, Free Radical Biol. Med. 15, 311–328.
van Acker, S.A.B.E., de Groot, M.J., van den Berg, D.J., Tromp, M.N.J.L., den Kelder, G.D.O., van der Vijgh, W.J.F., and Bast, A. (1996) A Quantum Chemical Explanation of the Antioxidant Activity of Flavonoids, Chem. Res. Toxicol. 9, 1305–1312.
Tanaka, K., Sakai, S., Tomiyama, S., Nishiyama, T., and Yamada, F. (1991) Molecular Orbital Approach to Antioxidant Mechanisms of Phenols by an ab initio Study, Bull. Chem. Soc. Jpn. 64, 2677–2680.
Tomiyama, S., Sakai, S., Nishiyama, T., and Yamada, F. (1993) Factors Influencing the Antioxidant Activity of Phenols by an ab initio Study, Bull. Chem. Soc. Jpn. 66, 299–304.
Rice-Evans, C.A., Miller, N.J., and Paganga, G. (1996) Structure-Antioxidant Activity Relationships of Flavonoid and Phenolic Acids, Free Radical Biol. Med. 20, 933–956.
Shahidi, F., Janitha, P.K., and Wanasundara, P.D. (1992) Phenolic Antioxidants, Crit. Rev. Food Sci. Nutr. 32, 67–103.
Brigelius-Flohe, R., and Traber, M.G. (1999) Vitamin E: Function and Metabolism, FASEB J. 13, 1145–1155.
Azzi, A., and Stocker, A. (2000) Vitamin E: Non Antioxidant Roles, Prog. Lipid Res. 39, 231–255.
van Acker, S.A.B.E., van den Berg, D.J., Tromp, M.N.J.L., Griffionen, D.H., van Bennekom, W.P., van der Vijgh, W.J.F., and Bast, A. (1996) Structural Aspects of Antioxidant Activity of Flavonoids, Free Radical Biol. Med. 20, 331–342.
Frankel, N.E. (1998) Lipid Oxidation, pp. 161–186, The Oily Press Ltd., Dundee.
Migliavacca, E., Carrupt, P.A., and Testa, B. (1997) Theoretical Parameters to Characterize Antioxidants. Part 1. The Case of Vitamin E and Analogs, Helv. Chim. Acta 80, 1613–1626.
He, D., Wang, Z., and Mo, F. (1996) Studies on the Relationship Between Electronic Structure and Antioxidant Activity of Vitamin E, Chin. J. Med. Chem. 6, 157–161.
Liu, S.L., Pan, J.H., Shi, D.Y., Chen, K.X., Wang, Q.M., Chen, S.M., and Yan, X.M. (1998) Relationship Between Structure and Anti-Oxidation of Tocopherol with Molecular Orbital Theory, Acta Pharmacol. Sin. 19, 513–518.
Zhang, H.Y. (1999) Investigation of the Effectiveness of HOMO to Characterize Antioxidant Activity, J. Am. Oil Chem. Soc. 76, 1109–1110.
Nagaoka, S.I., Kuranaka, A., Tsuboi, H., Nagashima, U., and Mukai, K. (1992) Mechanism of Antioxidant Reaction of Vitamin E. Charge Transfer and Tunneling Effect in Proton-Transfer Reaction, J. Phys. Chem. 96, 2754–2761.
Nagaoka, S.I., Mukai, K., Itoh, T., and Katsumata, S. (1992) Mechanism of Antioxidant Reaction of Vitamin E. 2 Photoelectron Spectroscopy and Ab Initio Calculation, J. Phys. Chem. 96, 8184–8187.
Ono, S., Okazaki, K., Sakurai, M., and Inoue, Y. (1997) Density Functional Study of the Radical Reactions of 3-Methyl-1-phenyl-2-pyrazolin-5-one (MCI-186): Implication for the Biological Function of MCI-186 as a Highly Potent Antioxidant Radical Scavenger, J. Phys. Chem. 101, 3769–3775.
Korzekwa, K.R., Jones, J.P., and Gillette, J.R. (1990) Theoretical Studies on Cytochrome P-450 Mediated Hydroxylation: A Predictive Model for Hydrogen Atom Abstractions, J. Am. Chem. Soc. 112, 7042–7046.
Bors, W., Heller, W., Michel, C., and Saran, M. (1990) Flavonoids as Antioxidants: Determination of Radical-Scavenging Efficiencies, Methods Enzymol. 186, 343–355.
Hueso-Urena, F., Jimenez-Pulido, S.B., Moreno-Carretero, M.N., and Rodriguez-Avi, J. (1998) Quantitative Structure Liquid Chromatographic Retention Time Relationship on Natural Phenols Found in Olive Oil, J. Am. Oil Chem. Soc. 75, 793–799.
Garcia-Granda, S., Beurskens, G., Beurskens, P.T., Krishna, T.S.R., and Desiraju, G.R. (1987) Structure of 3,4-Dihydroxy-trans-cinnamic Acid (caffeic acid) and Its Lack of Solid-State Topochemical Reactivity, Acta Cryst. C43, 683–685.
Nethaji, M., Pattabhi, V., and Desiraju, G.R. (1988) Structure of 3-(4-Hydroxy-3-methoxyphenyl)-2-propenoic acid, Acta Cryst. C44, 275–277.
Pouchert, C.J. (1970) The Aldrich Library of IR Spectra, Aldrich Chemical Co., St. Louis.
Hehre, W.J., Random, L., Schleyer, P.v.R., and Pople, J.A. (1986) Ab Initio Molecular Orbital Theory Wiley, New York.
Becke, A.D. (1993) Density-Functional Thermochemistry. III. The Role of Exact Exchange, J. Chem. Phys. 98, 5648–5652.
Ditchfield, R., Hehre, W.J., and Pople, J.A. (1971) Self-Consistent Molecular-Orbital Methods. IX. An Extended Gaussian-Type Basis for Molecular-Orbital Studies of Organic Molecules, J. Chem. Phys. 54, 724–728.
Hehre, W.J., Ditchfield, R., and Pople, J.A. (1972) Self-Consistent Molecular Orbital Methods. XII. Further Extensions of Gaussian-Type Basis Sets for Use in Molecular Orbital Studies of Organic Molecules, J. Chem. Phys. 56, 2257–2261.
Hariharan, P.C., and Pople, J.A. (1974) Accuracy of AHn Equilibrium Geometries by Single Determinant Molecular Orbital Theory, Mol. Phys. 27, 209–214.
Hariharan, P.C., and Pople, J.A. (1973) Influence of Polarization Functions on MO Hydrogenation Energies, Theor. Chim. Acta 28, 213–222.
Clark, T., Chandrasekhar, J., Spitznagel, G.W., and Schleyer, P.v.R. (1983) Efficient Diffuse Function Augmented Basis Sets for Anion Calculations. III The 3-21+G Basis Set for First-Row Elements, Lithium to Fluorine, J. Comp. Chem. 4, 294–301.
Frisch, M.J., Pople, J.A., and Binkley, J.S. (1984) Self-Consistent Molecular Orbital Methods 25. Supplementary Functions of Gaussian Basis Set, J. Chem. Phys. 80, 3265–3269.
Pulay, P., Fogarasi, G., Pang, F., and Boggs, J.E. (1979) Systematic Ab Initio Gradient Calculation of Molecular Geometries, Force Constants and Dipole Moment Derivatives, J. Am. Chem. Soc. 101, 2550–2560.
Peng, C., Ayala, P.Y., Schlegel, H.B., and Frisch, M.J. (1996) Using Redundant Internal Coordinates to Optimize Equilibrium Geometries and Transition States, J. Comput. Chem. 17, 49–56.
Frisch, M.J., Trucks, G.W., Schlegel, H.B., Gill, P.M.W., Johnson, B.G., Robb, M.A., Cheeseman, J.R., Keith, T., Petersson, G.A., Montgomery, J.A., et al. (1995) Gaussian 94, Revision D.4, Gaussian, Inc., Pittsburgh.
Brand-Williams, W., Cuvelier, M.E., and Berset, C. (1995) Use of a Free Radical Method to Evaluate Antioxidant Activity, Lebensm. Wiss. Technol. 28, 25–30.
Lampi, A.M., Hopia, A., Ekholm, P., and Piironen, V. (1992) Method for the Preparation of Triacylglycerol. Fractions from Rape Seed and Other Oils for Autoxidation Studies, Lebensm. Wiss. Technol. 25, 386–388.
Psomiadou, E., and Tsimidou, M. (1998) Simultaneous HPLC Determination of Tocopherols, Carotenoids, and Chlorophylls, for Monitoring their Effect on Virgin Olive Oil Oxidation, J. Agric. Food Chem. 46, 5132–5138.
Wu, Y.-D., and Lai, D.K.W. (1996) Density Functional Study of Substituent Effect on the O−H and O−CH3 Bond Dissociation Energies in Phenol and Anisole, J. Org. Chem. 61, 7904–7910.
Baesjou, P.J., Driessen, W.L., Challa, G., and Reedijk, J. (1997) Ab Initio Calculations on 2,6-Dimethylphenol and 4-(2,6-Dimethylphenoxy)-2,6-dimethylphenol. Evidence of an Important Role for the Phenoxyonium Cation in the Copper-Catalyzed Oxidative Phenol Coupling Reaction, J. Am. Chem. Soc. 119, 12590–12594.
Huang, C.-L., Wu, C.-C., and Lien, M.-H. (1997) Ab Initio Studies of Decarboxylations of the β-Keto Carboxylic Acids XCOH2COOH (X=H, OH and CH3) J. Phys. Chem. A 101, 7867–7873.
Foresman, J.B., and Frisch, E. (1996) Exploring Chemistry with Electronic Structure Methods, Gaussian Inc., Pittsburgh.
Pople, J.A., Scott, A.P., Wong, M.W., and Random, L. (1993) Scaling Factors for Obtaining Fundamental Vibrational Frequencies and Zero-Point Energies from HF/6-31G-Asterisk and Mp2/6-31G-Asterisk Harmonic Frequencies, Isr. J. Chem. 33, 345–350.
Wong, M.W. (1996) Vibrational Frequency Prediction Using Density Functional Theory, Chem. Phys. Lett. 256, 391–399.
Lin-Vien, D., Coltharp, N.B., Fateley, W.G., and Grasselli, J.G. (1991) The Handbook of Infrared and Raman Characteristic Frequencies of Organic Molecules, Academic Press, San Diego.
Welti, D. (1970) Infrared Vapor Spectra, Heyden and Son, New York.
Nyquist, R.A. (1985) The Interpretation of Vapor-Phase Infrared Spectra, Group Frequency Data The Sadtler Research Labs, Philadelphia.
Isaacs, N.S. (1995) Physical Organic Chemistry, 2nd edn., Longman Group Ltd., New York.
Papasavva, S., Illinger, K.H., and Kenny, J.E. (1996) Ab Initio Calculations on Fluoroethanes: Geometries, Dipole Moments, Vibrational Frequencies, and Infrared Intensities, J. Phys. Chem. 100, 10100–10115.
Papadopoulos, G., and Boskou, D. (1991) Antioxidant Effect of Natural Phenols on Olive Oil, J. Am. Oil Chem. Soc. 68, 669–671.
Marinova, E.M., and Yanishlieva, V.I.N. (1992) Effect of Temperature on the Antioxidative Action of Inhibitors in Lipid Autoxidation, J. Sci. Food Agric. 60, 313–318.
Marinova, E.M., and Yanishlieva, V.I.N. (1996) Antioxidative Activity of Phenolic Acids on Triacylglycerols and Fatty Acids Methylesters from Olive Oil, Food Chem. 56, 139–145.
Foti, M., Piattelli, M., Baratta, T.M., and Ruberto, G. (1996) Flavonoids, Coumarins, and Cinnamic Acids as Antioxidants in a Micellar System. Structure-Activity Relationship, J. Agric. Food Chem. 44, 497–501.
Terao, J., Karasawa, H., Arai, H., Nagao, A., Suzuki, T., and Takama, K. (1993) Peroxyl Radical Activity of Caffeic Acid and its Related Phenolic Compounds in Solution, Biosci. Biotechnol. Biochem. 57, 1204–1205.
Meyer, A.S., Donovan, J.L., Pearson, D.A., Waterhouse, A.L., and Frankel, E.N. (1998) Fruit Hydroxycinnamic Acids Inhibit Human Low-Density Lipoprotein Oxidation in Vitro, J. Agric. Food Chem. 46, 1783–1787.
Natella, F., Nardini, M., di Felice, M. and Scaccini, C. (1999) Benzoic and Cinnamic Acid Derivatives as Antioxidants: Structure-Activity Relation, J. Agric. Food Chem. 47, 1453–1459.
Gresch, N., Leboeuf, M., and Salahub, D. (1994) Energetics and Structure in Model Neutral, Anionic and Cationic Hydrogen-bonded Complexes: Combined ab initio SCF/MP2 Supramolecular, Density Functional and Molecular Mechanics Investigation, in Modeling the Hydrogen Bond, American Chemical Society Symposium Series (Smith, D.A., ed.), Vol. 569, pp. 82–112, American Chemical Society, Washington, DC.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Formrly at Laboratory of Applied Quantum Chemistry, School of ChemistryAristotle University of Thessaloniki, Thessaloniki, Greece
Supplementary material, Tables S1–S9, are available from the author E.G.B. upon request.
About this article
Cite this article
Bakalbassis, E.G., Chatzopoulou, A., Melissas, V.S. et al. Ab initio and density functional theory studies for the explanation of the antioxidant activity of certain phenolic acids. Lipids 36, 181–191 (2001). https://doi.org/10.1007/s11745-001-0705-9
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11745-001-0705-9