Abstract
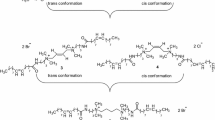
A new family of glucosamide-based trisiloxane gemini surfactants of the general formula (CH2OCH2)n (Me3SiOSiMeR1OSiMe3)2 (where R1=(CH2)3NR2(CH2)2NHCO (CHOH)4CH2OH; R2=CH2CH(OH)CH2OCH2; and n=0, 1, or 2) was prepared and characterized, both structurally and as aqueous surfactants. The monomer was prepared by amidation of the precursor amine functional trisiloxane with d-gluconic acid δ-lactone. Gemini surfactants were then prepared by the alkylation of the precursor secondary amine with oligoethylene glycol diglycidyl ethers. They were structurally and elemental analysis. Members of this family reduced the surface tension of water to approximately 21 mN/m at concentration levels of 10−5 mol/L. These gemini compounds showed two critical aggregation concentration values. This behavior resulted from the formation of premicellar aggregates before true micelles were formed.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- CAC:
-
critical aggregation concentration
- CMC:
-
critical micelle concentration
- NMR:
-
nuclear magnetic resonance
- PTC:
-
phasetransfer conditions
- TMAH:
-
tetramethyl ammonium hydroxide
References
Zana, R., M. Benrraou, and R. Rueff, Alkanediyl-α,ω-bis(dimethylalkylammonium bromide) Surfactants. 1. Effect of the Spacer Chain Length on the Critical Micelle Concentration and Micelle Ionization Degree, Langmuir 7:1072 (1991).
Alami, E., H. Levy, and R. Zana, Alkanediyl-α,ω-bis(dimethylalkylammonium bromide) Surfactants. 2. Structure of the Lyotropic Mesophases in the Presence of Water, Langmuir 9:940 (1993).
Alami, E., P. Beinert, P. Marie, and R. Zana, Alkanediyl-α,ω-bis(dimethylalkylammonium bromide) Surfactants. 3. Behavior at the Air-Water Interface, Langmuir 9:1465 (1993).
Dreja, M., W.P. Hintzen, H. Mays, and B. Tieke, Cationic Gemini Surfactants with Oligo (oxyethylene) Spacer Groups and Their Use in the Polymerization of Styrene in Ternary Microemulsion, Langmuir 15:391 (1999).
Rosen, M.J., J.H. Mathias, and L. Davenport, Aberrant Aggregation Behavior in Cationic Gemini Surfactants Investigated by Surface Tension, Interfacial Tension, and Fluorescence Methods, Langmuir 15:7340 (1999).
Menger, F.M., J.S. Keiper, and V. Azov, Gemini Surfactants with Acetylenic Spacers, Langmuir 16:2062 (2000).
Fang, L., M.J. Rosen, and S.B. Sulthana, Surface Properties of Cationic Gemini Surfactants and Their Interaction with Alkylglucoside or-maltoside Surfactants, Langmuir 17:1037 (2001).
Mathias, J.H., Fluorescence Study of Premicellar Aggregation in Cationic Gemini Surfactants, Langmuir 17:6148 (2001).
Rosen, M.J., and L.T. Liu, Surface Activity and Premicellar Aggregation of Some Novel Diquaternary Gemini Surfactants, J. Am. Oil Chem. Soc. 73:885 (1996).
Taiga, T., W. Zhang, T. Kida, Y. Nakatsuji, D. Ono, T. Takeda, and I. Ikeda, Novel Hydrolyzable and Biodegradable Cationic Gemini Surfactants: 1, 3-Bis[(acyloxyalkyl)-dimethylammonio]-2-hydroxypropane Dichloride, J. Surfact. Deterg. 3:167 (2000).
Taiga, T., W. Zhang, T. Kida, Y. Nakatsuji, K. Miyake, K. Matsushima, M. Tanaka, T. Furuta, and I. Ikeda, Preparation, Surface-Active Properties, and Antimicrobial Activities of Bis(alkyllammonium) Dichlorides Having a Butenylene or a Butynylene Spacer, J. Surfact. Deterg. 4:271 (2001).
Zhu, Y.P., K. Ishihara, A. Masuyama, Y. Nakatsuji, and M. Okahara, Preparation and Properties of Double-Chain Bis(quaternary ammonium) Compounds, J. Jpn. Oil Chem. Soc. 42:161 (1993).
Danit, D., Y. Talmon, and R. Zana, Vesicle-to-Micelle Transformation in Systems Containing Dimeric Surfactants, J. Colloid and Interface Sci. 185:84 (1997).
Zhu, Y.P., A. Masuyama, T. Nagata, and M. Okahara, Preparation and Properties of Double-Chain Surfactants Bearing Two Sulfonate Groups, J. Jpn. Oil Chem. Soc. 40:473 (1991).
Zhu, Y.P., A. Masuyama, T. Nagata, and M. Okahara, Synthesis and Properties of Bis(sulfonate) Types of Double-Chain Surfactants Bearing a Sulfur Atom in the Connecting Part, J. Jpn. Oil Chem. Soc. 42:86 (1993).
Zhu, Y.P., A. Masuyama, T. Nagata, and M. Okahara, Preparation and Surface Active Properties of Amphipathic Compounds with Two Sulfate Groups and Two Lipophilic Alkyl Chains, J. Am. Oil Chem. Soc 67:459 (1990).
Zhu, Y.P., A. Masuyama, T. Nagata, and M. Okahara, Preparation and Surface Active Properties of New Amphipathic Compounds with Two Phosphate Groups and Two Long-Chain Alkyl Groups, J. Am. Oil Chem. Soc. 68:268 (1991).
Zhu, Y.P., A. Masuyama, Y. Kobata, Y. Nakatsuji, M. Okahara, and M.J. Rosen, Double-Chain Surfactants with Two Carboxylate Groups and Their Relation to Similar Double-Chain Compounds, J. Colloid Interface Sci. 158:40 (1993).
Masuyama, A., C. Endo, S. Takeda, M. Nojima, D. Ono, and T. Takeda, Ozone-Cleavable Gemini Surfactants. Their Surface-Active Properties, Ozonolysis, and Biodegradability, Langmuir 16:368 (2000).
Duivenvoorde, F.L., M.C. Feiters, S.J. Gaast, and B.F.N. Engberts, Synthesis and Properties of Di-n-dodecyl α,ω-Alkyl Bisphosphate Surfactants, Langmuir 13:3737 (1997).
Zana, R., H. Levy, and K. Kwetkat, Mixed Micellization of Dimeric (gemini) Surfactants and Conventional Surfactants. I. Mixtures of an Anionic Dimeric Surfactant and of the Nonionic Surfactants C12E5 and C12E8, J. Colloid Interface Sci. 197:370 (1998).
Takeda, T., S. Yamamura, K. Honda, T. Stujisaka, M. Nakamura, and K. Horikawa, Synthesis and Properties of α,ω-Bis(amidopropyl-hydroxysulfobetaine)-type Amphoteric Surfactants, J. Jpn. Oil Chem. Soc. 39:576 (1990).
Tsubone, K., and K. Tajima, Aqueous Properties of Mixed Anionic Gemini Surfactant and Conventional Anionic Surfactant, J. Oleo Sci. 51:123 (2002).
Kunieda, H., N. Masuda, and K. Tsubone, Comparison Between Phase Behavior of Anionic Dimeric (gemini-type) and Monomeric Surfactants in Water and Water-Oil, Langmuir 16:6438 (2000).
Masuyama, A., T. Hirono, and Y.P. Zhu, Synthesis and Properties of Bis(taurine) Types of Double-Chain Surfactants, J. Jpn. Oil Chem. Soc. 41:301 (1992).
Jones, G.P., S. Regismond, K. Kwetkat, and R. Zana, Micellization of Nonionic Surfactant Dimers and of the Corresponding Surfactant Monomers in Aqueous Solution, J. Colloid Interface Sci. 243:496 (2001).
Pestman, J.M., K.R. Terpstra, M.C.A. Stuart, H.A. Doren, A. Brisson, R.M. Kellogg, and J.B.F.N. Engberts, Nonionic Bolaamphiphiles and Gemini Surfactants Based on Carbohydrates, Langmuir 13:6857 (1997).
Bergsma, M., M.L. Fielden, and J.B.F.N. Engberts, pH-Dependent Aggregation Behavior of Sugar-Amine Gemini Surfactant in Water: Vesicles, Micelles, and Monolayers of Hexane-1, 6-bis(hexadecyl-1′-deoxyglucitylamine), J. Colloid Interface Sci. 243:401 (2001).
Castro, M.J.L., J. Kovensky, and A.F. Cirelli, New Dimeric Surfactants from Alkyl Glucosides, Tetrahedron 55:12711 (1999).
Tracy, D.J., R.X. Li, and J. Yang, U.S. Patent 5, 863, 886 (1999).
Wilk, K.A., L. Syper, B.W. Domagalska, U. Komorek, I. Maliszewska, and R. Gancarz, Aldonamide-type Gemini Surfactants: Synthesis, Structural Analysis, and Biological Properties, J. Surfact. Deterg. 5:235 (2002).
Briggs, C.B.A., and A.R. Pitt, U.S. Patent 4,892,806 (1990).
Eastoe, J., P. Rogueda, B.J. Harrison, A.M. Howe, and A.R. Pitt, Properties of a Dichained “Sugar Surfactant”, Langmuir 10:4429 (1994).
Eastoe, J., P. Rogueda, A.M. Howe, and A.R. Pitt, Properties of New Glucamide Surfactants, Langmuir 12:2701 (1996).
Menger, F.M., and B.N.A. Mbadugha, Gemini Surfactants with a Disaccharide Spacer, J. Am. Chem. Soc. 123:875 (2001).
Wagner, R., L. Richter, R. Wersig, G. Schmaucks, B. Weiland, J. Weissmuller, and J. Reiners, Silicon-Modified Carbohydrate Surfactants I: Synthesis of Siloxanyl Moieties Containing Straight-Chained Glycosides and Amides, J. Appl. Organomet. Chem. 10:421 (1996).
Wagner, R., L. Richter, B. Weiland, J. Reiners, and J. Weissmuller, Silicon-Modified Carbohydrate Surfactants II: Siloxanyl Moieties Containing Branched Structures, J. Appl. Organomet. Chem. 10:437 (1996).
Wagner, R., L. Richter, B. Weiland, J. Weissmuller J. Reiners, and W. Kramer, Silicon-Modified Carbohydrate Surfactants III: Cationic and Anionic Compounds, J. Appl. Organomet. Chem. 11:523 (1997).
Wagner, R., L. Richter, B. Weiland, J. Weissmuller, J. Reiners, D. Schaefer, and S. Stadtmuller, Silicon-Modified Carbohydrate Surfactants IV: The Impact of Substructures on the Wetting Behaviour of Siloxanyl Modified Carbohydrate Surfactants on Low-Energy Surfaces, J. Appl. Organomet. Chem. 11:617 (1997).
Wagner, R., L. Richter, Y. Wu, J. Reiners, J. Weissmuller, E. Hengge, A. Kleewein, and K. Hassler, Silicon-Modified Carbohydrate Surfactants V: The Wetting Behaviour of Low-Molecular-Weight Siloxane, Carbosilane, Silane and Polysilane Precursors on Low-Energy Surfaces, J. Appl. Organomet. Chem. 11:645 (1997).
Wagner, R., L. Richter, Y. Wu, B. Weiland, J. Weissmuller, J. Reiners, E. Hengge, and A. Kleewein, Silicon-Modified Carbohydrate Surfactants VI: Synthesis of Carbosilane, Silane, Polysilane and Non-permethylated Siloxane Derivatives: The Wetting Behaviour of Epoxy-Modified Precursor Liquids on Nonpolar Surfaces, J. Appl. Organomet. Chem. 11:47 (1997).
Wagner, R., L. Richter, Y. Wu, J. Weissmuller, A. Kleewein, and E. Hengge, Silicon-Modified Carbohydrate Surfactants VII: Impact of Different Silicon Substructures on the Wetting Behaviour of Carbohydrate Surfactants on Low-Energy Surfaces—Distance Decay of Donor-Acceptor Forces, J. Appl. Organomet. Chem. 11:265 (1997).
Gradzielski, M., H. Hoffmann, P. Robisch, and W. Ulbricht, The Aggregation Behaviour of Silicone Surfactants in Aqueous Solutions, Tenside Surfactants Deterg. 27:366 (1990).
Snow, S.A., W.N. Fenton, and M.J. Owen, Synthesis and Characterization of Zwitterionic Silicone Sulfobetaine Surfactants, Langmuir 6:385 (1990).
Snow, S.A., W.N. Fenton, and M.J. Owen, Zwitterionic Organofunctional Siloxanes as Aqueous Surfactants: Synthesis and Characterization of Betaine Functional Siloxanes and Their Comparison to Sulfobetaine Functional Siloxanes Langmuir 7:868 (1991).
Snow, S.A., Synthesis, Characterization, Stability, Aqueous Surface Activity, and Aqueous Solution Aggregation of the Novel, Cationic Siloxane Surfactants (Me3SiO)2Si(Me)−(CH2)3 +NMe2(CH2)2ORX− [R=H,C(O)Me, C(O)NH(Ph); X=Cl,Br,I,NO3,MeOSO3], Langmuir 9:424 (1993).
Gentle, T.E., and S.A. Snow, Adsorption of Small Silicone Polyether Surfactants at the Air/Water Interface, Langmuir 11:2905 (1995).
Menger, F.M., and C.A. Littau, Gemini Surfactants: A new Class of Self-Assembling Molecules, J. Am. Chem. Soc. 115:10083 (1993).
Frindi, M., and B. Michels Alkanediyl-α,ω-bis(dimethylalkylammonium bromide) Surfactants. 4. Ultrasonic Absorption Studies of Amphiphile Exchange Between Micelles and Bulk Phase in Aqueous Micellar Solutions, Langmuir 10:1140 (1994).
Hattori, N., H. Hirata, H. Okabayashi, and C. O’Connor, Small-Angle Neutron-Scattering Study and Micellar Model of the Gemini (phenylenedimethylene)bis-(n-octylammonium)dibromide Surfactant Micelles in Water, Colloid Polym. Sci. 277:361 (1999).
Author information
Authors and Affiliations
About this article
Cite this article
Han, F., Zhang, G. Synthesis and characterization of glucosamide-based trisiloxane gemini surfactants. J Surfact Deterg 7, 175–180 (2004). https://doi.org/10.1007/s11743-004-0301-2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11743-004-0301-2