Abstract
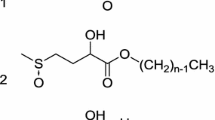
The synthesis and characterization of novel surfactants derived from tall oil fatty acid methyl esters are presented. The tall oil fatty acid methyl esters, which mainly contain methyl oleate, were converted to 9,10-epoxy derivatives and further transformed to the 9 (or 10) polyethylene glycol (PEG) ethers. Compounds with three different monomethylated PEG chain lengths with molecular weights of 350, 550, and 750, corresponding roughly to 7, 11, and 16 ethylene oxide units, were prepared. Surfactants were formed at an 89% overall yield from tall oil fatty acid. Cloud points were 46, 63, and 84°C, respectively, and surface tensions at the CMC (2.0, 1.0, and 0.4 mM, respectively) varied from 33 to 38 mN/m. Equilibrium surface tension and reflectometry measurements were made and results were similar to those of conventional long alkyl chain PEG surfactants. Results of the tensiometry and reflectometry measurements showed that the surfactants aligned better at the air-water interface than conventional surfactants. The adsorption properties for the three surfactants were similar to those of polyoxyethylene glycol alkyl ethers.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- (C9)2-PEG350:
-
tall oil fatty acid methyl ester hydroxy monomethyl-PEG 350-ether
- (C9)2-PEG550:
-
tall oil fatty acid methyl ester hydroxy monomethyl-PEG 550-ether
- (C9)2-PEG750:
-
tall oil fatty acid methyl ester hydroxy monomethyl-PEG 750-ether
- CMC:
-
critical micelle concentration
- DDS:
-
dimethyldichlorosilane
- DST:
-
dynamic surface tension
- GC:
-
gas chromatography
- IR:
-
infrared
- LCST:
-
lower critical solution temperature
- MPLC:
-
medium-pressure liquid chromatography
- NMR:
-
nuclear magnetic resonance
- PEG:
-
polyethylene glycol
References
Nilsson, F., O. Söderman, and I. Johansson, Physical-Chemical Properties of the n-Octyl β-d-glucoside/Water System. A Phase Diagram, Self-Diffusion NMR, and SAXS Study, Langmuir 12:902 (1996).
Nilsson, F., O. Söderman, and I. Johansson, Physical-Chemical Properties of Some Branched Alkyl Glucosides, Langmuir 13: 3349 (1997).
Svensson, M., Sterol Surfactants and Other Alicyclic Compounds, in Novel Surfactants: Preparation, Applications and Biodegradability, edited by K. Holmberg, Marcel Dekker, New York, 1998, p. 179.
Laughlin, R.G., Y.-C. Fu, F.C. Wireko, J.J. Scheibel, and R.L. Munyon, The Physical Science of N-Dodecanoyl-N-methylglucamine and Its Aqueous Mixtures, in Novel Surfactants: Preparation, applications and Biodegradability, edited by K. Holmberg, Marcel Dekker, New York, 1998, p. 5.
Zinkel, D.F., and J. Russel, Naval Stores, Pulp Chemicals Association, New York, 1988, p. 373.
Schmitz, W.R., and J.G. Wallace, Epoxidation of Methyl Oleate with Hydrogen Peroxide, J. Am. Oil Chem. Soc. 31:363 (1954).
Gall, R.J., and F.P. Greenspan, Recent Advances in in-situ Epoxidation Reactions with Resin Catalysts, J. Am. Oil Chem. Soc. 34: 161 (1957).
Ishii, Y., and Y. Sakata, A Novel Oxidation of Internal Alkynes with Hydrogen Peroxide Catalyzed by Peroxotungsten Compounds, J. Org. Chem. 55:5545 (1990).
Venturello, C., E. Alneri, and M. Ricci, A New, Effective Catalytic System for Epoxidation of Olefins by Hydrogen Peroxide Under Phase-Transfer Conditions, J. Org. Chem. 48:3831 (1983).
Ericsson, H., E. Sandberg, and E. Gottberg-Klingskog, U.S. Patent 5,101,051 (1992).
Alami, E., and K. Holmberg, Heterogemini Surfactants Based on Fatty Acid: Synthesis and Interfacial Properties, J. Colloid Interface Sci. 239:230 (2001).
Alami, E., K. Holmberg, and J. Eastoe, Adsorption Properties of Novel Gemini Surfactants with Nonidentical Head Groups, J. Colloid Interface Sci. 247:447 (2002).
Zana, R., M. Benrraou, and R. Rueff, Alkanediyl-α,ω-bis(dimethylalkylammonium bromide) Surfactants. 1. Effect of the Spacer Chain Length on the Critical Micelle Concentration and Micelle Ionization Degree, Langmuir 7:1072 (1991).
Alami, E., G. Beinert, P. Marie, and R. Zana, Alkanediyl-α,ω-bis(dimethylalkylammonium bromide) Surfactants. 3. Behavior at the Air-Water Interface, Langmuir 9:1467 (1993).
Menger, F.M., and C.A. Littau, Gemini Surfactants: A New Class of Self-Assembling Molecules, J. Am. Chem. Soc. 115:10083 (1993).
Seredyuk, V., E. Alami, M. Nydén, and K. Holmberg, Micellization and Adsorption Properties of Novel Zwitterionic Surfactants, Langmuir 17:5160 (2001).
Holmberg, B., Improved Gas Chromatographic Analysis of Fatty and Resin Acid Mixtures with Special Reference to Tall Oil, J. Am. Oil. Chem. Soc. 54:289 (1977).
Jay, R.R., Direct Titration of Epoxy Compounds and Aziridines, Anal. Chem. 36:667 (1964).
Miller, R., V.B. Fainerman, K.H. Schano, A. Hofmann, and W. Heyer, Dynamic Surface Tension Determination Using an Automated Bubble Pressure Tensiometer, Tenside Surfact. Deterg. 34:357 (1997).
Elwing, H., S. Welin, A. Askendal, U. Nilsson, and I. Lundström, Gradient Method for Studies of Macromolecular Interactions at the Liquid/Solid Interface, J. Colloid Interface Sci. 119: 35 (1987).
Corti, M., C. Minero, and V. Degiorgio, Cloud Point Transition in Nonionic Micellar Solutions, J. Phys. Chem. 88:309 (1984).
Eastoe, J., J.S. Dalton, P.G.A. Rogueda, E.R. Crooks, A.R. Pitt, and E.A. Simister, Dynamic Surface Tensions of Nonionic Surfactant Solutions, Colloid Interface Sci. 188:423 (1997).
Mitchell, D.J., G.J.T. Tiddy, L. Waring, T. Bostock, and M.P. McDonald, Phase Behaviour of Polyoxyethylene Surfactants with Water, J. Chem. Soc., Faraday Trans. 79:975 (1983).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Hedman, B., Piispanen, P., Alami, EO. et al. Synthesis and characterization of surfactants via epoxidation of tall oil fatty acid. J Surfact Deterg 6, 47–53 (2003). https://doi.org/10.1007/s11743-003-0247-4
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11743-003-0247-4