Abstract
Colorectal cancer can be a painful event, generally associated with changes in lifestyle for many patients. We studied the quality of life of the patients operated for colorectal malignancies 5 years after the diagnosis. Using detailed questionnaires, we investigated 220 patients of both sexes (mean age 66.5 years) 5 years (or more) after a curative operation for cancer of the large bowel. The short form 36 (SF-36) questionnaire took into consideration several aspects concerning work activity, physical activity, psychological attitude, alimentation, familial relationships, and other relevant components of lifestyle. Moreover, we compared the perception of the so-called SF-36 score between our patients and a comparison group in the general population. Both univariate and multivariate analysis were used. The obtained results revealed that familial and social relations were equally unchanged or tended to improve. Sexual activity declined in only 61(31.3%) subjects. Rather surprisingly (because of the average age at diagnosis), work activity remained unchanged in about half of the patients. Using the SF-36 questionnaire, the main differences from the general Italian population were seen in bodily pain (especially in the few individuals in whom a permanent stoma was necessary), social functioning and general physical health. In conclusion the results seem to suggest that the majority of patients who survive for more than 5 years after an operation for colorectal malignancy return to an almost normal life. The awareness among individuals about their disease, the improvements in surgical techniques and medical treatments are among the factors responsible for these positive results.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Colorectal neoplasms occur typically in economically developed populations, and is one of the leading causes of neoplastic morbidity and mortality in the Western World [1]. Over the years 1998–2005, in Italy, colorectal cancer accounted for 13.4% of cancer incidence in the male population (female 13.8%) and for 10.8% of all cancer deaths [ 2]. Survival at 5 years is around 60%, but varies depending on the stage, at diagnosis [3] from 50–90% for localized tumours to 5–10% for metastatic disease. Colorectal neoplasia is caused due to the interaction of different environmental factors (many of which are largely unknown) with a more or less marked genetic predisposition [4].
Cancer patients often experience distress. When symptoms of distress and anxiety persist over months, patients require psychosocial support [5, 6]. Quality of life (QoL) is a broad multi-dimensional concept encompassing many aspects of an individual’s life. It is difficult to define as it is a perceived subjective outcome. The World Health Organization (WHO) has defined quality of life as “Individuals’ perception of their position in the context of the culture and value systems in which they live, and in relation to their goals, expectations, standards and concerns” [7]. The term ‘quality of life’ includes the perceptions related to physical conditions, the psychological and social status of patients, and the subjective perception of the position in the social context of their relationships [8]. A study conducted on the quality of life involving the families of hundreds of patients shows that more than half of the patients are forced to leave work, while 35% show an increase in their liability for emotional stress and lack of energy due to various disorders [9]. Even family members of the affected patients reduce their job activity [10], and this has a negative impact on the physical and emotional status of patients [11].
The quality of life in cancer patients may be measured with psychometric questionnaires that analyze different aspects, [12] for instance, through the five qualities of lives dimension (physical function, role function, fatigue, pain, and dyspnoea). The Short Form 36 (SF-36) is a promising new instrument for measuring health perception in the general population. It is easy to use, acceptable to patients, and fulfils stringent criteria of reliability and validity. Its use in other contexts and with different disease groups requires further research [13]. We used a generic, SF-36 questionnaire to measure the quality of life in cancer patients [14]. There are different randomized studies on alternative techniques (like yoga), performed after the disease, demonstrating better quality of life in cancer patients [15].
The purpose of the present study is twofold: first, to investigate the quality of life in patients operated and treated for colorectal cancer who are in good health for at least five years after surgery; second, to determine the impact of ‘cancer experience’ on the individuals physical, mental and general health.
Methods
The study was observational and retrospective. All patients in our study were identified through the Specialized Colorectal Cancer Registry of the Health Care District of Modena. A total of 1,917 patients with colorectal malignancies were registered between 1995 and 2003; of these, 567 were considered suitable for the study (all stage I, II, or III individuals who survived 5 years or more after the diagnosis, and were in good physical conditions, i.e., without relevant comorbidities). Of the 567 patients, 220 (39%) agreed to participate, while 347 refused for several reasons, in particular: 42 were not interested in the project, 100 could not move due to advanced age, 175 declared lack of time, 26 changed addresses and 4 died before being contacted. The subjects were initially contacted through a letter requesting them to participate in the study. The letter was followed by a telephone call to explain the purpose of the research, and to verify the individual’s interest in being interviewed.
At the time of the interview, we provided detailed information on the design, purpose and duration of the study. For all the participants, we collected clinical data through the questionnaires and for the control group, healthy men and women were given with the SF-36 questionnaires.
Examination group
This study was carried out at the Department of Internal Medicine, Modena Policlinico (Italy). Patients completed the questionnaires while waiting for their follow-up appointment in the clinic. Data for this investigation originated from individuals of both sexes affected by colorectal cancer who survived 5 years or more after curative resection. The criteria were the following:
-
age between 40 and 75 years at diagnosis;
-
clinical stage TNM I, II or III (corresponding to Dukes’ stage A, B and C);
-
registration in Specialized Colorectal Cancer Registry of the Health Care District of Modena between 1995 and 2003;
-
ability to understand the questionnaire concerning quality of life.
Comparison group
The norm-based scoring can be illustrated by comparing the SF-36 profile using the original 0–100 scoring algorithms based on the totalised ratings method and the norm-based scoring algorithms for a representative sample of healthy Italian adults selected by the Research Institute of Statistics and Analysis of Public Opinion (Milan) who completed the SF-36 questionnaire (scores for all scales were transformed to a score where 0 represents worst possible health and 100 indicates best) [16].
Short form 36 (SF-36 questionnaire) [13]
SF-36 was developed in the United States over the last 80 years as a generic multidimensional questionnaire having 36 questions. The SF-36 comprises 36 items measuring eight health scales: physical functioning (limitations to everyday activities), role limitations due to physical problems (the degree by which physical health interferes with work and other activities), bodily pain, general health, vitality, social functioning (interference with social activities), role limitations due to emotional problems (the degree in which emotional problems interfere with work or other activities) and mental health (sense of nervousness and depression). According to standard scoring procedures, the sub-categories were linearly converted to a 0–100 scale, with higher scores indicating better functioning and lower scores worse (Table 1).
Along with the SF-36 questionnaire, we gave all the examined subjects a questionnaire concerning changes in lifestyle after surgery (relations with family, social relationships, physical activity, sexual activity, cultural interest, religious aspects, work activity and alimentation).
Specialized Colorectal Cancer Registry of the Healthcare District of Modena
The Specialized Colorectal Cancer Registry of the Healthcare District of Modena was established in 1984 with the main objective of evaluating the role of environmental and especially genetic factors in the pathogenesis of colorectal tumours [17].
The registration of a case usually begins with the morphological diagnosis of cancer and an accurate staging. In the successive phase, clinical charts are obtained and all other relevant information (personal and familial data, other instrumental clinical investigations—including endoscopies—details on surgery and other treatments) is transferred into the registration form. Familiarity was frequently evaluated through telephone calls (usually with the family doctors or directly with the patient or close relatives).
Statistical analysis [ 13]
Of the 220 questionnaires which were administered, only 89 were also analyzed with the SF-36 test. The eight sub-categories of the SF-36 and the total scores of physical health and mental health were calculated using scoring mathematical operations (algorithms).
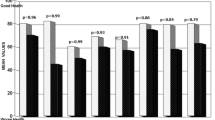
We performed paired t tests for comparing the SF-36 average scores as shown in Figs. 1, 2 and 3 between answers obtained on physical and mental health questions and the average reference scores of the Italian population, reported to the manual of instruction and guide to the analysis of the results of SF-36 (Fig. 1 and 2) [16], and in Fig. 3 between sexes in patient recovered from disease.
Comparison of the average values of the SF-36 score between the total sample and those of healthy Italian men as a reference *(the statistical significance of differences between groups was assessed with Student’s t tests). Grey bar sample of healthy Italian men, black bar men recovered from disease
Comparison of the average values of the SF-36 score between the total sample and those of healthy Italian women as a reference *(the statistical significance of differences between groups was assessed with Student’s t tests). Grey bar sample of healthy Italian women, black bar women recovered from disease
Moreover, we carried out univariate and multivariate linear regression analysis on the 81 patient who answered the questionnaire; eight patients were excluded from the model due to lack of information in the covariates. The covariates included in the model were: age, sex, marital status, Duke’s stage, site, therapy and stoma. All calculations were performed with Statistical Package for Social Sciences (SPSS) release 12.1 (SPSS, Munich).
Results
Sample characteristics
A total of 220 patients returned the questionnaires; 127 (57.3%) were men and 93 (42.7%) women, all were operated for colorectal cancer, 5 years after the diagnosis. The mean age was 66.6 years (range 43–81 years). As for marital status, 189 (85.9%) were married or living with a partner, 18 (10%) were divorced, and 13 (4.9%) were ‘single’. Fifty-nine patients (26.8%) had a TNM stage I disease, 104 (49.3%) stage II, 55 (26%) stage III, and 2 cases were classified as not-staged (NS). The majority of the tumours were located in the colon (79.9%), and remaining in the rectum (20.1%). Only 10% of all patients had a permanent stoma 5 years after colorectal cancer surgery (Table 2).
Changes of lifestyle 5 years after curative surgery for colorectal malignancy
Table 3 shows changes 5 years after curative surgery for colorectal malignancy. Familial relations were unchanged for 174 (79.1%) subjects while improved in 31 (14.1%). Social relationships remained unchanged in 177 (80.5%) subjects, improved in 22 (10%) and worsened in 21 (9.5%). Sexual activity worsened in 69 subjects (31.3%). The religious aspect improved for 35 patients (15.9%) and worsened in 12 (5.5%), but remained unchanged in the majority of them. Work activity after the disease was unchanged for more than half of the subjects, while worsened in 69 (31.9%). The last aspect evaluated was alimentation; this changed in 24 patients (10.9%), because some foods were no longer tolerated, but remained unchanged in the large majority of individuals (150, 68.2%). Most of the subjects reported their health as being generally good (49.5%) or very good (34.5%), and only a minority reported it as acceptable (15%).
SF-36 score results
We compared the perception of SF-36 score of healthy Italian men and women with colorectal cancer patients.
Figure 1 shows the perception of SF-36 score in healthy Italian men and patients 5 years after surgery. The highest differences were found in bodily pain, with a difference of 25 points between mean scores. The greatest differences between the groups were found in physical health (11 points), social functioning (18 points), and mental health (21 points). Differences between income strata were not significant in the role limitations functioning due to physical health and emotional health, general health and vitality of subjects.
Figure 2 represents a comparison of the perception of the SF-36 score between healthy Italian women and women included in our study group. There were significant differences in physical Health (21 points), bodily Pain (10 points), social functioning (31 points), role limitations functioning due to emotional (10 points) and mental health (15 points).
Figure 3 represents a comparison of the perception of SF-36 Score between men and women apparently cured of the disease. There were significant differences in the physical health (19 points), social functioning (20 points), and role limitations functioning due to emotional health (17 points).
The results of multivariate regression analysis (not described in detail) showed that only the age of the patients—but none of the clinical variables taken into consideration—was significantly associated with the 8 covariates of the SF-36 questionnaire (p-values between 0.05 and 0.01).
Discussion
The results of the present study can be summarized as follows. First, in general terms, several factors—including social relationships, physical activity, sexual activity, cultural interests, work activity, religious aspects, and diet—can be affected by the colorectal cancer event in a given individual. Second, quality of life in these patients can provide useful information concerning physical and emotional experiences. Third, in our investigation, familial relationships improved in 14% and social relations in 22% of the patients questioned; moreover, cultural interests tended to increase, presumably because affected individuals spent more time in them. Fourth, in the majority of patients work activity, physical activity and diet remained virtually unchanged 5 years after the diagnosis of the tumour, and more than 80% of them reported being in good or very good health. Finally, the use of more sophisticated SF-36 questionnaire revealed subtle differences—especially in physical health, social functioning and mental health—between patients with colorectal cancer and the comparison group.
As far as the method used in our study was concerned, we would like to make a few points. Broadly speaking, the patients who participated in this study were satisfied with the type of investigation, and the way it was conducted. However, several patients expressed their concerns in answering certain questions, since these were perceived as too personal, and, consequently, affecting their privacy; for other investigated subjects it was extremely painful to remember facts and episodes related to the disease despite the fact that at least 5 years had elapsed. Family doctors were of great help in some circumstances. Moreover, it should be noted that our sample, although representative of the registered population, was limited to 39% (220 of 567) of all individuals who were considered suitable for being enclosed in the study. This fact underlines the difficulty often encountered in gathering the sufficient number of patients in a disease that is typical of advanced age.
Previous studies on the same topic were of great help in designing our investigation. For instance, Dalton et al. [18] show that neoplasm occurrence in a given family tends to induce a profound emotional involvement of the family in addition to the patient. Dunn et al. [19] report that cancer patients obtain great benefit from spiritual support for overcoming disease-related distress. Moreover, Sultan et al. [20], underline that an adequate emotional support is associated with an improved quality of life after the ‘cancer event’. Thus, these investigations lend some support to the close relation between the quality of life after cancer and social interests shown in the present study.
It is well known from the literature that some kinds of alimentation (low calorie, high vegetable intake) and increased physical activity are beneficial in patients with colorectal malignancies and other neoplasms [21–25]. From our data we are unable to comment on changes in any of these variables in qualitative terms. However, in more general terms it may appear as positive, the fact that most investigated individuals maintained good physical activity, and that a fraction of patients —presumably influenced by the media, or by family doctors—shifted to a vegetarian-type diet [26, 27].The present study, however, was not designed in the order to explore the complex role that diet might have after a curative resection for colorectal cancer [28].
In conclusion, our population-based investigation seems to suggest that the large majority of individuals who survive more than 5 years after treatment for colorectal cancer—that includes surgery and, in specific cases, chemotherapy and radiotherapy—can tolerate this painful event without dramatizing the situation, and, more importantly, without changing their habits or lifestyle. This positive conclusion can be viewed as a composite effect of several factors, including a better education of patients, and the undoubted improvements of surgical techniques (which have almost abolished permanent stomas), together with medical management of pain and the possibility of supportive care.
References
Eustace K (2005) Colorectal cancer. Lancet 365(8):83–166
Airtum Working Group (2006) Cancer trend (1998–2005) Epidemiologia e Prevenzione, Suppl.2 pages 1–147
Parkin DM, Olsen AH, Sasieni P (2009) The potential for prevention of colorectal cancer in the UK. Eur J Cancer Prev 18:179–190
Movsas B (2003) Quality of life in oncology trials: a clinical guide. Semin Radiat Oncol 13:235–247
Brown P, Clark MM, Atherton P, Huschka M, Sloan JA, Gamble G, Girardi J, Frost MH, Piderman K, Rummans TA (2006) Will improvement in quality of life (QOL) impact fatigue in patients receiving radiation therapy for advanced cancer? Am J Clin Oncol 29:52–58
Hennessy EM, Stevinson C, Fox KR (2005) Preliminary study of the lived experience of exercise for cancer survivors. Eur J Oncol Nurs 9(2):155–166
Rogers SN (2010) Quality of life perspectives in patients with oral cancer. Oral Oncol 46(6):445–447
Akin S, Can G, Aydiner A, Ozdilli K, Durna Z (2010) Quality of life, symptom experience and distress of lung cancer patients undergoing chemotherapy. Eur J Oncol Nurs 14(5):400–409
Mastracci TM, Hendren S, O’Connor B, McLeod RS (2006) The impact of surgery for colorectal cancer on quality of life and functional status in the elderly. Dis Colon Rectum 49(12):1878–1884
Eriksson E, Arve S, Lauri S (2006) Informational and emotional support received by relatives before and after the cancer patient’s death. Eur J Oncol Nurs 10(1):48–58
Ballatori E, Porzio G, Roila F, Ruggeri B, Mattei A, Cortesi E (2007) Is there still a role for the uniscale assessment of quality of life? Tumori 93(1):78–81
Tsunoda A, Nakao K, Hiratsuka K, Tsunoda Y, Kusano M (2007) Prospective analysis of quality of life in the first year after colorectal cancer surgery. Acta Oncol 46(1):77–82
Brazier JE, Harper R, Jones NM, O’Cathain A, Thomas KJ, Usherwood T, Westlake L (1992) Validating the SF–36 health survey questionnaire: new outcome measure for primary care. BMJ 305(6846):160–164
Siassi M, Hohenberger W, Lösel F, Weiss M (2008) Quality of life and patient’s expectations after closure of a temporary stoma. Int J Colorectal Dis 23(12):1207–1212
Ulger O, Yağli NV (2010) Effects of yoga on the quality of life in cancer patients. Complement Ther Clin Pract 16(2):60–63
Apolone G, Mosconi P (1998) The Italian SF-36 Health Survey: translation, validation and norming. J Clin Epidemiol 51(11):1025–1036
Janaki MG, Kadam AR, Mukesh S, Nirmala S, Ponni A, Ramesh BS, Rajeev AG (2010) Magnitude of fatigue in cancer patients receiving radiotherapy and its short term effect on quality of life. J Cancer Res Ther 6(1):22–26
Dalton SO, Laursen TM, Ross L, Mortensen PB, Johansen C (2009) Risk for hospitalization with depression after a cancer diagnosis: a nationwide, population–based study of cancer patients in Denmark from 1973 to2003. J Clin Oncol 27(9):1440–1445
Dunn J, Lynch B, Rinaldis M et al (2006) Dimensions of quality of life and psychosocial variables most salient to colorectal cancer patients. Psycho oncology 15:20–30
Sultan S, Fisher DA, Voils CI, Kinney AY, Sandler RS, Provenzale D (2004) Impact of functional support on health-related quality of life in patients with colorectal cancer. Cancer 101:2737–2743
Ware JE Jr (1993) Measuring patients’ views: the optimum outcome measure. BMJ 306:1429–1430
Giovannucci E et al (1995) Physical activity, obesity, and risk for colon cancer and adenoma in men. Ann Int Med 122:327
Slattery ML et al (2003) Physical activity and colorectal cancer. Am J Epidemiol 158:214
Hou L (2004) Commuting physical activity and risk of colon cancer in Shangai, China. Am J Epidemiol 160:860
Slattery ML et al (2004) Associations between BMI, energy intake, energy expenditure, VDR genotype and colon and rectal cancers (United States). Cancer Causes 15(9):863–872
Chao A et al (2004) Amount, type and timing of recreational physical activity in relation to colon and rectal cancer in older adults:the cancer prevention study II nutrition cohort. Cancer Epidemiol Biomarkers Prev 13:2187
Stephenson LE, Bebb DG, Reimer RA, Culos–Reed SN (2009) Physical activity and diet behaviour in colorectal cancer patients receiving chemotherapy: associations with quality of life. BMC Gastroenterol 9:60
Meyerhardt JA, Niedzwiecki D, Hollis D, Saltz LB, Hu FB, Mayer RJ, Nelson H, Whittom R, Hantel A, Thomas J, Fuchs CS (2007) Association of dietary patterns with cancer recurrence and survival in patients with stage III colon cancer. JAMA 298(7):754–764
Acknowledgments
The authors wish to thank the Consiglio Nazionale della Ricerca (CNR), The Italian Association for Cancer Research (AIRC), The Italian Ministry of Education (Grants COFIN–PRIN), The Region Emilia–Romagna (Screening Project) and The LegaItalianaLottaTumori (LILT), for generous support during the study.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Domati, F., Rossi, G., Benatti, P. et al. Long-term survey of patients with curable colorectal cancer with specific reference to the quality of life. Intern Emerg Med 6, 529–535 (2011). https://doi.org/10.1007/s11739-011-0590-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11739-011-0590-y