Abstract
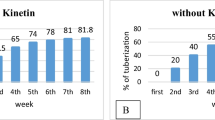
A 2-stage in vitro tuberization process comprising first micropropagation via nodal explants and then tuber induction in the resultant in vitro plantlets was studied using 2 cultivars of potato, Iwa and Daeji. In particular, the effects on both plantlet growth and subsequent in vitro tuberization of Murashige and Skoog (1962) basal medium containing either sucrose or maltose, each at 3 % (w/v), used for micropropagation were investigated. Sucrose and maltose were found to be equally effective in supporting development of vigorous plantlets from the nodal explants of both potato cultivars. Upon transfer to a medium with an optimised level of sucrose (i.e. 8 %, w/v) for in vitro tuberization, only the plantlets previously grown in the sucrose-containing medium were capable of forming more microtubers of the larger size category (greater than 0.5 g). The relative importance of sucrose supply at the mircropropagation stage was further confirmed when the resultant plantlets grown in the 3 % sucrose-containing medium were transferred to an in vitro tuberization medium containing either sucrose or maltose, each at 8 % (w/v). In this experiment, maltose and sucrose had indistingushable effects on in vitro tuberization.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Batty N., Dunwell J.M. 1989. Effect of carbohydrate source on the response of potato anthers in culture. Plant Cell Tissue Organ Cult. 18: 221–226.
El Maataouri M., Espagnac H., Jaber B., Alonso-Lopez A. 1998. Regulation of in vitro callogenesis and organogenesis from Albizzia root explants by carbohydrate source modifications. J. Plant Physiol. 152: 494–501.
Estrada R. Tovar J.H., Dodds J.H. 1986. Induction of in vitro tubers in a broad range of genotypes. Plant Cell Tissue Organ Cult. 7: 3–10.
Fung M.L., Irvine B.R., Barker W.G. 1972. In vitro tuberization of the common potato (Solanum tuberosum L.) is not a response to the osmotic concentration of the medium. Can. J. Bot. 50: 603–605.
Gopal J., Minocha J.L., Dhaliwal H.S. 1998. Microtuberization in potato (Solanum tuberosum L.). Plant Cell Rep. 17: 794–798.
Khuri S., Moorby J. 1995. Investigations into the role of sucrose in potato cv. Estima microtuber production in vitro. Ann. Bot. 75: 295–303.
Leclerc Y., Donnelly D.J., Seabrook J.E.A. 1994. Microtuberization of layered shoots and nodal cuttings of potato: The influence of growth regulators and incubating periods. Plant Cell Tissue Organ Cult. 37: 113–120.
Murashige T., Skoog F. 1962. A revised medium for rapid growth and bioassays with tobacoo tissue cultures. Physiol. Plant. 15: 473–497.
Norgaard J. V. 1997. Somatic embryo maturation and plant regeneration in Albies nordmanniana Lk. Plant Sci. 124: 211–221.
Requin C. 1983. Utilization of different sugars as carbon sources for in vitro cultures of petunia. Z. Pfanzenphysiol. 111: 453–457.
Stickland S.G., Nichol J.W., McCall C.M., stuart D.A. 1987. Effect of carbohydrate source on alfalfa embryogenesis. Plant Sci. 48: 113–121.
Xu X, van Lammeren A.A.M., Vermeer E., Vreugdenhil D. 1998. The role of gibberellin, abscisic acid, and sucrose in the regulation of potato tuber formation in vitro. Plant Physiol. 117: 575–584.
Yu W.C., Joyce P.J., Cameron D.C., McCown B.H. 2000. Sucrose utilization during potato microtuber growth in bioreactors. Plant Cell Rep. 19: 407–413.
Zarrabeitia A., Lejarcegui X., Veramendi J., Mingo-Castel A.M. 1997. Influence of nitrogen supply on micropropagation and subsequent microtuberization of four potato cultivars. Am. Potato J. 74: 369–378.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yoon, K.S., Leung, D.W.M. Relative importance of maltose and sucrose supplied during a 2-step potato microtuberization process. Acta Physiol Plant 26, 47–52 (2004). https://doi.org/10.1007/s11738-004-0043-6
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11738-004-0043-6