Abstract
Locally advanced prostate cancer (PCa) with pathological seminal vesicle invasion (pT3b) is a very-high-risk disease associated with biochemical recurrence (BCR), local recurrence, distant metastases, or mortality following definitive therapies. This study aimed to evaluate the risk factors associated with BCR following robot-assisted radical prostatectomy (RARP) in PCa patients with pT3b. A retrospective multicenter cohort study was conducted on 3,195 patients with PCa who underwent RARP at nine domestic centers between September 2011 and August 2021. Biochemical recurrence-free survival (BRFS) after RARP in PCa patients with pT3b was the primary end-point of the study. The secondary end-point was to determine the association between BCR and covariates. We enrolled 188 PCa patients with pT3b. The median follow-up period was 32.8 months. At the end of the follow-up period, 76 patients (40.4%) developed BCR, of whom 15 (8.0%) were BCR at the date of surgery. The 1-, 2-, and 3-year BRFS rates were 76.4, 65.9, and 50.8%, respectively. Multivariate analysis identified initial prostate-specific antigen level and positive surgical margins (PSM) as significant predictors of BCR in PCa patients with pT3b undergoing RARP. In this study, we investigated the BRFS in PCa patients with pT3b. As PSM was an independent predictor of BCR in PCa patients with pT3b, these patients may require a combination of therapies to improve the BCR.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Locally advanced prostate cancer (PCa) with pathological seminal vesicle invasion (pT3b) [1] is a very high-risk disease associated with biochemical recurrence (BCR), local recurrence, distant metastases, or mortality after definitive therapies [2, 3]. The European Association of Urology guidelines indicate that radical prostatectomy (RP) is an effective treatment modality for selected patients with locally advanced PCa [2]. According to the National Comprehensive Cancer Network (NCCN) guidelines, locally advanced PCa is classified as very-high-risk PCa and is recommended for RP with pelvic lymph node dissection (PLND), androgen deprivation therapy (ADT), or trimodality therapy combining ADT, brachytherapy, and external beam radiation therapy (EBRT) [4]. Although robotic-assisted RP (RARP) is one of the treatment modalities for localized PCa, it may currently be the treatment option for high-risk PCa because of its potential for improved outcomes [2, 4,5,6]. Cancer control using surgical approaches for PCa depends on clinical and pathological factors, such as tumor stage, biopsy Gleason grade group (GG) [7], initial prostate-specific antigen (PSA) level, treatment, extracapsular extension, lymphovascular invasion, positive surgical margins (PSM), and seminal vesicle invasion (SVI) [8]. Among these, PSM is an important predictor of BCR because it may require secondary treatment for PCa [9]. Several recent reports have shown that RARP reduces the incidence of PSM compared with laparoscopic or open RP [6, 9,10,11]. Thus, it has been suggested that RARP may improve the oncologic outcome of PCa with pT3b [7, 12].
Regarding overall survival (OS), RP for locally advanced PCa has been reported to be more useful than other therapies, whereas cancer control for this type of PCa with RP alone is difficult based on the results of various studies [3, 7, 12, 13]. For cancer control of locally advanced PCa, adjuvant EBRT and/or ADT may be used as postoperative therapy in patients with poor pathology [14,15,16]. Several studies have suggested that EBRT and ADT as adjuvant therapy for locally advanced PCa may be beneficial with respect to BCR, whereas in PCa with pT3b, there is no statistical evidence of benefit from adjuvant therapy [14,15,16]. Therefore, it is necessary to identify biomarkers to predict the prognosis of PCa with pT3b and to consider treatment strategies that combine various therapeutic modalities.
The purpose of this study was to evaluate the risk factors associated with the BCR in PCa patients with pT3b and to consider which patients should be treated with multidrug therapy.
Methods
Patients
This study was conducted with the approval of the Institutional Review Board of Gifu University (approval number: 2021-A050) and the institutional review boards of the participating institutions. Since this was a retrospective study, the requirement for informed consent from the patients was omitted. In addition, written consent was not required because the results of the retrospective and observational studies using existing data and other materials had already been published in accordance with the provisions of the Japanese Ethics Committee and Ethics Guidelines. For more information on this study, please visit https://www.med.gifu-u.ac.jp/visitors/disclosure/docs/2021-B039.pdf.
A retrospective multicenter cohort study was conducted on 3,195 patients with PCa receiving RARP at nine institutions between September 2011 and August 2021. Preoperative clinical covariates were collected as follows: age, height, weight, body mass index (BMI), serum prostate-specific antigen (PSA) level, prostate volume (PV), clinical stage according to the American Joint Committee on Cancer eighth edition cancer staging manual [1], biopsy GG group (bGG) [7], NCCN risk stratification [4], Eastern Cooperative Oncology Group performance status [17], history of neoadjuvant therapy, serum albumin level, hemoglobin level, serum C-reactive protein level, neutrophil count, lymphocyte count, thrombocyte count, neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio, and systemic immune-inflammation index. Clinical T stage is generally diagnosed using magnetic resonance imaging, depending on the surgeon and facility, using the American Joint Committee on Cancer eighth edition cancer staging manual [1]. Pathological characteristics were recorded, including T and N stages of the surgical specimen, pathological GG group (pGG) [7], SVI, and PSM statuses.
In this study, RARP was performed in the enrolled patients, and the presence or absence of PLND, its extent, and the method of nerve preservation were determined by the surgeon's preference and each institution's policy. The PLND extent was classified as limited (including only the obturator fossa) and extended (up to the common iliac vessel–ureter crossing, sacral anterior lymph nodes may or may not be present) [18].
Follow-up schedule
Serum PSA levels were evaluated at 3-month intervals following RARP in all patients. Patients whose postoperative serum PSA levels increased to > 0.2 ng/mL were classified as having BCR. If the postoperative PSA level was not ≤ 0.2 ng/mL, the date of RARP was defined as the time of BCR.
Pathological analysis
To evaluate all prostatectomy specimens, we used the whole-mount staining technique and based our diagnosis on the International Society of Urologic Pathology 2005 guidelines [7]. The apex of the prostate was cut perpendicularly to the prostatic urethra. The bladder neck margin was conically cut from the specimen and sectioned vertically. For the remaining prostatic tissue, sections were cut completely along a plane perpendicular to the urethral axis at 3 mm or 5 mm intervals.
Statistical analysis
The biochemical recurrence-free survival (BRFS) was the primary end-point of this study. The secondary end-point was to determine the association between BCR and pre- and postoperative covariates. JMP Pro 16 (SAS Institute Inc., Cary, NC, USA) was used for data analyses. Patient characteristics entered in the study were described using median and interquartile range for continuous variables and using number and percentage for categorical variables. BRFS following RARP was analyzed using the Kaplan–Meier method, and the relationship between BCR and subgroup classification was analyzed using log-rank test. Multivariate analysis was performed using the Cox proportional hazards model. All p-values were two-tailed, and p-values < 0.05 were considered statistically significant.
Results
Patients and characteristics
The demographic data of the enrolled patients are shown in Table 1. Patients who received neoadjuvant or adjuvant therapy and those with an unknown pathologic T stage were excluded from the study. As a result, 188 PCa patients with pT3b met the criteria for this study.
Oncological outcome
For all enrolled patients, the median follow-up was 32.8 months (interquartile range, 15.2–56.5 months). At the end of the follow-up period, 76 patients (40.4%) were diagnosed with BCR, of whom 15 (8.0%) underwent PSA recurrence at the date of surgery. In contrast, 10 patients (5.3%) developed clinical recurrence, and eight had castration-resistant PCa. None of the enrolled patients died of PCa, although one patient (0.5%) died of other causes (details are unknown).
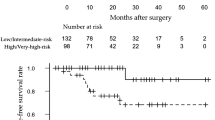
The 1-, 2-, and 3-year BRFS rates for the entire study population were 76.4%, 65.9%, and 50.8%, respectively (Fig. 1). Preoperative BRFS factors are shown in Fig. 2. The 2-year BRFS in patients with bGG ≤ 4 was 69.5% and in those with bGG 5 was 49.0% (P = 0.401, Fig. 2A). For initial PSA values, 2-year BRFS rates were 78.8% and 53.3% for patients with initial PSA values < 10.1 ng/mL and ≥ 10.1 ng/mL, respectively (P = 0.001; Fig. 2B). According to BMI value, the 2-year BRFS rate was 71.1% for < 23.6 kg/m2 and 60.6 ≥ 23.6 kg/m2 (P = 0.042; Fig. 2C). The relationship between the postoperative factors and BRFS is shown in Fig. 3. With respect to PSM, the 2-year BRFS rate was 81.0% in patients with negative surgical margins and 57.4% in those with PSM (P < 0.001; Fig. 3A). For microvascular invasion of prostatectomy specimens, the 2-year BRFS rate was 74.5% in patients with non-microvascular invasion and 47.0% in those with microvascular invasion (P < 0.001; Fig. 3B). Regarding pGG, the 2-year BRFS rate was 69.2% and 56.1% for patients with pGG ≤ 4 and pGG 5, respectively (P = 0.040; Fig. 3C).
The Kaplan–Meier estimates of biochemical recurrence-free survival (BRFS) according to biopsy Gleason grade group (bGG) stratified by 4 and 5 (Fig. 2A), prostate-specific antigen (PSA) levels stratified by a cutoff of 10.1 ng/mL (Fig. 2B) and body mass index (BMI) levels stratified by cutoff of 23.6 (Fig. 2C). Based on bGG, the 2-year BRFS rate was 69.5% in patients with bGG ≤ 4 and 49.0% in those with bGG 5 (P = 0.008; Fig. 2A). According to the initial PSA level, the 2-year BRFS rates were 78.8% and 53.3% in patients with initial PSA levels of < 10.1 ng/mL and ≥ 10.1 ng/mL, respectively (P = 0.001; Fig. 2B). According to BMI, the 2-year BRFS rates were 71.1% and 60.6% in patients with BMI levels of < 23.6 ng/mL and ≥ 23.6 ng/mL, respectively (P = 0.042; Fig. 2C)
The Kaplan–Meier estimates of biochemical recurrence-free survival (BRFS) according to positive surgical margin (Fig. 3A), microvascular invasion stratified positive and negative (Fig. 3B) and pathological Gleason grade group (pGG) (Fig. 3C). Regarding PSM, the 2-year BRFS rate was 81.0% in negative surgical margin (RM-) and 57.4% in those with PSM (RM +) (P < 0.001; Fig. 3A). Based on microvascular invasion on specimen, the 2-year BRFS rate was 74.5% in patients with non-microvascular invasion and 47.0% in those with microvascular invasion (P = 0.014; Fig. 3B). According to pGG, the 2-year BRFS rate was 69.2% in patients with pGG ≤ 4 and 56.1% in those with pGG 5 (P = 0.040; Fig. 3C)
Multivariate analysis revealed that PSA level and PSM were statistically independent factors for predicting BCR in PCa patients with pT3b undergoing RARP (Table 2).
Discussion
Locally advanced PCa with pT3b is a high-risk disease; in addition, this type of PCa is recognized as a cancer prone to local recurrence and distant metastasis [2, 3, 19]. Even today, more than 10% of PCa cases are diagnosed as locally advanced PCa, and local control is recognized as critical for PCa that is non-metastatic or has oligometastases [3, 19, 20]. According to the Southwest Oncology Group (SWOG) 8794 trial, the 5- and 10-year BRFS rates in PCa patients with pT3b were 49 and 22%, respectively [14]. A slight trend toward improved BRFS was observed with adjuvant EBRT, with a 5-year and 10-year BRFS of 62 and 36%, respectively [14]. In this regard, the results were similar to those of patients with negative SVI who received postoperative adjuvant EBRT [14]. These results are similar to those of the present study. Although recent studies reported the outcomes of RP in PCa with pT3b (Table 3), our investigation represents the second study specifically focusing on oncologic outcome of PCa with pT3b performed RARP [3, 21,22,23,24]. Currently, PCa with pT3b is considered difficult to cure by RP alone; therefore, multidisciplinary treatment combining surgery, EBRT, and various drug therapies are being attempted [3, 6].
Recently, it was reported that RARP is likely to be a safe and oncologically effective procedure for PCa, especially in high-risk PCa [20], which allows fine three-dimensional visualization of the entire prostate with a magnified field of view, and a larger area can be resected, which may be associated with a risk of PSM and BCR reduction [6]. The reported rates of PSM and BCR in RARP for locally advanced PCa ranged from 20 to 60% and 18.5% to 28.6%, respectively [6]. Gandaglia et al. reported using the multivariate Cox regression analysis that PSM was a predictor of BCR in patients undergoing RARP for locally advanced PCa (hazard ratio, 6.28; 95% confidence interval CI [1.82–21.4]; p = 0.010) [20]. However, although Ceylan et al. showed that PSM was the only significant negative predictor of BCR (relative risk: 0.163; 95% CI [0.062–0.433]; p < 0.001), they also concluded that PSM did not predict BCR in patients with locally advanced PCa [25]. In this study, PSM was a significant predictor of BCR with respect to RARP for PCa with pT3b (risk ratio, 1.85; 95% CI [1.14–3.02], p = 0.010), suggesting that reducing PSM may improve BCR. In contrast, we thought that reducing PSM using RARP alone may require additional treatment, since it is highly dependent on the skill of the individual surgeon and the progression of PCa in each patient.
Micro-metastasis before RP is considered to be related to postoperative BCR in patients with PCa [26]. Ito et al. reported that microvascular invasion is an independent prognostic factor in patients with PCa who underwent RP [26]. Van den Ouden et al. showed that the relative risk of microvascular invasion is 2.3 for BCR and 2.7 for cancer-specific survival [27]. Hashimoto et al. showed that patients with microlymphatic invasion and a pathologic Gleason score ≥ 4 + 3 tended to experience BCR more frequently postoperatively [28]. In the present study, microvascular and microlymphatic invasion were not independent prognostic factors for BCR, although the former tended to worsen BCR. With regard to the pT3b subclassification, patients with extraprostatic extension (EPE) or microscopic bladder neck invasion (mBNI) were reported to have a significantly higher risk of disease progression than the respective control group, and it has been suggested that EPE and mBNI should be incorporated into the risk classification in the future [29]. Therefore, additional therapy should be considered in cases of postoperative microvascular invasion.
Adjuvant EBRT is often the treatment of choice for cancer control in locally advanced PCa, such as when PSM is present [30]. However, the recommended method, adjuvant EBRT or BCR followed by systemic therapy, is still debated [6]. A meta-analysis examining this debate showed that adjuvant EBRT was superior to the wait-and-see strategy in terms of BRFS, progression-free survival, hormone-free survival, and 10-year metastasis-free survival (MFS), but not OS [31]. Studies on the benefit of adjuvant EBRT in T3 diseases (the European Organisation for Research and Treatment of Cancer 22,911, SWOG8794, and the Arbeitsgemeinschaft Radiologische Onkologie96-02) have suggested a benefit for BRFS [14,15,16]. However, the actual outcomes and benefits of additional treatment, especially for PCa with SVI, have not reached the point of recommendation, as subgroup analyses have failed to show statistically significant differences [3]. Mazzone et al. suggested that patients with pathologic grades (pGG 4/5, SVI, and lymph node metastasis) could benefit from adjuvant EBRT [30]. Swanson et al. reported that postoperative EBRT is useful for BCR but is not associated with MFS and OS in SVI [14]. Adjuvant ADT was reported to be an effective treatment option for patients with pT3N0M0 prostate cancer [32]. However, SVI was found to be an independent predictor of poor prognosis, subsequent hormone refractoriness, and further biochemical progression, even after ADT [32]. Recently, meta-analysis suggested that adjuvant EBRT does not improve event-free survival in patients with localized or locally advanced PCa even if pT3b. Early salvage treatment would seem the preferable treatment policy as it offers the opportunity to spare many men radiotherapy and its associated side-effects [33]. Adjuvant therapy alone could not improve OS in PCa patients with pT3b and PSM. Therefore, it is necessary to consider the other treatment strategy.
If PCa with pT3b could be predicted preoperatively such as nomogram, neoadjuvant therapy may be considered. Neoadjuvant ADT for RP has been reported to significantly contribute to increased surgical margin negative rates and tumor downstaging, but not to improve BRFS or OS [34]. Koie et al. reported a favorable BRFS rate in high-risk patients with PCa who received neoadjuvant chemohormonal therapy consisting of a gonadotropin-releasing hormone agonist or antagonist and low-dose estramustine phosphate prior to RP [35], and Fujita et al. reported that neoadjuvant chemohormonal therapy before RARP may improve BCR [12]. Antagonist and neoadjuvant therapy consisting of low-dose estramustine phosphate reported favorable BRFS rates in high-risk patients with PCa [35]. Fujita et al. reported that the aforementioned neoadjuvant chemohormonal therapy before RARP may improve BCR and OS [36]. Therefore, neoadjuvant chemohormonal therapy may contribute to the improvement of PSM and BCR in PCa patients with pT3b.
This study had several limitations. First, this was a multicenter, retrospective cohort study that may be susceptible to potential selection bias due to differences in diagnostic and surgical approaches among participating centers. Second, because of the short observation period, it was not possible to examine the overall or cancer-specific survival. Therefore, the extent to which SVI and PSM contribute to survival could not be determined in this study. Third, a single pathologist did not reevaluate the prostate biopsy and prostatectomy specimens. However, Ghadjar et al. reported an agreement between central and regional pathology reviews in their central pathology analysis [37], and we believe that diagnostic variability is unlikely to be significantly greater in a multicenter cluster of cases. Finally, since the adjuvant treatment was decided at the discretion of the attending physician and/or at the patient's request, the decision to administer it was not made after taking into account the pathological results and must be followed as having a potentially large bias. Therefore, the results of this study should be cautiously interpreted.
Conclusion
We report the results of our study on PCa patients with pT3b undergoing RARP. This study identified several potential predictors of BCR after RARP in PCa patients with pT3b. In particular, patients with high PSA levels or PSM at initial presentation may require optimal neoadjuvant or adjuvant therapy. The findings of this study need to be validated in a larger prospective trial.
Data availability
Yes.
References
Buyyounouski MK, Choyke PL, McKenney JK, et al (2017) Prostate cancer – major changes in the American Joint Committee on Cancer eighth edition cancer staging manual. CA Cancer J Clin 67:245–253. https://doi.org/10.3322/caac.21391
Mottet N, van den Bergh RCN, Briers E et al (2020) EAU-EANM-ESTRO-ESUR-SIOG guidelines on prostate Cancer-2020 update. part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol 79:243–262. https://doi.org/10.1016/j.eururo.2020.09.042
Poelaert F, Joniau S, Roumeguère T et al (2019) Current management of pT3b prostate cancer after robot-assisted laparoscopic prostatectomy. Eur Urol Oncol 2:110–117. https://doi.org/10.1016/j.euo.2018.05.005
Prostate cancer [NCCN guidelines] ® 2022 (July 2021). Accessed Apr 4
Ilic D, Evans SM, Allan CA et al (2018) Laparoscopic and robot-assisted vs open radical prostatectomy for the treatment of localized prostate cancer: a cochrane systematic review. BJU Int 121:845–853. https://doi.org/10.1016/j.euo.2018.05.005
Saika T, Miura N, Fukumoto T, Yanagihara Y, Miyauchi Y, Kikugawa T (2018) Role of robot-assisted radical prostatectomy in locally advanced prostate cancer. Int J Urol 25:30–35. https://doi.org/10.1111/iju.13441
Epstein JI, Allsbrook WC, Amin MB, Egevad LL, ISUP Grading Committee (2015) The 2005 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma. Am J Surg Pathol 29:1228–1242 https://doi.org/10.1097/01.pas.0000173646.99337.b1.
Pessoa RR, Maroni P, Kukreja J, Kim SP (2021) Comparative effectiveness of robotic and open radical prostatectomy. Transl Androl Urol 10:2158–2170. https://doi.org/10.21037/tau.2019.12.01
Yossepowitch O, Bjartell A, Eastham JA et al (2009) Positive surgical margins in radical prostatectomy: outlining the problem and its long-term consequences. Eur Urol 55:87–99. https://doi.org/10.1016/j.eururo.2008.09.051
Morizane S, Yumioka T, Makishima K et al (2021) Impact of positive surgical margin status in predicting early biochemical recurrence after robot-assisted radical prostatectomy. Int J Clin Oncol 26:1961–1967. https://doi.org/10.1007/s10147-021-01977-x
Fujimura T, Fukuhara H, Taguchi S et al (2017) Robot-assisted radical prostatectomy significantly reduced biochemical recurrence compared to retro pubic radical prostatectomy. BMC Cancer 17:454. https://doi.org/10.1186/s12885-017-3439-6
Fujita N, Koie T, Hashimoto Y et al (2018) Neoadjuvant chemohormonal therapy followed by robot-assisted and minimum incision endoscopic radical prostatectomy in patients with high-risk prostate cancer: comparison of perioperative and oncological outcomes at single institution. Int Urol Nephrol 50:1999–2005. https://doi.org/10.1007/s11255-018-1985-8
Chierigo F, Wenzel M, Würnschimmel C et al (2022) Survival after radical prostatectomy versus radiation therapy in high-risk and very high-risk prostate cancer. J Urol 207:375–384. https://doi.org/10.1097/JU.0000000000002250
Swanson GP, Goldman B, Tangen CM et al (2008) The prognostic impact of seminal vesicle involvement found at prostatectomy and the effects of adjuvant radiation: data from southwest oncology group 8794. J Urol 180:2453–2457. https://doi.org/10.1016/j.juro.2008.08.037
Bolla M, van Poppel H, Tombal B et al (2012) Postoperative radiotherapy after radical prostatectomy for high-risk prostate cancer: long-term results of a randomised controlled trial (EORTC trial 22911). Lancet 380:2018–2027. https://doi.org/10.1016/S0140-6736(12)61253-7
Wiegel T, Bottke D, Steiner U et al (2009) Phase III postoperative adjuvant radiotherapy after radical prostatectomy compared with radical prostatectomy alone in pT3 prostate cancer with postoperative undetectable prostate-specific antigen: ARO 96–02/AUO AP 09/95. J Clin Oncol 27:2924–2930. https://doi.org/10.1200/JCO.2008.18.9563
Oken MM, Creech RH, Tormey DC et al (1982) Toxicity and response criteria of the Eastern cooperative oncology group. Am J Clin Oncol 5:649–655
Clark T, Parekh DJ, Cookson MS et al (2003) Randomized prospective evaluation of extended versus limited lymph node dissection in patients with clinically localized prostate cancer. J Urol 169:145–147. https://doi.org/10.1097/01.ju.0000039647.16278.17
Takagi K, Kawase M, Kato D et al (2022) Robot-assisted radical prostatectomy for potential cancer control in patients with metastatic prostate cancer. Curr Oncol 29:2864–2870. https://doi.org/10.3390/curroncol29040233
Gandaglia G, De Lorenzis E, Novara G et al (2017) Robot-assisted radical prostatectomy and extended pelvic lymph node dissection in patients with locally advanced prostate cancer. Eur Urol 71:249–256. https://doi.org/10.1016/j.eururo.2016.05.008
Inoue T, Kinoshita H, Terada N et al (2015) Evaluation of prognostic factors after radical prostatectomy in pT3b prostate cancer patients in Japanese population. Jpn J Clin Oncol 45:780–784. https://doi.org/10.1093/jjco/hyv077
Lee HJ, Han JH, Lee DH et al (2016) Does bilateral seminal vesicle invasion at radical prostatectomy predict worse prognosis than unilateral invasion among patients with pT3b prostate cancers? Int J Urol 23:758–763. https://doi.org/10.1111/iju.13144
Kim JK, Lee HJ, Hwang SI, Choe G, Hong SK (2020) Prognostic value of seminal vesicle invasion on preoperative multiparametric magnetic resonance imaging in pathological stage T3b prostate cancer. Sci Rep 10:5693. https://doi.org/10.1038/s41598-020-62808-z
Suh J, Jeong IG, Jeon HG et al (2020) Long-term oncologic outcomes of robot-assisted versus open radical prostatectomy for prostate cancer with seminal vesicle invasion: a multi-institutional study with a minimum 5-year follow-up. J Cancer Res Clin Oncol. https://doi.org/10.1007/s00432-022-04243-3
Ceylan C, Tonyali S, Keles I (2016) Impact of positive surgical margin on biochemical recurrence following radical prostatectomy in locally advanced prostate cancer. Kaohsiung J Med Sci 32:514–517. https://doi.org/10.1016/j.kjms.2016.08.007
Ito K, Nakashima J, Mukai M et al (2003) Prognostic implication of microvascular invasion in biochemical failure in patients treated with radical prostatectomy. Urol Int 70:297–302. https://doi.org/10.1159/000070139
Van den Ouden D, Kranse R, Hop WC, Van der Kwast TH, Schroder FH (1998) Microvascular invasion in prostate cancer: prognostic significance in patients treated by radical prostatectomy for clinically localized carcinoma. Urol Int 60:17–24. https://doi.org/10.1159/000030197
Hashimoto T, Nakashima J, Inoue R et al (2020) The significance of micro-lymphatic invasion and pathological Gleason score in prostate cancer patients with pathologically organ-confined disease and negative surgical margins after robot-assisted radical prostatectomy. Int J Clin Oncol 25:377–383. https://doi.org/10.1007/s10147-019-01561-4
Numbere N, Teramoto Y, Gurung PMS, Goto T, Yang Z, Miyamoto H (2022) The clinical impact of pT3a lesions in patients with pT3bProstate cancer undergoing radical prostatectomy. Arch Pathol Lab Med 146:619–625. https://doi.org/10.5858/arpa.2021-0069-OA
Mazzone E, Gandaglia G, Stabile A et al (2022) Not all adverse pathology features are equal: identifying optimal candidates for adjuvant radiotherapy among patients with adverse pathology at radical prostatectomy. J Urol 208:1046–1055. https://doi.org/10.1097/JU.0000000000002855
Shaikh MP, Alite F, Wu MJ, Solanki AA, Harkenrider MM (2018) Adjuvant radiotherapy versus wait-and-see strategy for pathologic T3 or margin-positive prostate cancer: a meta-analysis. Am J Clin Oncol 41:730–738. https://doi.org/10.1097/COC.0000000000000358
Tsurumaki Sato Y, Fukuhara H, Suzuki M et al (2014) Long-term results of radical prostatectomy with immediate adjuvant androgen deprivation therapy for pT3N0 prostate cancer. BMC Urol 14:13. https://doi.org/10.1186/1471-2490-14-13
Vale CL, Fisher D, Kneebone A et al (2020) Adjuvant or early salvage radiotherapy for the treatment of localized and locally advanced prostate cancer: a prospectively planned systematic review and meta-analysis of aggregate data. Lancet 396:1422–1431. https://doi.org/10.1016/S0140-6736(20)31952-8
Gandaglia G, Sun M, Trinh QD et al (2014) Survival benefit of definitive therapy in patients with clinically advanced prostate cancer: estimations of the number needed to treat based on competing-risks analysis. BJU Int 114:E62–E69. https://doi.org/10.1111/bju.12645
Koie T, Mitsuzuka K, Yoneyama T et al (2015) Neoadjuvant luteinizing-hormone-releasing hormone agonist plus low-dose estramustine phosphate improves prostate-specific antigen-free survival in high-risk prostate cancer patients: a propensity score-matched analysis. Int J Clin Oncol 20:1018–1025. https://doi.org/10.1007/s10147-015-0802-y
Fujita N, Koie T, Ohyama C et al (2017) Overall survival of high-risk prostate cancer patients who received neoadjuvant chemohormonal therapy followed by radical prostatectomy at a single institution. Int J Clin Oncol 22:1087–1093. https://doi.org/10.1007/s10147-017-1160-8
Ghadjar P, Hayoz S, Genitsch V et al (2017) Importance and outcome relevance of central pathology review in prostatectomy specimens: data from the SAKK 09/10 randomized trial on prostate cancer. BJU Int 120:E45–E51. https://doi.org/10.1111/bju.13742
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
MK: Data analysis, manuscript writing/editing; SE: Protocol/project development, data collection and management; TT: Data collection and management; TS: Data collection and management; YI: Data collection and management; AN: Data collection and management; MT: Data collection and management; TY: Protocol/project development, data collection and management; KS: Data collection and management; JT: Protocol/project development and supervision; KM: Protocol/project development and supervision; TI: Protocol/project development and supervision; HK: Protocol/project development and supervision; KS: Protocol/project development and supervision; FK: Protocol/project development and supervision; SU: Protocol/project development and supervision; TK: Protocol/project development, data management, manuscript writing/editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
Received from the Japanese Ethics Committee and Ethics Guidelines.
Consent to participate
Retrospective study, no consent required.
Consent for publication
Taken from the Institute.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kawase, M., Ebara, S., Tatenuma, T. et al. Clinical factors associated with biochemical recurrence of prostate cancer with seminal vesicle invasion followed by robot-assisted radical prostatectomy: a retrospective multicenter cohort study in Japan (the MSUG94 group). J Robotic Surg 17, 1609–1617 (2023). https://doi.org/10.1007/s11701-023-01567-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11701-023-01567-1