Abstract
Background
Pre-operative diet may play an important role as far as patients’ fitness for surgery, post-operative outcomes, and successful weight loss. Our aim was to compare surgical outcome and weight loss in two groups of patients who were offered two different pre-operative kinds of diet: very low calorie diet (VLCD) and very low calorie ketogenic diet (VLCKD).
Methods
Patients candidate for bariatric surgery (laparoscopic sleeve gastrectomy) were registered and assessed according to pre- and post-diet BMI, operative time, hospital stay, drainage output, and hemoglobin (Hb) levels. Patients’ preference influenced the type of diet.
Results
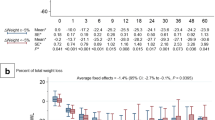
From January to December 2016, 178 patients (139 F and 39 M) were enrolled in this study. The mean age was 43 years. In total, 72 patients were on VLCKD while 106 patients on VLCD. Pre-diet mean BMI was 46.3 ± 6.3 kg/m2 for VLCKD group and 43.1 ± 6.9 kg/m2 for VLCD group, while immediately pre-op BMI were 43.9 ± 5.9 kg/m2 and 41.9 ± 6.8 kg/m2, respectively. Drainage output and hemoglobin levels after surgery resulted significantly correlated with diet induced BMI reduction (141.2 ± 75.8 vs. 190.7 ± 183.6 ml, p = 0.032; 13.1 ± 1.2 vs. 12.7 ± 1.5 g/l, p = 0.04). The percentage of patients requiring a hospital stay longer than anticipated (> 3 days) was 2.8% in the VLCKD group and 10.4% in the VLCD group (p = 0.048).
Conclusions
In our experience, VLCKD showed better results than VLCD on surgical outcome, influencing drainage output, post-operative hemoglobin levels, and hospital stay.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Accurate pre-operative multidisciplinary assessment of bariatric patients plays an important role in improving performance status, surgical outcome, weight loss, and reducing the risk of weight regain. Dietary assessment and pre-operative weight loss has to be considered integrated in the surgical treatment.
Inadequate accessibility to the abdominal cavity due to large fatty liver and visceral obesity is one of the major limiting factors for surgery and has a negative impact on exposition of the surgical field. Reducing hepatomegaly and visceral adipose tissue means having several technical advantages, reducing potential injuries and complications during surgery, and in the peri-operative period.
Very low calorie diet (VLCD) regimens and intragastric balloon placement have already been investigated and have an established role in the pre-operative weight loss [1, 2]. The role of very low calorie ketogenic diet (VLCKD ) is consolidated and increasing in importance for obesity treatment and type 2 diabetes mellitus (T2DM) [3, 4], but their role before bariatric surgery is still less considered. In addition, in the pre-operative period, some issues have to be taken into consideration as the influence of the catabolic state and the oxidative stress induced by ketogenic diets.
Aim
Our purpose was to compare the effects on weight loss between a consolidated pre-operative diet (VLCD) and a new dietetic approach in bariatric surgery (VLCKD). In particular, we concentrated our analysis on the benefits provided by the two regimens on surgical outcomes (mean operative time, hospital stay, drainage output, hemoglobin levels after surgery).
Patients and Methods
From January to December 2016, 178 patients with morbid obesity (139 F, 39 M) underwent laparoscopic sleeve gastrectomy (LSG) at our institution. Eligibility for bariatric surgery was based on standard international guidelines [5]. Seventy-two patients followed a pre-operative VLCKD and 106 a VLCD.
Both regimens were applied for 3 weeks and were proposed according to patients’ preference after a detailed discussion on both diet schemes with the case manager and the bariatric surgeon (Tables 1 and 2).
VLCKD
This regimen contains 1.4 g of proteins per kg of ideal body weight according to Lorenz formula, < 20–30 g carbohydrates, and 15–20 g lipid die. It is divided in three main meals with maximum caloric intake of 700 kcal die. Breakfast and dinner are replaced by a diluted power containing (whey proteins enriched with amino acids).
The lunch includes animal or vegetal derived proteic natural food and 200 g of vegetables. Integration of trace elements diluted in water 2 l during the day is recommended. VLKCD scheme is reported in Table 3.
This regimen is contraindicated in kidney failure with creatinine levels > 1.3 mg/dl, mild-severe liver failure, type 1 diabetes mellitus (T1DM), QT > 0.44 ms, cardiac arrhythmias, autoimmune diseases, hypokalemia, chronic therapies with diuretics as furosemide and hydrochlorthiazide, persistent diarrhea, heart attack, TIA or ICTUS in the last 12 months, pregnancy and breastfeeding, malignances, psychiatric disorders, age > 70 or < 14 years, allergies to milk proteins, and galactosemia.
VLCD
It is structured with three main meals and two snacks with maximum caloric intake of 800 kcal die. It includes proteins 0.8-1.5 g/kg/die; carbohydrates 80 g/die; lipids 15 g die; and 2 l of noncaloric fluid die [6]. VLCD scheme is reported on Table 4.
All patients are invited to send to our Case Manager a certificated weight report (by their General Practitioner for example) once a week before surgery. If their weight loss results are unsatisfactory, patients are contacted and invited to check their weight at our clinic. Patients are informed that weight loss before surgery is mandatory at our center.
Data Collection and Statistical Analysis
Data were collected by the retrospective analysis of a prospectical database. We recorded anthropometric data (weight, pre-diet BMI, pre-operative BMI, %excess BMI loss), comorbidities, parameters related to surgical outcome (mean operative time, hospital stay, drainage output, hemoglobin levels after surgery).
We decided to estimate weight loss by %EBL according to guidelines that encourage the use of BMI and %EBL [7].
Patient characteristics and results were described by statistical frequency, mean, and standard deviation.
Differences between results of VLCD and VLCKD groups were compared by the parametric tests Student’s t and chi-square for numeric data and Fisher exact test for categorical data.
Statistical analysis was performed by the program Statistical Package for Social Sciences, version 18.0 (SPSS, Chicago, IL). p value < 0.05 was considered statistically significant.
Surgical Technique
All the procedures were carried out under laparoscopy, the patient lying supine, surgeon in between the legs, and assistants on both sides. Pneumoperitoneum (16–18 mmHg) was achieved by inserting a Verres needle under the left subcostal margin. A five-trocar approach was used in all patients. The dissection and the division of the vascular supply of the greater curve of the stomach was done with the harmonic scalpel starting at the antrum, 5 cm proximal to the pylorus up to the angle of His, with complete exposure of the left crus. LSG was modeled on a 40-Fr gastric bougie. Multiple fires of an Echelon Endopath were used to resect the stomach. The staple line was tested with methylene blue through the bougie. Perigastric drain was left in place.
Post-operative Management
All patients underwent a fast-track protocol with early mobilization and upper-GI series with water-soluble contrast medium on the first post-operative day (POD). If good gastric emptying through the duodenum was shown, patients were allowed to sip liquids and start a soft diet. Post-operative bleeding was monitored by drainage quality and quantitative output and by lab tests. Drain was usually removed on the 2nd POD. It was left in place according to quality and quantitative output. Hemoglobin levels were checked on the 1st POD and on discharge. Discharge was planned in the 3rd POD.
Results
From December to January 2016, 178 patients (139 F, 39 M) underwent LSG at our institution. Pre-operatively, 72 patients followed a pre-operative VLCKD and 106 a VLCD for 3 weeks.
Anthropometric data for the two groups are reported in Table 1. In the VLCKD group, 16.7% were smokers, and in the VLCD group 19.8%. Hypertension, diabetes mellitus type 2, and OSAS were represented among the VLCKD and VLCD groups in the 44.4% and 50%, 19.4% and 27.4%, and 19.4% and 22.6%, respectively. No significant differences were observed between the two groups in smoking and comorbidity status (Table 2).
Absolute weight loss was significantly better in the VLCKD than in the VLCD group (5.8 ± 2.4 vs. 4.8 ± 2.5 kg; p = 0.008), while no significant differences were observed in %EBMIL (10.4 ± 4.0 vs. 10.0 ± 5.6%; p = 0.658) (Fig. 1).
Mean operative time and hospital stay were slightly but not significantly shorter in the VLCKD group (59.8 ± 18.7 vs. 69 ± 317 min, p = 0.28; 3.0 ± 0.2 vs. 3.2 ± 2.4 days, p = 0.31). The percentage of patients requiring a hospital stay longer than anticipated (> 3 days) was 2.8% in the VLCKD group and 10.4% in the VLCD group (p = 0.048 by chi-square test).
Post-operative hemoglobin levels in g/l were greater in the VLCKD group (13.1 ± 1.2 vs. 12.7 ± 1.49 g/l, p = 0.04). Drainage output resulted also less in the VLCKD group (141.2 ± 75.8 vs. 190.7 ± 183. 6 ml, p = 0.032). No gastric leaks were registered in both groups.
Discussion
It is well established in literature that sensitivity and responsiveness to bariatric surgery as well as surgical and anesthesiological risks are influenced by physical activity and pre-operative diet [8, 9]. Hepatomegaly and visceral obesity especially in the left upper quadrant can affect the exposition of surgical field and increase operative time and conversion rate to laparotomy [10, 11]. According to these evidences, we consider the pre-operative diet part of the surgical act and weight loss in the 3 weeks before surgery is mandatory in our center.
Efficacy of VLCD regimens and BIB as bridging therapy before bariatric surgery is consolidated in literature [1, 2], while the role of VLCKD diets is arising in importance on the obese and diabetic patient, but still under discussion and not well determined in the pre-operative period. Therefore, we decided to test a VLCKD regimen in the bariatric patient prior to surgery considering the better weight loss reported in comparison to regimens with more carbohydrates [12]. The low compliance reported in literature of patients toward VLCD regimens was another point of interest that brought us to consider a VLCKD scheme. Leonetti et al. reported a high compliance rate to a pre-operative diet composed by a sequential scheme of VLCKD and VLCD (96% of patients followed and completed successfully the treatment) [13]. VLCKD can also improve the patient compliance thanks to the anorexigenic effect and hunger reduction of ketone bodies.
We did not experience low compliance to both regimens due to our weekly check of patients’ weight curve.
Our data did not document a strong difference on weight loss between the VLCD and the VLCKD groups, especially EBMIL did not result statistically greater in the VLCKD group. Only weight loss in kg resulted greater and statistically significant after VLCKD.
In our experience, the main advantage of VLCKD was not on weight loss, but on parameters strongly related to surgical outcome. Hematic drainage outputs resulted lower and post-operative hemoglobin levels higher, respectively, post VLCKD. Considering that weight loss and mean operative time resulted comparable among the two groups, we can suppose that this advantage was not influenced only by easier surgical maneuvers due to hepatomegaly and visceral adipose tissue reduction. We suppose that VLCKD patients gained a better metabolic and nutritional status influencing tissue healing and response to surgery. More studies on laboratory parameters regarding pre- and post-operative nutritional assessment are needed to support our hypothesis.
Our suspicion is in contrast with the current fear of metabolic derangements and oxidative stress induced by VLCKD and the negative impact of carbohydrate deprivation before surgery [12, 13]. Moreover, the oxidative stress induced by ketogenic regimens is still unclear. The other side effect of protein metabolism, the increase of glomerular pressure, and hyperfiltration was documented only in animal models [14].
The overall significance of our study was limited by the nonrandomized design, by the relatively small number of patients, and by the lack of direct reliable data on the reduction of liver and visceral fat volumes.
A larger randomized study with the assessment of liver and visceral fat volumes by CT could help in the future to support our preliminary findings.
Conclusion
Otherwise, according to the results, diet can be an important instrument in surgeon’s hands because of its direct influence on surgical outcome. VLCKD showed better results than VLCD on drainage output and post-operative hemoglobin levels. Lower bleeding and better hemoglobin levels are related to a better healing and a lower risk of life-threatening complication as gastric leak after LSG [15].
References
Busetto L, Segato G, De Luca M, et al. Pre-operative weight loss by intragastric balloon in super-obese patients treated with laparoscopic gastric banding: a case-control study. Obes Surg. 2004;14:671–6.
Andrianzen Vargas M, Cassinello Fernandez N, Ortega SJ. Preoperative weight loss in patients with indication of bariatric surgery: which is the best method? Nutr Hosp. 2011;26(6):1227–30.
Al Khalifa A, Mathew TC, Al Zaid NS, et al. Therapeutic role of low carbohydrate ketogenic diet in diabetes. Nutrition. 2009;25:1177–85.
Dashti HM, Mathew TC, Khadada M, et al. Beneficial effects of ketogenic diet in obese diabetic subjects. Mol Cell Biochem. 2007;302:249–56.
Fried M, Yumuk V, Oppert J-M, et al. Interdisciplinary European guidelines on metabolic and bariatric surgery. Obes Facts. 2013;6:449–68.
Gilden Tsai A, Wadden TA. The evolution of very low calorie diets: an update and meta-analysis. Obesity. 2006;14:1283–93.
Deitel M, Greenstein JR. Recommendations for reporting weight loss. Obes Surg. 2003;13:159–60.
Gerber P, Anderin c, Thorell A. Weight loss prior to bariatric surgery: an updated review of the literature. Scand J Surg. 2015 Mar;104(1):33–9.
Schiavo L, Sanas A, Scalera G, et al. Why pre-operative weight loss in preparation for bariatric surgery is important. Obes Surg. 2016 Nov;26(11):2790–2.
Fris RJ. Preoperative low energy diet diminishes liver size. Obes Surg. 2004;4(9):1165–70.
Schwartz ML, Drew RL, Chazin Caldie M. Laparoscopic Roux-en-Y gastric bypass: preoperative determinants of prolonged operative times, conversion to open gastric bypasses and post-operative complications. Obes Surg. 2003;13:734–8.
Heisson M, Rolland C, KulKarni U, et al. Systematic review of randomized controlled trials of low carbohydrate vs low fat/low calorie diet s in the management of obesity and its comorbidities. Obes Rev. 2009;10:36–50.
Gustafsson UO, Scott MJ, Schwenk W, et al. Guidelines for perioperative care in elective colonic surgery: Enhanced Recovery After Surgery (ERAS) Society recommendations. Clin Nutr. 2012;31:783–800.
Gupta L, Khandelwal D, Kalra S, et al. Ketogenic diet in endocrine disorders: current perspectives. J Postgrad Med. 2017;63(4):242–51.
Iossa A, Abdelgawad M, Watkins BM, et al. Leaks after laparoscopic sleeve gastrectomy: overview of pathogenesis and risk factors. Langenbeck's Arch Surg. 2016;401(6):757–66.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Statement of Human and Animal Rights
For this type of study, formal consent is not required.
Rights and permissions
About this article
Cite this article
Albanese, A., Prevedello, L., Markovich, M. et al. Pre-operative Very Low Calorie Ketogenic Diet (VLCKD) vs. Very Low Calorie Diet (VLCD): Surgical Impact. OBES SURG 29, 292–296 (2019). https://doi.org/10.1007/s11695-018-3523-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-018-3523-2