Abstract
Background
In bariatric surgery patients, urinary tract infections (UTIs) are one of the most common postoperative infections. In this study, we sought to determine if preoperative patient factors and perioperative processes contribute to an increased risk of UTI.
Methods
A retrospective analysis was performed of patients who underwent bariatric surgery at a single institution between March 2012 and May 2016. Standard protocol was antibiotic prophylaxis with cefazolin. Patients with a penicillin allergy received clindamycin. Urinary catheters were placed selectively. A univariate and multivariate analyses were performed to determine risk factors for patients who developed a UTI within 30 days postoperatively.
Results
Six hundred ninety-four patients (82.7% female) underwent bariatric surgery in the study interval. UTIs were more common in females (4.9 vs. 1.7%, p = 0.12). On univariate analysis age, operative time, length of stay, urinary catheter placement, clindamycin prophylaxis, and revisional surgery were significantly correlated with UTI. A multivariate logistic regression model revealed the risk of UTI increased 5.38-fold [95% confidence interval (CI) 2.41–12.05] with clindamycin use, 6.37-fold [95% CI 2.22–18.18] with revision surgery, and 1.25-fold [95% CI 1.05–1.49] for every 5 years gained in age.
Conclusions
Older age, clindamycin prophylaxis, and revisional procedures are significantly associated with an increased rate of UTI following bariatric surgery. Several identified variables are modifiable risk factors and targets for a quality improvement initiative to decrease the rate of UTI in bariatric surgery patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urinary tract infections (UTIs) are common, representing up to 40% of all nosocomial infections impacting 1.7 million patients annually [1]. Previous studies have demonstrated that up to 80% of UTIs are catheter-associated (CAUTI) and an average CAUTI event results in direct and indirect costs of $676 and $2386, respectively [2, 3]. In patients undergoing surgery at an American College of Surgeons National Surgical Quality Improvement Program (ACS-NSQIP) hospital, UTIs account for 16.9% of all post-discharge complications in bariatric surgery patients [4].
Our institution participates in the Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program (MBSAQIP). In 2015, we received a semiannual report that identified our institution as a high outlier for “all occurrence morbidity” following both sleeve gastrectomy and laparoscopic gastric bypass. Analysis determined that greater than 50% of recorded postoperative morbidities were UTIs. We sought to identify modifiable risk factors for UTI that could be addressed in an effort to decrease the rate of this postoperative morbidity.
Materials and Methods
Subjects were identified from our institution’s MBSAQIP database. Variables not available in the MBSAQIP database were captured from the electronic medical record. All patients included had undergone a primary or revisional bariatric surgery between March 2012 and May 2016 at a single academic medical center. The occurrence of a UTI within 30 days of bariatric surgery was the primary outcome, defined as an infection in the urinary tract (kidneys, ureters, bladder, or urethra). Patients were diagnosed with a UTI if their urine culture had > 100,000 colony-forming units per milliliter (cfu/mL) with two or fewer species of organisms and at least one of the following criteria: fever (> 100.4 ° F), urgency, frequency, dysuria, suprapubic tenderness, or costovertebral angle pain or tenderness. A patient that complained of two or more of the above symptoms who was started on antimicrobial therapy prior to attaining a culture was also considered to have had a UTI.
Demographic information, comorbidities, preoperative medications, perioperative events, and perioperative complication data were collected. The results of microbiologic data from urine cultures were recorded. The standard of care for patients included in this analysis included preoperative antibiotic prophylaxis with cefazolin, except in the instance of a penicillin allergy in which case clindamycin was administered most often. Urinary catheters were placed selectively, usually when a long operative time was anticipated and especially for revisional cases. Postoperative urine cultures were obtained when patients complained of symptoms consistent with a UTI or as part of an evaluation for fever.
The following variables were compared between patients with and without a UTI diagnosed within 30 days of surgery: sex, age, body mass index (BMI), diabetes, history of urinary condition (defined as any condition affecting the urinary tract noted as a chronic problem), length of hospital stay, operating room time, urinary catheter use, duration catheter was in place if used, preoperative antibiotic prophylaxis, and the bariatric procedure performed. A linear transformation on age was performed by dividing each patients’ age by five to create interpretable intervals.
All statistical analyses were performed using SPSS, version 21 (IBM Corp.). Categorical variables were analyzed with chi-square and Fisher’s exact tests, whereas continuous variables were analyzed using independent samples t tests or, when data was non-parametric, Mann-Whitney U tests. Predictive variables in univariate analysis were input into multivariate logic regression models in a stepwise fashion. A stratified analysis was also performed on females due to the low number of observed UTIs in males. All analyses were two-sided and p values of < 0.05 were considered significant. Our institutional review board approved this study.
Results
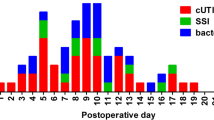
In total, 694 bariatric surgery patients met inclusion criteria. The study cohort included 574 females (82.7%) and 120 males (17.3%). Thirty patients (4.3%) were diagnosed with a postoperative UTI, of which there were 28 females (4.9%) and two males (1.7%). A summary of patient demographic information is displayed in Table 1. The mean age of the study cohort was 44.9 ± 11.7 years and the mean BMI was 46.8 ± 8.2 kg/m2. UTIs were diagnosed an average of 13.1 ± 10.3 days after surgery. In total, 92.3% of pathogens causing UTI were gram negative, with 62.5% growing E. coli. Primary pathogens that resulted in the diagnosis of postoperative UTI for the purposes of this study are illustrated in Fig. 1. Of the 28 UTIs in females, 20 had a positive urine culture with > 100,000 cfu/mL, three patients were treated for symptoms without a culture, two had symptoms and were treated despite a contaminated urine culture, and three patients had UTI symptoms and were treated with an antibiotic and a urine culture that grew < 100,000 cfu/mL of a solitary pathogen.
Age, operating room time, length of stay, urinary catheter placement, clindamycin antibiotic prophylaxis, and bariatric revision procedures were significantly associated with the development of a UTI (p ≤ 0.05) on univariate analysis (Table 1). Multivariate logistic regression revealed clindamycin as preoperative prophylaxis (p ≤ 0.0001), bariatric revisions (p = 0.001), and increased age (p = 0.012) were statistically significant in predicting the development of UTIs. Specifically, the risk of developing a UTI increased 5.38-fold with clindamycin use (95% CI 2.41–12.05), 6.37-fold with bariatric revision procedures (95% CI 2.22–18.18), and 1.25-fold for every 5 years advanced in age (95% CI 1.05–1.49) (Table 2). Multicollinearity was observed between urinary catheter use, operating room time, and revision procedures (Table 3), likely due to the longer operating time for bariatric revision procedures and routine placement of a urinary catheter in these patients. Due to the issue of multicollinearity, urinary catheter placement and operating room time were not included in the final model.
Discussion
We identified an opportunity for improvement in our bariatric surgery program based on the results of our risk adjusted semiannual MBSAQIP report. This retrospective review over the past several years revealed that urinary catheter use, longer operative time, revision procedures, and use of a prophylactic antibiotic other than a cephalosporin were significantly associated with UTI following laparoscopic bariatric surgery.
The association between the use of urinary catheters and UTI is well described. Catheters allow pathogens direct access to the urinary tract [5, 6]. A crystalline biofilm has been demonstrated to form on the indwelling catheter itself [7]. Being highly resistant to the innate and adaptive immune system, the biofilm continues to seed the urine with bacteria and increases resistance to both antibiotics and the patient’s immune response. E.coli is the primary causative agent in over 90% of ambulatory patients diagnosed with a UTI and approximately 50% of nosocomial CAUTIs [8]. CAUTIs, however, do not always present similarly to traditional UTIs. A recent analysis revealed that more than 90% of catheterized patients with 100,000 cfu/mL of bacteria who did not have another confounding site of infection in addition to the urinary tract were asymptomatic [9]. Patients with urinary catheters in place longer than 2 days have been shown to be twice as likely to develop a UTI when compared to patients with catheterization of 2 days or less [10]. Our study did not demonstrate that the duration of catheterization was associated with UTI. As a quality intervention, we have designed a workflow where our patients void immediately prior to going to the operating room for their bariatric surgery and a catheter is avoided when possible. We now only place urinary catheters in patients with an anticipated case duration of > 4 h. In our practice, these prolonged operative times are limited to patients undergoing complex reoperative procedures.
In addition to urinary catheter use, risk factors for UTI identified in previous studies include age [5], longer operative times [11], longer duration of hospital stay [12], and even BMI [13]. We found that the administration of clindamycin as prophylaxis was associated with a significantly increased rate of UTI. These UTIs were mostly from gram-negative organisms. While the primary intent of preoperative antibiotics is to decrease surgical site infections, we found that there was a notable association between the antibiotic class and incidence of UTI. Cefazolin covers many gram-positive and some gram-negative bacteria, whereas clindamycin covers primarily gram-positive bacteria. Since E. coli is both a gram-negative bacteria and is the most common cause of urinary tract infections, clindamycin monotherapy may leave patients susceptible to gram-negative organisms [8, 14]. The Joint Commission has created Surgical Care Improvement Project (SCIP) guidelines for antibiotic prophylaxis prior to gastroduodenal procedures where the gastrointestinal tract is entered (as in bariatric surgery) [12]. For the procedures included in this study, SCIP guidelines recommend prophylaxis with cefazolin. In patients with penicillin allergies, clindamycin or vancomycin combined with an aminoglycoside or a fluoroquinolone is recommended. Failure to follow SCIP guidelines has been demonstrated to be associated with higher rates of surgical site infection [15], but not with UTI. As a quality intervention, we have partnered with our pharmacy colleagues to address these issues. Many patients with a penicillin allergy noted in their medical record can safely receive a cephalosporin as there is a low incidence of cross reactivity between penicillin and cephalosporins [16]. Historically, for patients who have reported an allergic reaction to penicillin that was administered in the 1970s, the allergen may actually have been a contaminant in the penicillin preparation [17, 18]. Furthermore, penicillin-specific IgE diminishes over time and a patient who had an allergic reaction as a child may not have a reaction as an adult [19]. It is likely that administration of a cephalosporin is safe for patients with a reported penicillin allergy with the exception of patients reporting a true anaphylactic reaction. When there is doubt about the extent of the allergy, testing can be considered prior to surgery. For patients truly requiring an alternative antibiotic, a combination of clindamycin and gentamicin provides adequate gram-negative coverage. This regimen is supported by the SCIP guidelines and is also affordable with minimal side effects. As part of our quality improvement initiative, we now more routinely administer a prophylactic cephalosporin to patients with a listed penicillin allergy that have not had an anaphylactic reaction. For those who have a true anaphylactic allergy, we use two antibiotics as described. These decisions are made in close collaboration with our pharmacy colleagues and antimicrobial stewardship groups.
As we have paid increasing attention to our UTIs in the bariatric population, we have come to realize that some patients meet criteria for a UTI prior to surgery. These infections are considered to be present at the time of surgery (PATOS), and for the purposes of our registry are removed from the numerator and denominator when calculating UTI rates. Given the changes in process and protocol described including avoiding urinary catheters and changes to prophylactic antibiotic selection as described, our program returned to the median for all occurrence morbidity and we have not had a UTI within 30 days of surgery in the past year.
This study has several limitations. This is a single-center retrospective review of prospectively collected data. Certain factors that may correlate with UTI could not be attained from the medical record such as who placed the catheter (medical students often learn to place catheters on our patients). Nonetheless, the changes we have made in our protocols including avoiding a urinary catheter when feasible, changes in antibiotic prophylaxis in patients with a reported penicillin allergy, and increased awareness of the possibility a UTI may be present at the time of surgery have led to improved outcomes for our patients. UTIs are a potential complication of bariatric surgery, and focused perioperative interventions can minimize the incidence of this event.
References
Klevens RM, Edwards JR, Richards CL, et al. Estimating health care-associated infections and deaths in U.S. hospitals, 2002. Public Health Rep. 2007;122(2):160–6. https://doi.org/10.1177/003335490712200205.
Saint S. Clinical and economic consequences of nosocomial catheter-related bacteruria. Am J Infect Control. 2000;28(1):68–75. https://doi.org/10.1016/S0196-6553(00)90015-4.
Weber DJ, Sickbert-Bennett EE, Gould CV, et al. Incidence of catheter-associated and non-catheter-associated urinary tract infections in a healthcare system. Infect Control Hosp Epidemiol. 2011;32(08):822–3. https://doi.org/10.1086/661107.
Chen SY, Stem M, Schweitzer MA, et al. Assessment of post-discharge complications after bariatric surgery: a National Surgical Quality Improvement Program analysis. Surgery. 2015;158(3):777–86. https://doi.org/10.1016/j.surg.2015.04.028.
Foxman B. Urinary tract infection syndromes: occurrence, recurrence, bacteriology, risk factors, and disease burden. Infect Dis Clin N Am. 2014;28(1):1–13. https://doi.org/10.1016/j.idc.2013.09.003.
Saint S, Chenoweth CE. Biofilms and catheter-associated urinary tract infections. Infect Dis Clin N Am. 2003;17(2):411–32. https://doi.org/10.1016/S0891-5520(03)00011-4.
Trautner BW, Hull RA, Darouiche RO. Prevention of catheter-associated urinary tract infection. Curr Opin Infect Dis. 2005;18(1):37–41. https://doi.org/10.1097/00001432-200502000-00007.
Jacobsen SM, Stickler DJ, Mobley HLT, et al. Complication catheter-associated urinary tract infections due to Escherichia coli and Proteus mirabilis. Clin Microbiol Rev. 2008;21(1):26–59. https://doi.org/10.1128/CMR.00019-07.
Tambyah PA, Maki DG. Catheter-associated urinary tract infection is rarely symptomatic: a prospective study of 1497 catheterized patients. Arch Intern Med. 2000;160(5):678–82.
Wald HL, Ma A, Bratzler DW, et al. Indwelling urinary catheter use in the postoperative period: analysis of the national surgical infection prevention project data. Arch Surg. 2008;142:551–7.
Alvarez AP, Demzik AL, Alvi HM, et al. Risk factors for postoperative urinary tract infections in patients undergoing total joint arthroplasty. Adv Orthop. 2016; Epub 2016 Nov 28, 2016;1:5. https://doi.org/10.1155/2016/7268985.
Mitchell BG, Ferguson JK, Anderson M, et al. Length of stay and mortality associated with healthcare-associated urinary tract infections: a multi-state model. J Hosp Infect. 2016;93(1):92–9. https://doi.org/10.1016/j.jhin.2016.01.012.
Semins MJ, Shore AD, Makary MA, et al. The impact of obesity on urinary tract infection risk. Urology. 2012;79(2):266–9. https://doi.org/10.1016/j.urology.2011.09.040.
Bratzler DW, Dellinger EP, Olsen KM, et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health-Syst Pharm. 2013;70(3):195–283. https://doi.org/10.2146/ajhp120568.
Smith BP, Fox N, Fakhro A, et al. “SCIP” ping antibiotic prophylaxis guidelines in trauma. J Trauma Acute Care Surg. 2012;73(2):452–6. https://doi.org/10.1097/TA.0b013e31825ff670.
Campagna JD, Bond MC, Schabelman E, et al. The use of cephalosporins in penicillin-allergic patients: a literature review. J Emerg Med. 2012;42(5):612–20. https://doi.org/10.1016/j.jemermed.2011.05.035.
Bernstein IL, Gruchalla RS, Lee RE, et al. Disease management of drug hypersensitivity: a practice parameter. Ann Allergy Asthma Immunol. 1999;83:665–700.
Sogn DD, Evens R, Shepherd GM, et al. Results of the National Institute of Allergy and Infectious Diseases collaborative clinical trial to test the predictive value of skin testing with major and minor penicillin derivatives in hospitalized patients. Arch Intern Med. 1992;152(5):1025–32. https://doi.org/10.1001/archinte.1992.00400170105020.
Wall GC, Peters L, Leaders C, et al. Pharmacist-managed service providing penicillin allergy skin tests. Am J Health Syst Pharm. 2004;61(12):1271–5.
Author information
Authors and Affiliations
Contributions
1. Jon C. Gould was responsible for the concept and design of this study. Furthermore, Jon C. Gould takes full responsibility for the integrity of the work as a whole, from inception to published article.
2. Authors Zachary M. Helmen, Melissa C. Helm, and Alexander Nielsen were responsible for acquisition and interpretation of the data.
3. Authors Zachary M. Helmen, Melissa C. Helm, and Alexander Nielsen were responsible for analyzing and interpretation of the data in depth from the statistical point of view.
4. All authors were involved in drafting the manuscript and revising it critically for important intellectual content and have given final approval of the version to be published. Furthermore, all authors have participated sufficiently in the work to take public responsibility for its content.
Corresponding author
Ethics declarations
Conflict of Interest
JG is a consultant for Torax Medical. The remaining authors declare that they have no competing interests.
Ethical Approval
For this type of study formal consent is not required.
Informed Consent
Does not apply.
Additional information
Presented as a poster at the SAGES 2017 Annual Meeting, Houston, TX, March 2017
Rights and permissions
About this article
Cite this article
Helmen, Z.M., Helm, M.C., Helm, J.H. et al. Predictors of Postoperative Urinary Tract Infection After Bariatric Surgery. OBES SURG 28, 1950–1954 (2018). https://doi.org/10.1007/s11695-017-3095-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-017-3095-6