Abstract
We aim to review the available literature on obese patients treated with ursodeoxycholic acid (UDCA) in order to prevent gallstone formation after bariatric surgery. A systematic literature search was performed in PubMed, Cochrane library, and Scopus databases, in accordance with the PRISMA guidelines. Eight studies met the inclusion criteria incorporating 1355 patients. Random-effects meta-analysis showed a lower incidence of gallstone formation in patients taking UDCA. Subgroup analysis reported fewer cases of gallstone disease in the UDCA group in relation to different bariatric procedures, doses of administered UDCA, and time from bariatric surgery. Adverse events were similar in both groups. Fewer patients required cholecystectomy in UDCA group. No deaths were reported. The administration of UDCA after bariatric surgery seems to prevent gallstone formation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obesity is a rising epidemic with more than 30% of Americans being obese [1]. In this context, bariatric surgery continues to be the main therapeutic mode for a high rate of sustainable weight loss [2,3,4]. However, morbid obesity, along with bariatric surgery and the subsequent weight loss, is associated with an increased risk for the development of gallstones. In fact, a study [5] that incorporated over 90,000 morbid obese patients reported a sevenfold risk of gallstone formation compared with normal weight population. The risk is higher during a period of acute weight loss [6]. Approximately one fourth (25%) of patients carrying gallstones develop complications, such as cholecystitis, cholangitis, or pancreatitis [7], and may require hospitalization, thus representing a major economic burden [8].
Ursodeoxycholic acid (UDCA) is a secondary bile acid that was associated with prevention of gallstone formation in obese patients undergoing acute weight loss. During weight loss, it is believed to decrease bile lithogenicity [9]. In fact, UDCA reduces the intestinal absorption and biliary secretion of cholesterol [10]. The main advantages regarding administration of UDCA are its short-term duration and safety [6].
Currently, there is no consensus regarding gallstone prevention in obese patients undergoing bariatric procedures. Prophylactic cholecystectomy has been proposed as a preventive strategy [11] given that it has not been associated with increased morbidity or mortality [12, 13]. Even though concomitant cholecystectomy at laparoscopic RYGB is much more challenging [11], it has been proven feasible and safe [14, 15], especially in cases of ultrasonography-confirmed gallbladder pathology [14]. In contrast, cholecystectomy performed after RYGB has been associated with increased incidence of adverse events [16]. However, the cost-effectiveness of this option remains debatable. Consequently, UDCA remains an attractive alternative during rapid weight loss.
Nearly 1 decade after the first meta-analysis [17] that assessed the role of UDCA in the prevention of gallstone formation after bariatric surgery, it is necessary to reexamine its effectiveness considering new studies have emerged. For this purpose, we conducted an updated meta-analysis in order to summarize the existing evidence regarding the effectiveness of UDCA for prevention of gallstone formation after bariatric surgery.
Materials and Methods
Search Strategy and Articles Selection
The present systematic review and meta-analysis was conducted in accordance with the protocol agreed by all authors and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [18]. A thorough literature search was performed in PubMed (Medline), Cochrane Central Register of Controlled Studies (CENTRAL), and Scopus (ELSEVIER) databases (last search: February 15, 2017) using the following terms in every possible combination: “ursodesoxycholic acid,” “udca,” “bariatric surgery,” “sleeve gastrectomy,” “sg,” “roux-en-y gastric bypass,” “rygb,” “cholelithiasis,” “gallbladder complication,” “gallbladder disease,” “gallbladder stone,” “gallstone,” and “gallstones.” Inclusion criteria were (1) original reports with > 10 patients, (2) written in the English language, (3) published from 1980 to 2017, (4) conducted on human subjects, and (5) reporting outcomes of UDCA in the prevention of gallstone formation after bariatric surgery.
Two independent reviewers (DEM, VST) extracted the data from the included studies. Any discrepancies between the investigators about the inclusion or exclusion of studies were discussed with the guarantor author (DZ) in order to include articles that best matched the criteria, until consensus was reached. Moreover, the reference lists of all included articles were assessed for additional potentially eligible studies.
Data Extraction
For each eligible study, data were extracted relative to demographics (number of patients, sex, mean age, preoperative body mass index (BMI), comorbidities), dose and duration of UDCA administration, the incidence of cases presented with gallstones, and the mean weight loss along with the incidence of adverse events. Two authors (DEM, VST) performed the data extraction independently and compared the validity of the data. Any discrepancies were discussed with the guarantor author (DZ), until consensus was reached.
Statistical Analysis
Regarding the categorical outcomes, the odds ratio (ORs) and 95% confidence interval (CI) were calculated, based on the extracted data, by means of random-effects model (Mantel-Haenszel statistical method), where the number of studies providing data was sufficient. OR < 1 denoted outcome was more frequent in the control group. Continuous outcomes were evaluated by means of weighted mean difference (WMD) with its 95% CI, using random-effects (inverse variance statistical method) models, appropriately to calculate pooled effect estimates. In cases where WMD < 0, values in the control group were higher.
We chose the random-effects model because we did not expect that all the included studies would share a common effect size. Between-study heterogeneity was assessed through Cochran Q statistic and by estimating I 2 [19] (values of 25, 50, and 75% indicated low, moderate, and high heterogeneity, respectively [20]).
In cases that multiple studies analyzed the same population (i.e., series from the same hospital), only the larger study or the one with the longest follow-up (if the sample was similar) was included in the meta-analysis.
Quality and Publication Bias Assessment
The Newcastle-Ottawa Quality Assessment Scale (NOS) [21] was used as an assessment tool to evaluate non-RCTs. The scale’s range varies from zero to nine stars, and studies with a score equal to or higher than five were considered to have adequate methodological quality to be included. There were six RCTs in the literature to be included. The RCTs were assessed for their methodological quality with the tools that are used to evaluate the risk of bias according to the Cochrane Handbook for Systematic Reviews of Interventions [22]. Two reviewers (DEM, VST) rated the studies independently and final decision was reached by consensus.
The existence of publication bias could not be evaluated using the Egger’s formal statistical test [23] because the number of the included in the analyses studies was not adequate (less than 10), thus compromising substantially the power of the test.
Results
Article Selection and Patient Demographics
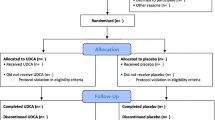
The flow diagram of the search of the literature is shown in Fig. 1 and the research strategy in Table S1. Among the 72 articles in PubMed, CENTRAL, and Scopus that were retrieved, eight comparative studies were included in the qualitative and quantitative synthesis [6, 24,25,26,27,28,29,30]. The study design was retrospective in one study [24], prospective non-randomized in one study [26], and randomized controlled in six studies [6, 25, 27,28,29,30]. The included studies were conducted in Egypt [24], USA [25, 28, 30], France [26], Austria [27], and Canada [6, 29] and were published between 1993 and 2016. The UDCA sample size was 816 patients and ranged from 10 to 247 patients. The total sample size was 1355 patients. Preoperative mean BMI was ≥ 30 kg/m2 in all included patients.
Characteristics of studies comparing the outcomes between patients treated with UDCA and control group are provided in Table 1 . The duration of treatment by UDCA for each study is shown in Table 1. The period of assessment was 3 months in one study [6], 6 months in two studies [28, 30], 12 months in three studies [24,25,26], 18 months in one study [29], and 24 months in one study [27]. The Newcastle-Ottawa rating scale assessment for all studies is shown in Table 1 . The risk assessment according to the Cochrane Handbook for Systematic Reviews of Interventions is shown in Table S2. The summary of the assessment of postoperative gallstone formation and adverse events is shown in Table 2. Pooled ORs, I 2, and p values of heterogeneity for all outcomes are summarized in Table 3 .
Ursodeoxycholic Acid and Incidence of Gallstones Formation in Relation to Different Procedures
In our study, we investigated the postoperative incidence of gallstone formation (Fig. 2). Gallstone formation was assessed with routine postoperative abdominal imaging. Our total analysis showed significantly lower incidence of gallstone formation in patients treated with UDCA (OR 0.25 [95% CI 0.17, 0.38]; p < 0.00001). According to subgroup analysis, the incidence of gallstone formation was lower in UDCA group both in patients treated with SG (OR 0.14 [95% CI 0.05, 0.39]; p = 0.0002) and RYGB (OR 0.25 [95% CI 0.15, 0.44]; p < 0.00001). Outcomes were similar regarding both groups treated with VBG (OR 0.30 [95% CI 0.04, 2.61]; p = 0.28).
Incidence of Gallstones Formation in Relation to Different Doses of UDCA
We performed a subgroup analysis of the postoperative incidence of gallstones formation in relation to different doses of UDCA (Fig. 3). Six studies [24,25,26,27,28, 30] with seven arms assessed the administration of 500–600 mg of UDCA. Patients treated with UDCA reported fewer postoperative cases of gallstone formation (OR 0.21 [95% CI 0.12, 0.38]; p < 0.00001). A two-arm analysis [6, 28] was performed regarding doses of 1000–1200 mg of UDCA. Patients treated with 1000–1200 mg UDCA reported fewer cases of postoperative gallstone disease (OR 0.13 [95% CI 0.13, 0.33]; p = 0.0002).
Incidence of Gallstones Formation in Relation to Time from Surgery
Three studies [24,25,26] assessed the incidence of gallstone formation 6 months after bariatric surgery (Fig. S1). Incidence of gallstones was significantly reduced in the UDCA group (OR 0.11 [95% CI 0.04, 0.26]; p < 0.00001). Five studies [24,25,26,27,28] were included in the subgroup analysis regarding gallstone formation 12 months after surgery. Gallstone formation was significantly lower in UDCA group (OR 0.18 [95% CI 0.12, 0.29]; p < 0.00001).
Adverse Events
No deaths were reported. According to our analysis, the incidence of adverse events was similar between the two groups (OR 1.67 [95% CI 0.67, 4.14]; p = 0.27) (Fig. S2). Considering all included studies, only a few cases of adverse events were reported. The main most commonly reported adverse events were nausea, constipation, vomiting, and medication intolerance.
Incidence of Post-bariatric Surgery Cholecystectomy
We performed a four-arm analysis [6, 26,27,28] in order to assess the incidence of urgent cholecystectomy after bariatric surgery (Fig. S2). According to our analysis, patients treated with UDCA presented lower rate of required cholecystectomy (OR 0.18 [95% CI 0.09, 0.34]; p < 0.00001).
Weight Loss
Bariatric surgery was associated with significant weight loss. The mean weight loss as shown in Table 2 was similar in both groups. The administration of UDCA did not affect the outcomes regarding postoperative mean weight loss.
Compliance
Compliance was reported in all included studies. Compliance for the trial drug regiment was similar between the UDCA and control groups in most studies [24,25,26,27,28] and compliance rate exceeded 80% [24,25,26,27,28]. However, two other studies [29, 30] reported poor compliance. According to Wudel et al. [30], compliance was 28.3% and 31.7% of patients were lost to follow-up. According to Adams et al. [25], compliance was associated with lower rate of gallstone/sludge formation. Sugerman et al. [28] have reported that compliance was increased in patients receiving UDCA 300 mg twice daily compared with those receiving 600 mg once daily.
Quality and Publication Bias
Quality assessment of each non-randomized study according to Newcastle-Ottawa Assessment Scale is shown in Table 1 and for each randomized controlled trial is presented in Table S2 . Heterogeneity was low in all variables except from gallstone formation in patients underwent VBG which associated with a moderate heterogeneity. Egger test was not performed for the outcomes because of the small number of the studies that were included.
Discussion
The increasing popularity of bariatric surgery as the main therapeutic method of morbid obesity [2] along with its effect on multiple metabolic and hormonal parameters [31] has led to an increased effort to improve the efficacy and reduce the complications of the method. This meta-analysis identified eight articles assessing the postoperative administration of UDCA in patients undergoing bariatric surgery, measuring patients’ outcomes and published between 1993 and 2016. Since 2008 when the previous similar meta-analysis [10] was published, new studies have emerged reporting on the administration of UDCA.
The present study demonstrates that the postoperative administration of UDCA is a well-tolerated, feasible, and effective approach in order to reduce postoperative incidence of gallstone formation. Gallstone formation represents a certain threat after bariatric procedures and its incidence ranges from 10 to 38% [3]. Several studies reported an incidence of complicated gallstone formation after bariatric surgery requiring cholecystectomy that ranged between 6.2 and 14.7% [32,33,34]. Furthermore, it has been reported that the postoperative complication risk after cholecystectomy was increased in patients with previous RYGB [16]. In fact, cholecystectomy after bariatric surgery, and especially after RYGB, is a challenging option due to the formation of adhesions and the consequent limitation of laparoscopic visualization of intraabdominal anatomy [11, 28] and has been associated with greater risk of adverse events [35]. The administration of UDCA as a preventive agent regarding gallstone formation during weight loss was first reported by Broomfield et al. [9] in 1998. A large amount of cholesterol can be mobilized from adipose tissue in obese persons undergoing weight loss. UDCA decreases biliary cholesterol and glycoprotein secretion to lower biliary cholesterol saturation and nucleating factors [29].
The included studies reported outcomes regarding patients treated with different surgical procedures. In fact, our findings showed significantly lower incidence of gallstone formation after SG and RYGB in patients treated postoperatively with UDCA compared to the control group. In contrast, outcomes were similar for both groups treated with VBG. However, the study of Wudel et al. [30] that was included in the two-arm analysis assessing the VBG is associated with high proportion of patients lost to follow-up (31.7%), along with poor compliance (28%), thus posing a certain limitation. In addition, in the second study included in the same subgroup analysis [6], the incidence of gallstone formation after VBG was lower in the UDCA group.
Another important aspect that our study examined was the impact of the different administered doses of UDCA on gallstone formation incidence. Doses of 500–600 mg UDCA and 1000–1200 mg UDCA were effective in reducing gallstone formation incidence. No significant difference was reported when doses of 250 mg twice and 500 mg once were compared. According to Coupaye et al. [26], 500 mg of UDCA once daily was more efficient to prevent gallstone disease after SG compared with RYGB, thus proposing different response of patients undergoing RYGB to certain dosages of UDCA. A possible explanation could be the differences in postoperative bile acid profile between the two procedures. Furthermore, according to our subgroup analysis, the incidence of gallstone disease was lower in patients treated with UDCA after 6 and 12 months.
Safety is a crucial parameter when comparing two different clinical practices. According to our meta-analysis, the administration of UDCA is a safe treating modality. No deaths were reported and the average incidence of adverse events was low in all included studies. The main adverse events were constipation, nausea, vomiting, and inconvenience. In addition, fewer patients required urgent cholecystectomy in UDCA group compared to the control group.
There was no significant difference between the two groups regarding weight loss. In all included studies, patients in both groups presented similar weight loss. Further meta-analysis could not be performed due to the diversity of the available data.
The compliance to treatment is another significant parameter that was examined in the present study. In fact, the increased compliance rate has been associated with lower risk of postoperative gallstone formation [25]. The compliance rate in UDCA group was over 80% and similar to control group in most studies [24,25,26,27,28]. Only two older studies reported low compliance rate [29, 30]. According to another study [28], the administration of UDCA 300 mg twice daily instead of 600 mg once daily was associated with increased compliance. Furthermore, it has been found that when the administered dose of UDCA is higher than 1000 mg, the compliance is lower [28, 29].
This meta-analysis demonstrates the safety and feasibility of UDCA in the prevention of gallstone formation after bariatric surgery. In fact, fewer patients in UDCA group required urgent cholecystectomy. According to our findings, administration of either 500–600 mg of UDCA once or 250–300 mg twice daily for a time period of 6 months was associated with reduced risk of postoperative risk of gallstone disease. In contrast, administrated dosages that exceed 1000 mg were associated with poor compliance.
The limitations of this meta-analysis reflect the limitations of the studies included. Six studies [6, 25, 27,28,29,30] (75%) were RCTs. There was one retrospective study [24] and one prospective non-randomized trial [26]. The inclusion of two non-randomized prospective trials poses a certain limitation in this study. Furthermore, the small number of the included studies represents another limitation.
On the other hand, the strengths of this study are (1) the clear data extraction protocol, (2) the well-specified inclusion-exclusion criteria, (3) the search in three different databases, (4) the quality assessment of the included studies, and (5) the detailed presentation of the results of data extraction and analysis.
Conclusion
This meta-analysis identified eight unique peer-reviewed studies assessing the efficiency and safety of administering UDCA in order to prevent gallstone formation after bariatric surgery. These studies suggest that administration of 500–600 mg of UDCA for a period of 6 months was associated with reduced incidence of gallstone formation. Moreover, fewer patients in the UDCA group required urgent cholecystectomy. There was no significant difference between the two groups regarding the %EWL and BMI reduction after 6 and 12 months. No deaths were reported. These results should be interpreted with caution due to the small number of the included studies. Given the latest reports regarding the effect of bariatric surgery on gut microbiota, metabolic profile, and diabetes remission [31], we suggest a new randomized controlled study to investigate the possible effect of UDCA on these parameters, along with the long-term incidence of gallstones and their complications.
References
Go AS, Mozaffarian D, Roger VL, et al. Executive summary: heart disease and stroke statistics—2013 update: a report from the American Heart Association. Circulation. 2013;127:143–52.
Colquit JL, Picot J, Loveman E, et al. Surgery for obesity. Cochrane Database Syst Rev. 2009;2:CD003641.
Magouliotis DE, Tasiopoulou VS, Sioka E, et al. Robotic versus laparoscopic sleeve gastrectomy for morbid obesity: a systematic review and meta-analysis. Obes Surg. 2017;27:245. https://doi.org/10.1007/s11695-016-2444-1.
Magouliotis DE, Tasiopoulou VS, Svokos AA, et al. One-anastomosis gastric bypass versus sleeve gastrectomy for morbid obesity: a systematic review and meta-analysis. Obes Surg. 2017;27(9):2479–87. https://doi.org/10.1007/s11695-017-2807-2.
Stampfer MJ, Maclure KM, Colditz GA, et al. Risk of symptomatic gallstones in women with severe obesity. Am J Clin Nutr. 1992;55:652–8.
Worobetz LJ, Inglis FG, Shaffer EA. The effect of ursodeoxycholic acid therapy on gallstone formation in the morbidly obese during rapid weight loss. Am J Gastroenterol. 1993;88(10):1705–10.
Friedman GD. Natural history of asymptomatic and symptomatic gallstones. Am J Surg. 1993;165:399–404.
Everhart JE, Ruhl CE. Burden of digestive diseases in the United States part III: liver, biliary tract, and pancreas. Gastroenterology. 2009;136:1134–44.
Broomfield PH, Chopra R, Sheinbaum RC, et al. Effects of ursodeoxycholic acid and aspirin on the formation of lithogenic bile and gallstones during loss of weight. N Engl J Med. 1998;319(24):1567–72.
Salvioli G, Igimi H, Carey MC. Cholesterol gallstone dissolution in bile: dissolution kinetics of crystalline cholesterol monohydrate by conjugated chenodeoxycholate-lecithin and conjugated ursodeoxycholate-lecithin mixtures—dissimilar phase equilibria and dissolution mechanisms. J Lipid Res. 1983;24:701–20.
Sarr MG. Patients developed symptomatic gallstones between 3 and 21 months after Roux-en-Y gastric bypass (RYGB), neither prophylactic cholecystectomy nor treatment with ursodeoxycholic acid is necessary after open RYGB. Surg Obes Relat Dis. 2006;2(2):233.
Kim JJ, Schirmer B. Safety and efficacy of simultaneous cholecystectomy at Roux-en-Y gastric bypass. Surg Obes Relat Dis. 2009;5(1):48–53. https://doi.org/10.1016/j.soard.2008.06.001.
Dorman RB, Zhong W, Abraham AA, et al. Does concomitant cholecystectomy at time of Roux-en-Y gastric bypass impact adverse operative outcomes? Obes Surg. 2013 Nov;23(11):1718–26. https://doi.org/10.1007/s11695-013-1001-4.
Tucker ON, Fajnwaks P, Szomstein S, et al. Is concomitant cholecystectomy necessary in obese patients undergoing laparoscopic gastric bypass surgery? Surg Endosc. 2008 Nov;22(11):2450–4. https://doi.org/10.1007/s00464-008-9769-3.
Weiss AC, Inui T, Parina R, et al. Concomitant cholecystectomy should be routinely performed with laparoscopic Roux-en-Y gastric bypass. Surg Endosc. 2015 Nov;29(11):3106–11. https://doi.org/10.1007/s00464-014-4033-5.
Wanjura V, Sandblom G, Österberg J, et al. Cholecystectomy after gastric bypass-incidence and complications. Surg Obes Relat Dis. 2017 Jun;13(6):979–87. https://doi.org/10.1016/j.soard.2016.12.004.
Uy MC, Talingdan-Te MC, Espinosa WZ, et al. Ursodeoxycholic acid in the prevention of gallstone formation after bariatric surgery: a meta-analysis. Obes Surg. 2008;18:1532–8.
Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009;6:e1000100.
Higgins JPT, Green S. Cochrane handbook for systematic reviews of interventions version 5.1.0 [updated March 2011]. The Cochrane Collaboration 2011 Available from http://www.cochrane-handbook.org.
Borenstein M, Hedges LV, Higgins JPT, et al. Introduction to meta-analysis. West Sussex: Wiley; 2009. p. 119. 283–287
Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010;25:603–5.
H.J. GSE. (2011) Cochrane handbook for systematic reviews of interventions version 5.1.0 [updated March 2011]
Egger M, Davey Smith G, Schneider M, et al. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–34.
Abdallah E, Emile SH, Elfeki H, et al. Role of ursodeoxycholic acid in the prevention of gallstone formation after laparoscopic sleeve gastrectomy. Surg Today. 2016; https://doi.org/10.1007/s00595-016-1446-x.
Adams LB, Chang C, Pope J, et al. Randomized, prospective comparison of ursodeoxycholic acid for the prevention of gallstones after sleeve gastrectomy. Obes Surg. 2016;26:990. https://doi.org/10.1007/s11695-015-1858-5.
Coupaye M, Calabrese D, Sami O, et al. Evaluation of incidence of cholelithiasis after bariatric surgery in subjects treated or not treated with ursodeoxycholic acid. Surg Obes Relat Dis. 2016:00–0. https://doi.org/10.1016/j.soard.2016.11.022.
Miller K, Hell E, Lang B, et al. Gallstone formation prophylaxis after gastric restrictive procedures for weight loss: a randomized double-blind placebo-controlled trial. Ann Surg. 2003;238(5):697–702.
Sugerman HJ, Brewer WH, Shiffman ML, et al. A multicenter, placebo-controlled, randomized, double-blind, prospective trial of prophylactic ursodiol for the prevention of gallstone formation following gastric-bypass-induced rapid weight loss. Am J Surg. 1995;169:91–7.
Williams CN, Gowan R, Perey BJ. A double-blind placebo-controlled trial of ursodeoxycholic acid in the prevention of gallstones during weight loss after vertical banded gastroplasty. Obes Surg. 1993;3:257–9.
Wudel LJ, Wright JK, Debelak JP, et al. Prevention of gallstone formation in morbidly obese patients undergoing rapid weight loss: results of a randomized controlled pilot study. J Surg Res. 2002;102(1):50–6.
Magouliotis DE, Tasiopoulou VS, Sioka E, et al. Impact of bariatric surgery on metabolic and gut microbiota profile: a systematic review and meta-analysis. Obes Surg. 2017; https://doi.org/10.1007/s11695-017-2595-8.
Caruana JA, McCabe MN, Smith AD, et al. Incidence of symptomatic gallstones after gastric bypass: is prophylactic treatment really necessary? Surg Obes Relat Dis. 2005;1:564–7.
Papasavas PK, Gagné DJ, Ceppa FA, et al. Routine gallbladder screening not necessary in patients undergoing laparoscopic Roux-en-Y gastric bypass. Surg Obes Relat Dis. 2006;2:41–6.
Swartz DE, Felix EL. Elective cholecystectomy after Roux-en-Y gastric bypass: why should asymptomatic gallstones be treated differently in morbidly obese patients? Surg Obes Relat Dis. 2005;1:555–60.
Moon RC, Teixeira AF, Du Coin C, et al. Comparison of cholecystectomy cases after Roux-en-Y gastric bypass, sleeve gastrectomy, and gastric banding. Surg Obes Relat Dis. 2014;10(1):64–8.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Magouliotis, D.E., Tasiopoulou, V.S., Svokos, A.A. et al. Ursodeoxycholic Acid in the Prevention of Gallstone Formation After Bariatric Surgery: an Updated Systematic Review and Meta-analysis. OBES SURG 27, 3021–3030 (2017). https://doi.org/10.1007/s11695-017-2924-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-017-2924-y