Abstract
Background
Postoperative nausea and vomiting (PONV) is common with bariatric surgery. We examined the PONV rate in bariatric surgical patients who received triple antiemetic prophylaxis (dexamethasone, droperidol, and ondansetron) with and without antiemetic aprepitant.
Methods
Medical records of female patients undergoing laparoscopic bariatric surgery from January 1, 2014, to July 28, 2016, were reviewed for PONV episodes during 48 postoperative hours.
Results
In total, 338 patients received triple antiemetic, of whom 172 (51%) also received aprepitant. Rates of PONV in the postanesthesia care unit (PACU) among patients with and without aprepitant therapy were 11 vs 17% (P = .09). Within 1 h after PACU discharge, fewer patients in the aprepitant group had PONV (19 vs 31%; odds ratio [OR] [95% CI], 0.5 [0.30–0.80]; P = .007). During the first 48 postoperative hours, PONV rates were similar between the groups (68 and 66%; P = .73), but fewer emesis episodes occurred in the aprepitant group (6 vs 13%; OR [95% CI], 0.45 [0.21–0.95]; P = .04). Analyses were also performed with a subset of patients matched on propensity for receiving aprepitant. In this subset, OR estimates quantifying aprepitant effect on PONV were similar to those obtained from multivariable regression analyses.
Conclusion
Addition of aprepitant to a multimodal antiemetic prophylactic regimen may be associated with significant reduction of PONV during early recovery and potentially with reduced incidence of vomiting during the first 48 postoperative hours. The high PONV rate in the first 48 postoperative hours is suggestive that introduction of scheduled anti-PONV prophylactic treatment may be desirable.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Postoperative nausea and vomiting (PONV) is common after laparoscopic bariatric surgery [1, 2]. Some characteristics of the surgical patients—female sex, nonsmoker status, and use of opioid medications—pose a cumulative risk of high PONV rates [3, 4]. PONV has adverse implications, such as intolerance to oral intake, dehydration, electrolyte imbalance, acute kidney injury [5, 6], pulmonary aspiration, and decreased patient satisfaction [7].
Because of multiple risk factors among patients with laparoscopic bariatric surgery, antiemetic prophylaxis is advocated with multimodal agents rather than a single medication [7, 8]. Our standard bariatric PONV prophylaxis includes a triple antiemetic regimen: droperidol, dexamethasone, and ondansetron. Despite this prophylaxis, the PONV rate continues to be increased for these patients, and we have demonstrated that PONV was the primary cause for delayed postanesthesia discharge [1]. In our practice, these observations have led to the adoption of more aggressive PONV prophylaxis with preoperatively applied transdermal scopolamine patch, intraoperative use of propofol infusion, and more recently, use of the neurokinin-1 (NK-1) inhibitor aprepitant. An NK-1 receptor antagonist, aprepitant, blocks substance P activity in centers of the brain and in viscera associated with nausea and vomiting [9]. It is commonly used for prophylaxis against nausea from chemotherapy [10]. Aprepitant is a superior agent for PONV prophylaxis compared with ondansetron used as a single agent [11]. Since, opioid-induced nausea and vomiting is mediated through up-regulation of NK-1 receptors in cortical neurons, it seems reasonable to consider aprepitant as a useful agent for treatment of PONV due to opioids [12]. Aprepitant has been used successfully in multimodal PONV prophylaxis [13] and has been shown to be effective for patients receiving intravenous patient-controlled analgesia with fentanyl [14].
The aim of this study was to test the hypothesis that addition of a single preoperative dose of aprepitant to a multimodal prophylactic regimen is associated with additional reduction of PONV among female patients undergoing laparoscopic bariatric surgery.
Methods
This study was approved by the institutional review board on May 5, 2015. Consistent with state statute, the study included only patients who had provided authorization for research use of their health records.
Study Design
This study was a retrospective chart review of postoperative PONV rate among female patients who underwent laparoscopic bariatric surgery. PONV was defined as use of rescue antiemetics or notation of nausea, vomiting, or retching (severe PONV) during phase I postanesthesia recovery in the postanesthesia care unit (PACU) at 1 h after discharge from the recovery room (PACU + 1 h). This approach accounted for the emetogenic effect of patient transport to the ward also during the first 24 (PACU + 24 h) and 48 h (PACU + 48 h) following discharge from the PACU. When health records did not contain notation of PONV or when documentation of PONV treatment was missing, we considered the patient PONV-free.
Study Participants
The present study included consecutive adult female patients from January 1, 2014, to July 28, 2016, who underwent laparoscopic bariatric weight loss surgery, received triple antiemetic therapy, were transferred from the operating room to the PACU, and had provided research authorization for use of their health records. As previously described [1], these health records were reviewed for clinical, anesthetic, and postanesthesia variables. We excluded from review all patients who had revision surgery or had the same type of surgery but for reasons other than weight loss. Finally, we included patients with failed laparoscopic banding who were referred for a major bariatric surgery.
Preoperative Treatment
All patients enrolled in the bariatric surgical program are initially evaluated by an endocrinologist. All obesity-associated comorbidities, such as hypertension and diabetes mellitus, are treated as indicated. In addition, special attention is directed toward detection of obstructive sleep apnea (OSA), and depending on indications, a pulmonologist conducts evaluation and orders either overnight pulse oximetry or polysomnography. All patients who have no indication for formal OSA evaluation are screened for OSA the day of surgery using Flemons criteria [15]. If needed, patients have psychological or psychiatric evaluation, or both, and if smokers, receive a smoking cessation program.
Preoperative Medications
Standard preoperative order consists of 1000 mg acetaminophen orally. Recognition that the bariatric surgical population is at high risk for PONV prompted 1 surgeon to start prescribing a single oral 40-mg dose of aprepitant, a change in practice that motivated the present study. Of note, as standard practice, all female patients are counseled to use two forms of birth control, including a barrier or spermicidal method, for the first six postoperative months, which makes interaction between aprepitant and hormonal contraceptives moot.
Anesthetic Management
All bariatric operations are performed with general endotracheal anesthesia. Anesthetic maintenance most commonly includes use of desflurane; nitrous oxide is not used. Because bariatric surgical patients have a high risk of PONV (due to the typical characteristics of age < 50 years, female sex, nonsmoker, and postoperative opioid analgesics administration) [1, 4, 16], use of triple antiemetic prophylaxis (i.e. 0.625 mg droperidol, 4 mg dexamethasone, and 4 mg ondansetron) is strongly encouraged [3, 17]. At the discretion of the supervising anesthesiologist, a scopolamine patch or propofol infusions, or both, are also used for high-risk patients. Rocuronium and vecuronium are the standard neuromuscular blocking drugs, and their effects are universally reversed with neostigmine and glycopyrrolate at the end of the operation. Standard intraoperative opioid management includes fentanyl and hydromorphone. At the end of the procedure, 15 mg ketorolac is administered and port sites are infiltrated with 0.25% bupivacaine.
PACU Management
Registered nurses who have received training in phase I recovery make up the PACU staff, together with a first- or second-year anesthesia resident. The attending anesthesiologist is accessible for any medical issue that requires advanced expertise. Discharge criteria for phase I anesthesia recovery have been described previously [1], but germane to this report is a goal PONV level of mild to none.
Nausea Management
An episode of PONV was identified when a patient required rescue antiemetic therapy or when documentation in the nursing records cited nausea or vomiting. Rescue therapy was administered for patients who had nausea or vomiting, with ondansetron as the first-line agent. For recalcitrant cases, dexamethasone, droperidol, promethazine, or metoclopramide was administered under the guidance of the attending anesthesiologist (during PACU stay) or the surgeon (remainder of hospitalization).
Data Abstraction
The electronic health records of study patients were reviewed for demographic information, American Society of Anesthesiologists physical status score, body mass index, smoking status (current smoker), surgical duration, type of bariatric procedure, and perioperative medications, including antiemetic use and opioid dose. Intraoperative opioid administration was converted to intravenous morphine equivalents. The postoperative records were reviewed for occurrence of PONV or emesis, or both.
Statistical Analysis
Data are summarized as mean and standard deviation (SD) for continuous variables and as frequency count and percentage for categorical variables. Baseline characteristics were compared between the patient group that received aprepitant and the patient group that did not have it. Two-sample t test analyzed continuous variables, and Fisher exact test analyzed categorical variables. The frequencies of PONV at PACU, PACU + 1 h, PACU + 24 h, and PACU + 48 h and of emesis at any time during the first 48 postoperative hours were analyzed with logistic regression. A propensity-matched analysis was also performed because many baseline patient characteristics differed significantly between the patient group that received aprepitant and the group that did not. To calculate propensity scores, a logistic regression model was fit with aprepitant use as the dependent variable. Patients who received aprepitant were matched 1 to 1 with patients who did not receive it, on the basis of logit of the propensity score (±0.2 SDs). Balance between propensity-matched groups was assessed by calculating standardized differences. For the propensity-matched sets, PONV and emesis outcomes were analyzed using generalized estimating equations with robust standard error estimates. For each outcome, findings are summarized by presenting the odds ratio (OR) and corresponding 95% confidence interval (CI) for aprepitant use. Two-sided tests were used, and P ≤ .05 denoted statistical significance. Analyses were performed with statistical software (SAS version 9.4; SAS Institute Inc.).
Results
Study Cohort
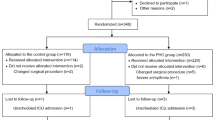
During the study time frame, 338 women underwent laparoscopic bariatric surgery; most (n = 257, 76%) underwent malabsorptive procedures of the 247 laparoscopic gastric bypass and 10 biliopancreatic diversion and duodenal switch procedures. In total, 81 patients (24%) had restrictive procedures (19 gastric banding and 62 sleeve gastrectomy). All patients received intraoperative triple antiemetic regimen, and of these patients, 172 (51%) also received aprepitant. Table 1 compares the characteristics of patients who received aprepitant with patients who did not receive it. A consistent anesthetic management was provided: Desflurane was used in 334 patients (99%) (in aprepitant group, 3 patients had sevoflurane, and in patients who did not receive aprepitant, 1 had isoflurane). All patients received succinylcholine for tracheal intubation followed by vecuronium or rocuronium for muscle relaxation, and all therapies (100%) were reversed with neostigmine (median [interquartile range] dose, 5 mg [5–5]).
Comparison Between Aprepitant and No-Aprepitant Groups
Compared with the patients who did not receive aprepitant, patients who received the agent were younger (P = .049), had longer operations (P = .008), and were more likely to receive preoperative scopolamine patch (P = .047) and intraoperative propofol infusion (P < .001) (Table 1). The PONV rates in the PACU of the aprepitant group and the no-aprepitant group were 11 and 17% (P = .09) (Table 2). Within 1 h after PACU discharge, fewer patients in the aprepitant group had PONV (19 vs 31%; OR [95% CI], 0.5 [0.30–0.80]; P = .007). However, the first 48 h after PACU discharge showed that the percentage of patients with PONV was similar between the two groups (68 and 66% in aprepitant group and no-aprepitant group; P = .73) (Fig. 1). The cumulative episodes of emesis and vomiting through 48 postoperative hours were fewer in the aprepitant group than the no-aprepitant group (6 vs 13%; OR [95% CI], 0.45 [0.21–0.95]; P = .04). The Appendix summarizes the number of patients and types of antiemetics administered at various postoperative times.
Because of imbalance in baseline variables (Appendix Table 3) between patients who did and did not receive aprepitant, an analysis was performed for a subset of patients matched on the propensity for receiving aprepitant. All characteristics listed in Table 1 are included as explanatory variables for propensity matching. From this propensity-matched analysis, OR estimates quantifying the effect of aprepitant on PONV were similar to those obtained from unadjusted analyses.
Discussion
In the present study, we found that addition of a single 40-mg dose of aprepitant to a multimodal antiemetic regimen was associated with reduction of PONV during early recovery (PACU + 1 h) and potentially with reduced incidence of vomiting during the first 48 postoperative hours. The effect of aprepitant in combination with triple antiemetic therapy in attenuating PONV compared with triple antiemetic therapy alone did not persist beyond the initial recovery period, with high cumulative rates of PONV observed at 48 h after PACU discharge.
Several studies have examined the role of aprepitant on PONV reduction—specifically on bariatric, plastic, gynecologic, and neurosurgical procedures [2, 13, 14, 18]. The studies uniformly report that aprepitant was more effective in preventing postoperative vomiting than reducing nausea scores. These observations from single-center studies agree with the results of large, multicenter double-blind trials reporting that selective NK-1 receptor antagonists (aprepitant and rolapitant) were superior to ondansetron for prevention of vomiting in the first 24 and 48 postoperative hours, but no significant differences were observed between aprepitant and ondansetron for nausea control [11, 19]. In the present study, we found immediate postoperative reduction of PONV in patients who received aprepitant and evidence of association with a reduced rate of vomiting during the first 48 postoperative hours.
In our study population, 67% of patients had PONV within 48 postoperative hours. This high incidence of PONV may reflect that the cohort was limited to female and mostly nonsmoking patients (>95%)—two characteristics highly associated with increased PONV risk [20]. A reduction in PONV may be expected with aggressive antiemetic prophylaxis, and indeed, the PONV rate in PACU was 11 and 19% in aprepitant versus no-aprepitant groups, which reflect the protective effect of our intense perioperative antiemetic prophylactic regimen. It further may suggest the presence of a cumulative antinausea effect with increasing number of antiemetic agents used. This rate is lower than our previous bariatric study, where the PONV rate in the PACU was 25% and was the primary reason for delayed PACU discharge [1]. By comparison, however, in the present study where all patients received triple antiemetic prophylaxis, only 42.3% of patients received triple antiemetics [1].
In the present study, the rate of PONV within 1 h after PACU discharge increased to 19% for aprepitant recipients and to even more for patients who did not receive aprepitant (31%). These findings are suggestive of a fading effect of single prophylactic doses of antiemetics that resulted in an increase in PONV during transition from an undisturbed state in the PACU to a more active period associated with patient motion during transport. Subsequently, the effect of the single dose of perioperative antiemetics faded, culminating in 67% of patients reporting PONV by 48 h. This high rate may be explained by the fact that in our practice, antiemetics are administered on an as-needed basis. This high rate suggests the need for practice improvement. For example, these patients may benefit from more intense and structured postoperative antiemetic management.
Limitations
This report has all the limitations inherent in a retrospective study design. Specifically, the definition of the primary end point PONV was determined by the use of rescue medications or notation of its presence in the health records. This approach raises the possibility that a subset of patients may have had untreated PONV, in which case we would underestimate the already high rate of PONV. The possibility also exists that in anticipation of nausea, some patients may have requested treatment, which would overestimate the true rate. Regardless, our rate indicates the clinical reality in our institution but may lack generalizability to other practices. The severity of nausea was not quantified using the formal nausea verbal rating scale, and therefore, association between aprepitant treatment and severity of PONV cannot be studied. Furthermore, no recommendations are available currently for the adjustment of perioperative aprepitant dose based on body weight in adults, and our institution follows the manufacturer recommended low dose (40 mg) for prevention of PONV [21]. Therefore, whether higher doses may be warranted for morbidly obese patients is unknown. Yet, several studies that used larger aprepitant doses of 80 and 125 mg suggested that higher doses may not necessarily provide better outcomes because of a possible ceiling effect [22]. Finally, our study cannot conclude about the size of the effect of aprepitant as a sole agent, but rather, it suggests that four antiemetics may have a stronger effect on prevention of PONV than three antiemetics.
Conclusions
In this study, we found that the addition of aprepitant to a multimodal antiemetic regimen was associated with a significant reduction of PONV during early recovery and potentially with a reduced incidence of vomiting during the first 48 postoperative hours. Regardless of antiemetic treatment used, a high cumulative rate of PONV was observed in bariatric patients at 48 h after PACU discharge, suggesting that scheduled anti-PONV treatment, rather than rescue therapy, may be a more desirable approach in this patient population.
Change history
11 February 2019
In the section ?Discussion? on page 41, the second sentence in the second paragraph in the right-hand column should read as follows.
11 February 2019
In the section ?Discussion? on page 41, the second sentence in the second paragraph in the right-hand column should read as follows.
11 February 2019
In the section ���Discussion��� on page 41, the second sentence in the second paragraph in the right-hand column should read as follows.
11 February 2019
In the section ���Discussion��� on page 41, the second sentence in the second paragraph in the right-hand column should read as follows.
11 February 2019
In the section ���Discussion��� on page 41, the second sentence in the second paragraph in the right-hand column should read as follows.
Abbreviations
- CI:
-
Confidence interval
- NK-1:
-
Neurokinin-1
- OR:
-
Odds ratio
- OSA:
-
Obstructive sleep apnea
- PACU:
-
Postanesthesia care unit
- PONV:
-
Postoperative nausea and vomiting
- SD:
-
Standard deviation
References
Weingarten TN, Hawkins NM, Beam WB, et al. Factors associated with prolonged anesthesia recovery following laparoscopic bariatric surgery: a retrospective analysis. Obes Surg. 2015;25(6):1024–30.
Sinha AC, Singh PM, Williams NW, et al. Aprepitant’s prophylactic efficacy in decreasing postoperative nausea and vomiting in morbidly obese patients undergoing bariatric surgery. Obes Surg. 2014;24(2):225–31.
Gan TJ, Diemunsch P, Habib AS, et al. Society for Ambulatory Anesthesia. Consensus guidelines for the management of postoperative nausea and vomiting. Anesth Analg. 2014;118(1):85–113.
Apfel CC, Laara E, Koivuranta M, et al. A simplified risk score for predicting postoperative nausea and vomiting: conclusions from cross-validations between two centers. Anesthesiology. 1999;91(3):693–700.
Moon HY, Baek CW, Choi GJ, et al. Palonosetron and aprepitant for the prevention of postoperative nausea and vomiting in patients indicated for laparoscopic gynaecologic surgery: a double-blind randomised trial. BMC Anesthesiol. 2014;14:68.
Weingarten TN, Gurrieri C, McCaffrey JM, et al. Acute kidney injury following bariatric surgery. Obes Surg. 2013;23(1):64–70.
White PF, O’Hara JF, Roberson CR, et al. The impact of current antiemetic practices on patient outcomes: a prospective study on high-risk patients. Anesth Analg. 2008;107(2):452–8.
Apfel CC, Korttila K, Abdalla M, et al. Impact investigators. A factorial trial of six interventions for the prevention of postoperative nausea and vomiting. N Engl J Med. 2004;350(24):2441–51.
Leslie RA. Neuroactive substances in the dorsal vagal complex of the medulla oblongata: nucleus of the tractus solitarius, area postrema, and dorsal motor nucleus of the vagus. Neurochem Int. 1985;7(2):191–211.
Gralla RJ, Roila F, Tonato M, et al. The 2004 Perugia antiemetic consensus guideline process: methods, procedures, and participants. Support Care Cancer. 2005;13(2):77–9.
Gan TJ, Apfel CC, Kovac A, et al. Aprepitant Ponv study group. A randomized, double-blind comparison of the NK1 antagonist, aprepitant, versus ondansetron for the prevention of postoperative nausea and vomiting. Anesth Analg. 2007;104(5):1082–9.
Smith HS, Smith JM, Seidner P. Opioid-induced nausea and vomiting. Ann Palliat Med. 2012;1(2):121–9.
Habib AS, Keifer JC, Borel CO, et al. A comparison of the combination of aprepitant and dexamethasone versus the combination of ondansetron and dexamethasone for the prevention of postoperative nausea and vomiting in patients undergoing craniotomy. Anesth Analg. 2011;112(4):813–8.
Lee SJ, Lee SM, Kim SI, et al. The effect of aprepitant for the prevention of postoperative nausea and vomiting in patients undergoing gynecologic surgery with intravenous patient controlled analgesia using fentanyl: aprepitant plus ramosetron vs ramosetron alone. Korean J Anesthesiol. 2012;63(3):221–6.
Flemons WW. Clinical practice: obstructive sleep apnea. N Engl J Med. 2002;347(7):498–504.
Weingarten TN, McGlinch BP, Liedl L, et al. Intranasal nicotine increases postoperative nausea and is ineffective in reducing pain following laparoscopic bariatric surgery in tobacco-naive females: a randomized, double blind trial. Obes Surg. 2015;25(3):506–13.
Weingarten TN, Bergan TS, Narr BJ, et al. Effects of changes in intraoperative management on recovery from anesthesia: a review of practice improvement initiative. BMC Anesthesiol. 2015;15:54.
Vallejo MC, Phelps AL, Ibinson JW, et al. Aprepitant plus ondansetron compared with ondansetron alone in reducing postoperative nausea and vomiting in ambulatory patients undergoing plastic surgery. Plast Reconstr Surg. 2012;129(2):519–26.
Gan TJ, Gu J, Singla N, et al. Rolapitant investigation group. Rolapitant for the prevention of postoperative nausea and vomiting: a prospective, double-blinded, placebo-controlled randomized trial. Anesth Analg. 2011;112(4):804–12.
Pierre S, Whelan R. Nausea and vomiting after surgery. Contin Educ Anaesth Crit Care Pain. 2013;13(1):28–32.
Reference withheld for anonymous review.
Singh PM, Borle A, Rewari V, et al. Aprepitant for postoperative nausea and vomiting: a systematic review and meta-analysis. Postgrad Med J. 2016 Feb;92(1084):87–98.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study. This study was approved by the institutional review board on May 5, 2015. Consistent with state statute, the study included only patients who had provided authorization for research use of their health records.
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Therneau, I.W., Martin, E.E., Sprung, J. et al. The Role of Aprepitant in Prevention of Postoperative Nausea and Vomiting After Bariatric Surgery. OBES SURG 28, 37–43 (2018). https://doi.org/10.1007/s11695-017-2797-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-017-2797-0