Abstract
Background
Serum bile acids (BAs) are elevated following bariatric surgery and have emerged as a potential glucose-lowering beneficial factor. The change of BA components and its underlying mechanisms may be of great significance during bariatric surgery. The aim of this study is to investigate the effects of different bariatric procedures on serum BA composition and explore the potential mechanisms using a diabetic rat model.
Methods
Duodenal-jejunal bypass (DJB), sleeve gastrectomy (SG), and sham operation were performed in diabetic rats induced by high-fat diet (HFD) and streptozotocin (STZ). Body weight, food intake, oral glucose tolerance test (OGTT), and insulin tolerance test (ITT) were measured at indicated time points. Serum BAs composition and the expression of cholesterol 7α hydroxylase (CYP7A1), bile acid: CoA synthase (BACS) and bile acid-CoA: amino acid N-acyltransferase (BAAT) at both transcriptional and protein levels in the liver were evaluated at 12 weeks postoperatively.
Results
Compared with sham group, DJB and SG both achieved rapid and sustained improvements in glucose tolerance and insulin sensitivity. They also resulted in increased serum BAs, especially the taurine-conjugated BAs by elevated conjugation. No obvious difference was detected between DJB and SG except that SG achieved decreased weight gain and food intake.
Conclusions
The preferentially elevated serum taurine-conjugated BAs were similar after different bariatric surgeries, and the enhanced conjugation of BAs in the liver might account for the changed serum BAs profiles.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Type 2 diabetes mellitus (T2DM) is a prevalent disease worldwide, which is hard for effective and long-term management [1]. Bariatric and metabolic surgeries have showed rapid and sustained effects in glucose homeostasis, which led them to be standard treatments for diabetic patients with morbid obesity (BMI >35 kg/m2) [2, 3]. Some bariatric surgeries involve the reconstruction of the gastrointestinal tract, such as Roux-en-Y gastric bypass (RYGB) [4], ileal transposition (IT) [5], and duodenal-jejunal bypass (DJB) [6], while others are with the characteristic of changing the volume of the stomach, such as sleeve gastrectomy (SG) [7]. RYGB and SG have been the most widely performed bariatric procedures in the world [8] and both of them could result in dramatic alleviation of T2DM [4, 9], but the underlying mechanisms are still far from completely elucidated.
Recently, it has been reported that serum bile acids (BAs) level is elevated following bariatric surgeries and BAs have emerged as potential glucose-lowering factors [10–13]. BAs are synthesized from cholesterol in the liver by cholesterol 7α hydroxylase (CYP7A1), which is the rate-limiting enzyme of BA biosynthesis. BAs were previously well recognized for their important roles in the absorption of lipids and fat-soluble vitamins in the gut [14, 15]. In addition, BAs could also work as a kind of endocrine factors by activating the farnesoid-X receptor (FXR) and transmembrane G protein-coupled receptor (TGR5), which could result in suppressed gluconeogenesis and lipid de novo synthesis [16], enhanced release of glucagon-like peptide-1 (GLP-1) and energy expenditure [17]. There are more than 20 kinds of BAs, which has diverse effects on metabolism [14]. The changes of BA components and the causes have become hot spots [11, 12] and may be of great significance during bariatric surgery.
DJB is an experimental procedure that was designed by Rubino and Marescaux to investigate the weight-independent anti-diabetic effects of RYGB [18]. Since DJB and SG are the representatives of bypass surgery and volume-limiting surgery separately, in this study, we performed DJB and SG procedures on diabetic rats induced by high-fat diet (HFD) and low dose of streptozotocin (STZ), and analyzed the BA component changes, compared the similarities and differences, and discussed potential mechanisms of diabetes control after different procedures.
Materials and Methods
Animals
All 8-week-old male Wistar rats (200 g on average) were purchased from the Laboratory Animal Center of Shandong University (Jinan, China). The rats were individually housed in independent ventilated cages (IVC) under constant ambient temperature (24–26 °C) and humidity in a 12-h light/dark cycle. T2DM was induced as previously described [19]: The rats were given free access to tap water and fed with HFD (40 % fat, 42 % carbohydrate, 18 % protein, as a total percentage of calorie, Huafukang Biotech, China) for 4 weeks to induce fat accumulation and insulin resistance, and then injected with a low dose of STZ (35 mg/kg) (Sigma, USA) intraperitoneally to induce hyperglycemia. Seventy-two hours after injection, random blood glucose was measured with a glucometer (Roche Diagnostics, Germany) from tail veins. Thirty rats with random blood glucose ≥16.7 mmol/l were considered diabetic and were matched into SG group (n = 10), DJB group (n = 10), and SHAM group (n = 10). All study protocols were approved by the Animal Care and Utilization Committee of Shandong University.
Surgical Procedures
All the rats were fed 10 % Ensure (Abbott, USA) for 2 days and then fasted overnight preoperatively. Anesthesia was achieved with 10 % chloral hydrate (3 ml/kg).
Duodenal-Jejunal Bypass
DJB surgery was performed as previously described [6]: The duodenum was transected at 0.5 cm distal to the pylorus and then the stump was closed by a 7–0 silk suture (Ningbo Medical Needle, China). The jejunum was transected at 10 cm distal to the ligament of Treitz, and the distal end was connected to the proximal duodenum (duodenojejunal anastomosis). After all, the biliopancreatic limb was sutured to the alimentary limb with 15 cm distal to the duodenojejunal anastomosis in a Roux-en-Y fashion.
Sleeve Gastrectomy
SG surgery [7, 20] involved (1) a 4-cm midline epigastric incision to identify the structures; (2) dissecting the gastric omentum and disclosing the gastric cardium, (3) ligating and transecting short gastric vessels, related gastroepiploic vessels, and the branches of left gastric vessels in the greater curvature using 5–0 silk suture (Ningbo Medical Needle, China), (4) removing the gastric fundus and a large portion of the gastric body, and (5) closing the residual stomach using 5–0 silk suture (Ningbo Medical Needle, China).
For rats in the sham group, laparotomy was performed to expose the stomach, esophagus, and small intestine. No other procedure was carried out. Furthermore, operative time was prolonged to induce a comparable degree of anesthetic stress as experienced by the operated rats. The role of sham group was to eliminate the influence of surgical stress anesthesia on experiments. That surgical procedure was also used by other research groups [21, 22].
Oral Glucose Tolerance Test (OGTT)
OGTT was performed preoperatively and at 2 and 12 weeks after surgery. Blood glucose was measured in conscious rats after an overnight fast and at 0, 10, 30, 60, and 120 min after the administration of 1 g/kg glucose by oral gavage.
Insulin Tolerance Test (ITT)
ITT was conducted to evaluate systemic insulin sensitivity preoperatively and at 2 and 12 weeks after operation. After overnight fasting, blood glucose was measured in conscious rats at 0, 10, 30, 60, and 120 min after intraperitoneal injection of human insulin (0.5 IU/kg).
Measurement Protocol of Body Weight, Food Intake, and Blood Sampling
In all groups, body weight and food intake were tracked for 12 weeks after surgery. At 12 weeks postoperatively, the rats were fasted overnight. Blood samples were collected from the retrobulbar venous plexus of rats into chilled EDTA tubes containing a dipeptidyl peptidase IV inhibitor. After centrifugation (3000 rpm) at 4 °C for 15 min, the supernatant was immediately collected. Aliquots of fasting serum samples were immediately analyzed for total bile acids (TBA) by Roche Cobas 8000 system using enzyme cycling method. The rest samples were stored at −80 °C for future analysis.
High-Pressure Liquid Chromatography Coupled with Tandem Mass Spectrometry (HPLC–MS/MS) Detection of Serum Bile Acids
Reverse-phase solid phase extraction (SPE) cartridges (Chromabond C18ec cartridges, Germany) were pre-conditioned by 1 ml methanol and 1 ml 0.05 % formic acid. Serum samples mixed with 1 ml 0.05 % formic acid for 1 min and then were loaded onto the cartridges. 1 ml 0.05 % formic acid and 1 ml methanol water solution (45:55, V/V) were used to rinse successively. After eluted with 1 ml methanol, the BAs were collected and dried under a nitrogen stream at 60 °C. The residue was dissolved in 200 μl mobile phase (ammonium acetate:methanol = 20:80, V/V), of which 5 μl were injected into the HPLC–MS/MS after mechanical stirring.
The chromatographic separation of BAs was carried out on a Zorbax Eclipse XDB-C18 4.6 × 250 mm column (5-μm particle size) fitted on an Agilent 6410 Triple Quadrupole LC/MS system (Agilent Technologies, USA). The column was thermostated at 30 °C. The mobile phase consisted of solvent A (ammonium acetate 5 mmol/L) and solvent B (methanol), using a gradient elution (0–20 min: 70–100 % B) with 8 min of balance back to 70 % B. The injection volume was 5 μl and the flow rate was set at 0.5 mL/min. The mass spectrometry conditions were as follows: ionization mode, ESI-negative; drying gas flow, 12 L/min; nitrogen drying gas temperature, 350 °C; capillary voltage, 4000 V; nebulizer, 35 psi. Data were acquired by the workstation software (version B.01.04, Agilent) in the Multiple Reaction Monitoring (MRM) mode. The reference standards of TαMCA, TβMCA, αMCA, and βMCA were purchased from Steraloids (USA) and the rest were from Sigma Aldrich (USA).
Quantitative Real-Time PCR
Twelve weeks after operation, all the rats were starved overnight and euthanatized. The liver was removed and immediately frozen in liquid nitrogen and stored at −80 °C until analysis. Total RNA was extracted using TRIzol reagent (Invitrogen, USA) The cDNA was synthesized in the presence of random primers (TOYOBO, Osaka, Japan) according to the manufacturer’s protocol. Quantitative real-time reverse transcription polymerase chain reaction (quantitative real-time RT-PCR) was performed in a Light Cycler System (Roche Diagnostics, Mannheim, Germany). Analyses were performed on 1 μg cDNA using the SYBR® Green Real Time PCR Master Mix (TOYOBO, Japan), in a total PCR reaction volume of 10 μl, containing 0.2–0.6 μM of each primer. The following primer pairs were used:
CYP7A1: 5′-TGAAAGCGGGAAAGCAAAGACCAC-3′(forward), 5′-TCTTGGACGGCAAAGAGTCTTCCA-3′(reverse);
BACS: 5′-TTCAGGGACCACTGGACTTCCAAA-3′ (forward), 5′-ACCACATCATCAGCTGTTCTCCCA-3′ (reverse);
BAAT: 5′-TGAAAGCGGGAAAGCAAAGACCAC-3′ (forward), 5′-TCTTGGACGGCAAAGAGTCTTCCA-3′ (reverse);
GAPDH: 5′-TCCCTCAAGATTGTCAGCAA-3′ (forward),
5′-AGATCCACAACGGATACATT-3′ (reverse)
Western Blotting
Liver were homogenized in lysis buffer containing protease and phosphatase inhibitors (BestBio, Shanghai, China) and centrifuged (12,000 rpm, 10 min, 4 °C). The supernatants were extracted and the protein concentration was determined by BCA Kit (Beyotime, Shanghai, China). Equivalent amount of samples were loaded on 8 % SDS-PAGE gels (Beyotime, China) and separated by electrophoresis. Then proteins were transferred onto PVDF membranes (Millipore, USA). After blocked in 5 % fat-free milk for 2 h, the membranes were incubated with primary antibodies to bile acid: CoA synthase (BACS) (Santa Cruz, USA), bile acid-CoA: amino acid N-acyltransferase (BAAT) (Sigma, USA), CYP7A1 and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (Abcam, USA) overnight, followed by incubation in horseradish peroxidase-conjugated secondary antibodies for 60 min. The protein bands were visualized by ECL solution (Beyotime, China), and the band intensity was quantified with the ImageJ software (http://rsb.info.nih.gov/ij; National Institutes of Health, Bethesda, MD, USA).
Abbreviations
alpha muricholic acid (αMCA); beta muricholic acid (βMCA); cholic acid (CA); chenodeoxycholic acid (CDCA); deoxycholic acid (DCA); ursodeoxycholic acid (UDCA);hyodeoxycholic (HDCA); lithocholic acid (LCA); the prefix T indicated conjugation with taurine.
Statistical Analysis
All quantitative data were reported as mean ± standard error. Areas under the curves (AUC) for OGTT (AUCOGTT) and ITT (AUCITT) were calculated by trapezoidal integration. Inter-group comparisons were evaluated using one-way analysis of variance (ANOVA) followed by Bonferroni post hoc comparisons. If data did not have a normal distribution or homogeneity of variance, Kruskal-Wallis H test with Benjamini-Hochberg correction was used. All statistical analyses were performed using SPSS version 20.0, and the threshold of statistical significance was set at P < 0.05.
Results
All the rats were survived until the end of the study.
Body Weight and Food Intake
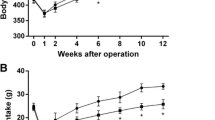
As shown in Fig. 1, body weight and food intake in all groups decreased during the first week postsurgery because of perioperative food restriction and surgical stress, and then gradually increased. Compared with DJB and sham groups, SG group showed significant weight loss at 6 weeks postoperatively (Fig. 1a), and also caused a significant decrease in the food intake of rats at 4 weeks after operation (Fig. 1b). There was no significant difference in body weight and food intake between DJB and sham groups both preoperatively and postoperatively.
Effects of Surgeries on Glucose Homeostasis
No difference in AUCOGTT and AUCITT was observed between the groups preoperatively (Fig. 2). SG and DJB groups both exhibited better glucose tolerance and insulin sensitivity than the sham group at 2 and 12 weeks postoperatively, as exhibited by lower values of AUCOGTT and AUCITT separately (Fig. 2). Notably, the values of AUCOGTT and AUCITT were similar between SG and DJB groups postoperatively.
Effects of Surgeries on Fasting Serum TBA
At 12 weeks after operation, SG and DJB groups exhibited significantly higher fasting serum TBA than sham group (Fig. 3), and there was no significant difference between those.
Effects of Surgeries on Serum BAs Composition
Absolute Value
CA was the most abundant BA species in all three groups. At 12 weeks after operation, all the taurine-conjugated species, plus βMCA, UDCA were higher in both DJB and SG groups than in the sham group (Fig. 4a) and were similar between DJB and SG groups. The SG group demonstrated statistically higher CDCA than sham group.
Percentage
CA accounted for more than 50 % of TBA in sham group while this percentage was less than 35 % in the DJB and SG group at 12 weeks after operation (Fig. 4b). Interestingly, though the absolute value of some BA species elevated, the relative percentage was unchanged at all (Fig. 4b). The percentage of TαMCA, TβMCA, TUDCA, and TCA in DJB and SG groups are still higher than the sham group without obvious differences.
Subgroup Analysis
The absolute value and percentage of conjugated BAs were found elevated in the DJB and SG groups (Fig. 4c, d), and it also trended to be higher in the DJB than in the SG group, but it did not reach statistical difference. No significant change was observed in the absolute value of unconjugated BAs.
Effects of Surgeries on BA Synthesis and Conjugation with Amino Acids in the Liver
Since we have observed increased conjugated BAs expression in the liver, we have investigated the CYP7A1 (Figs. 5a and 6), which can reflect the BAs synthesis, and BACS (Figs. 5b and 6) and BAAT (Figs. 5c and 6), which are important enzymes which can regulate the conjugation with amino acids. DJB and SG groups exhibited lower CYP7A1 and higher BACS and BAAT at both protein and transcriptional level.
Discussion
Numerous studies have showed that bariatric surgeries could achieve weight-independent amelioration of T2DM early after operation [6, 7, 13], and SG [9], instead of DJB [13], could effectively cause long-term reduction of weight and food intake. Our study supports the view and provides further evidence that DJB and SG have no significant difference in the improvement of glucose tolerance and insulin sensitivity by OGTT and ITT.
It was demonstrated that BAs, especially the serum BAs, activate various cell signaling pathways though receptors of extensive tissue distribution [23]. Serum BAs levels mainly reflect BAs synthesis [24], absorption from the intestine and uptake by hepatocytes [25, 26]. Previous studies have showed that serum BAs were elevated following RYGB [10], IT [11], and SG [12], and Ferrannini et al. found that BAs synthesis in the liver was suppressed after RYGB [27]. Our data provided further evidences that DJB and SG could similarly increase BAs, while the rate-limiting enzyme of BA synthesis was both downregulated after operation. In our study, although SG-operated rats underwent less weight gain than DJB-operated rats, these two groups exhibited similar BA profiles. In agreement with our study, Kohli et al. also reported that two sham groups with different feeding regime showed different body weight but comparable BAs [28], suggesting that weight loss might be not a critical reason for BAs changes. In this study, we did not investigate the effects of DJB and SG surgeries on fatty liver diseases of T2DM rats, but Han et al. [13] and Myronovych et al. [12] had respectively showed that DJB and SG could reduce liver fat content and increase serum BAs. We conjecture that improved fatty liver disease might be beneficial for BA metabolism after bariatric surgery. We speculate that for RYGB, IT, and DJB, the reconstruction of gastrointestinal tract results in expedited BAs flow to terminal ileum, and early absorption of BAs, which might account for the elevated serum BAs after operation [26]. For SG [29], accelerated gastric emptying might also play the same role. And, we speculate that the downregulation of CYP7A1 might be the results of negative feedback of increased serum BAs.
The conjugation of BAs with amino acids in the liver could increase the polarity and solubility of these molecules [23]. BAs are mainly conjugated with glycine or taurine in humans, while with taurine in rodents [30, 31]. Taurine is a sulfur amino acid which could be synthesized from cysteine or methionine endogenously or acquired from diet [32]. Researchers have found reduced levels of plasma taurine in obese and diabetic subjects [33] and rodents [34], and taurine supplementation could prevent obesity and improve glucose tolerance [35, 36]. Whether taurine-conjugated BAs have better physiological function is uncertain. In the BA composition analysis, we found the significant elevation of serum taurine-conjugated BAs in DJB and SG group at 12 weeks after operation, regardless of absolute value or percentage. Some species of unconjugated BAs also showed increase in the absolute value but did not have a statistical change except CA in the percentage. Elevated conjugated BAs were also observed after RYGB [27] and IT [28]. Myronovych et al. found that HFD induced obese C57BL/6 mice after vertical sleeve gastrectomy (VSG) had a higher level of TUDCA both in the absolute value and percentage but it is a pity that there is no data of other conjugated BAs [12]. Cummings et al. found no change of circulating BA profiles at 3 months after VSG in UCD-T2DM rats, but taurine-conjugated BAs were increased after a long-term observation [37], which may be related to the difference of animal models. It is noteworthy that Ferrannini et al. found greater increments in unconjugated BAs with increased BA synthesis after Biliopancreatic diversion (BPD) in humans, which does not agree with our results [27]. In addition, our study demonstrated that although there are some specific differences in the surgical procedure, the effect of DJB and SG on serum BAs is similar.
We also investigated the causes of elevated conjugated BAs after DJB and SG. Conjugation of BAs is catalyzed by two separate enzymes. BACS converts BAs to their CoA thioesters, and BAAT, which locates in the peroxisomes, catalyzes the amidation of bile acid-CoA thioester to form taurine conjugate [38–40]. Their relationships with diabetes and bariatric surgery are unsure, but the variants in gene of BACS may be etiological for type 2 diabetes [41]. Unlike the reduction of synthesis, the expression of BACS and BAAT was elevated, which means the conjugation is enhanced, and there was no significant difference between DJB and SG. Like the conjugation, the deconjugation of BAs after bariatric surgeries was unclear either. In the intestine, a significant portion of BAs is deconjugated by gut bacteria in humans [40, 42]. Because of the reduction of synthesis, deconjugated BAs may be the important source of newly taurine-conjugated BAs. BAs transport may also play a critical role in the elevated conjugated BAs. The human apical sodium dependent bile acid transporter (ASBT) transports BAs with a preference for the conjugates over the unconjugated forms [43].
There are some limitations in our study. Serum glycine-conjugated BAs were not evaluated. The measurements of BA synthesis were not systematic and comprehensive that need to be done in the future.
In conclusion, our study demonstrated that both DJB and SG could preferentially elevate the taurine-conjugated BAs, which might be explained by elevated BACS and BAAT.
References
Whiting DR, Guariguata L, Weil C, et al. IDF diabetes atlas: global estimates of the prevalence of diabetes for 2011 and 2030. Diabetes Res Clin Pract. 2011;94(3):311–21.
Buchwald H, Estok R, Fahrbach K, et al. Weight and type 2 diabetes after bariatric surgery: systematic review and meta-analysis. Am J Med. 2009;122(3):248–56. e5.
Sjostrom L. Review of the key results from the Swedish Obese Subjects (SOS) trial—a prospective controlled intervention study of bariatric surgery. J Intern Med. 2013;273(3):219–34.
Schauer PR, Burguera B, Ikramuddin S, et al. Effect of laparoscopic Roux-en Y gastric bypass on type 2 diabetes mellitus. Ann Surg. 2003;238(4):467–84. discussion 84–5.
Wang TT, Hu SY, Gao HD, et al. Ileal transposition controls diabetes as well as modified duodenal jejunal bypass with better lipid lowering in a nonobese rat model of type II diabetes by increasing GLP-1. Ann Surg. 2008;247(6):968–75.
Liu S, Zhang G, Wang L, et al. The entire small intestine mediates the changes in glucose homeostasis after intestinal surgery in Goto-Kakizaki rats. Ann Surg. 2012;256(6):1049–58.
Sun D, Liu S, Zhang G, et al. Sub-sleeve gastrectomy achieves good diabetes control without weight loss in a non-obese diabetic rat model. Surg Endosc. 2014;28(3):1010–8.
Angrisani L, Santonicola A, Iovino P, et al. Bariatric surgery worldwide 2013. Obes Surg. 2015
Himpens J, Dobbeleir J, Peeters G. Long-term results of laparoscopic sleeve gastrectomy for obesity. Ann Surg. 2010;252(2):319–24.
Kohli R, Bradley D, Setchell KD, et al. Weight loss induced by Roux-en-Y gastric bypass but not laparoscopic adjustable gastric banding increases circulating bile acids. J Clin Endocrinol Metab. 2013;98(4):E708–12.
Cummings BP, Bettaieb A, Graham JL, et al. Bile-acid-mediated decrease in endoplasmic reticulum stress: a potential contributor to the metabolic benefits of ileal interposition surgery in UCD-T2DM rats. Dis Model Mech. 2013;6(2):443–56.
Myronovych A, Kirby M, Ryan KK, et al. Vertical sleeve gastrectomy reduces hepatic steatosis while increasing serum bile acids in a weight-loss-independent manner. Obesity (Silver Spring). 2014;22(2):390–400.
Han H, Hu C, Wang L, et al. Duodenal-jejunal bypass surgery suppresses hepatic de novo lipogenesis and alleviates liver fat accumulation in a diabetic rat model. Obes Surg. 2014;24(12):2152–60.
de Aguiar Vallim TQ, Tarling EJ, Edwards PA. Pleiotropic roles of bile acids in metabolism. Cell Metab. 2013;17(5):657–69.
Li T, Chiang JYL. Bile acids as metabolic regulators. Curr Opin Gastroenterol. 2015;31(2):159–65.
Fiorucci S, Mencarelli A, Palladino G, et al. Bile-acid-activated receptors: targeting TGR5 and farnesoid-X-receptor in lipid and glucose disorders. Trends Pharmacol Sci. 2009;30(11):570–80.
Watanabe M, Houten SM, Mataki C, et al. Bile acids induce energy expenditure by promoting intracellular thyroid hormone activation. Nature. 2006;439(7075):484–9.
Patriti A, Facchiano E, Donini A. Effect of duodenal-jejunal exclusion in a non-obese animal model of type 2 diabetes: a new perspective for an old disease. Ann Surg. 2004;240(2):388–9. author reply 9–91.
Srinivasan K, Viswanad B, Asrat L, et al. Combination of high-fat diet-fed and low-dose streptozotocin-treated rat: a model for type 2 diabetes and pharmacological screening. Pharmacol Res. 2005;52(4):313–20.
Pereferrer FS, Gonzalez MH, Rovira AF, et al. Influence of sleeve gastrectomy on several experimental models of obesity: metabolic and hormonal implications. Obes Surg. 2008;18(1):97–108.
Bruinsma BG, Uygun K, Yarmush ML, et al. Surgical models of Roux-en-Y gastric bypass surgery and sleeve gastrectomy in rats and mice. Nat Protoc. 2015;10(3):495–507.
Kawasaki T, Ohta M, Kawano Y, et al. Effects of sleeve gastrectomy and gastric banding on the hypothalamic feeding center in an obese rat model. Surg Today. 2015;45(12):1560–6.
Zwicker BL, Agellon LB. Transport and biological activities of bile acids. Int J Biochem Cell Biol. 2013;45(7):1389–98.
Li T, Owsley E, Matozel M, et al. Transgenic expression of cholesterol 7 alpha-hydroxylase in the liver prevents high-fat diet induced obesity and insulin resistance in mice. Hepatology. 2010;52(2):678–90.
Trauner M, Boyer JL. Bile salt transporters: molecular characterization, function, and regulation. Physiol Rev. 2003;83(2):633–71.
Dawson PA, Lan T, Rao A. Bile acid transporters. J Lipid Res. 2009;50(12):2340–57.
Ferrannini E, Camastra S, Astiarraga B, et al. Increased bile acid synthesis and deconjugation after biliopancreatic diversion. Diabetes. 2015;64(10):3377–85.
Kohli R, Kirby M, Setchell KD, et al. Intestinal adaptation after ileal interposition surgery increases bile acid recycling and protects against obesity-related comorbidities. Am J Physiol Gastrointest Liver Physiol. 2010;299(3):G652–60.
Braghetto I, Davanzo C, Korn O, et al. Scintigraphic evaluation of gastric emptying in obese patients submitted to sleeve gastrectomy compared to normal subjects. Obes Surg. 2009;19(11):1515–21.
Sjovall J. Dietary glycine and taurine on bile acid conjugation in man; bile acids and steroids 75. Proc Soc Exp Biol Med. 1959;100(4):676–8.
Shonsey EM, Wheeler J, Johnson M, et al. Synthesis of bile acid coenzyme a thioesters in the amino acid conjugation of bile acids. Methods Enzymol. 2005;400:360–73.
Hayes KC, Sturman JA. Taurine in metabolism. Annu Rev Nutr. 1981;1:401–25.
Jeevanandam M, Ramias L, Schiller WR. Altered plasma free amino acid levels in obese traumatized man. Metabolism. 1991;40(4):385–90.
Tsuboyama-Kasaoka N, Shozawa C, Sano K, et al. Taurine (2-aminoethanesulfonic acid) deficiency creates a vicious circle promoting obesity. Endocrinology. 2006;147(7):3276–84.
Rosa FT, Freitas EC, Deminice R, et al. Oxidative stress and inflammation in obesity after taurine supplementation: a double-blind, placebo-controlled study. Eur J Nutr. 2014;53(3):823–30.
Carneiro EA, Latorraca MQ, Araujo E, et al. Taurine supplementation modulates glucose homeostasis and islet fanction. J Nutr Biochem. 2009;20(7):503–11.
Cummings BP, Bettaieb A, Graham JL, et al. Vertical sleeve gastrectomy improves glucose and lipid metabolism and delays diabetes onset in UCD-T2DM rats. Endocrinology. 2012;153(8):3620–32.
Chong CPK, Mills PB, McClean P, et al. Bile acid-CoA ligase deficiency—a new inborn error of bile acid metabolism. J Inherit Metab Dis. 2012;35(3):521–30.
Sayin SI, Wahlstrom A, Felin J, et al. Gut microbiota regulates bile acid metabolism by reducing the levels of Tauro-beta-muricholic acid, a naturally occurring FXR Antagonist. Cell Metab. 2013;17(2):225–35.
Rembacz KP, Woudenberg J, Hoekstra M, et al. Unconjugated bile salts shuttle through hepatocyte peroxisomes for taurine conjugation. Hepatology. 2010;52(6):2167–76.
Fisher E, Nitz I, Lindner I, et al. Candidate gene association study of type 2 diabetes in a nested case–control study of the EPIC-Potsdam cohort—role of fat assimilation. Mol Nutr Food Res. 2007;51(2):185–91.
Garbutt JT, Wilkins RM, Lack L, et al. Bacterial modification of taurocholate during enterohepatic recirculation in normal man and patients with small intestinal disease. Gastroenterology. 1970;59(4):553–66.
Alrefai W, Gill R. Bile acid transporters: structure, function, regulation and pathophysiological implications. Pharm Res. 2007;24(10):1803–23.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (no.81270888/H0713, no.81370496/H0308), the Fundamental Research Funds of Shandong University (no. 2014QLKY22), and the Taishan Scholar Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All study protocols were approved by the Animal Care and Utilization Committee of Shandong University.
Conflict of Interest
The authors declare that they have no competing interests.
Statement of Informed Consent
Does not apply.
Statement of Human and Animal Rights
All applicable institutional and national guidelines for the care and use of animals were followed.
Rights and permissions
About this article
Cite this article
Wu, Q., Zhang, X., Zhong, M. et al. Effects of Bariatric Surgery on Serum Bile Acid Composition and Conjugation in a Diabetic Rat Model. OBES SURG 26, 2384–2392 (2016). https://doi.org/10.1007/s11695-016-2087-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-016-2087-2