Abstract
Background
There are limited data quantifying national trends, post-operative readmissions, and revisional surgeries for bariatric procedures. We hypothesized that there is a trend away from Roux en Y gastric bypass (RYGB) and laparoscopic adjustable gastric bands (LAGB) in favor of vertical sleeve gastrectomies (VSG). We hypothesized that VSG was associated with fewer revisions and readmissions, and that demographics and comorbidities were associated with surgery received.
Methods
We used the US-based Premier database, 2008–2013 and 2014 first and second quarters to
-
1.
Examine trends in incidence of RYGB, LAGB and VSG.
-
2.
Quantify occurrence of revisional surgeries and readmissions.
-
3.
Identify predictors of receipt of procedure and of readmissions.
Results
The proportion of VSG increased from 3.0 to 54 % from 2008 to 2014. RYGB decreased from 52 % in 2008 to 32 % by 2014. Earlier year, female sex, white race, western (versus southern) region, and Medicaid predicted receipt of RYGB. Later year, male sex, nonwhite race, northeast or western (versus southern) regions, and insurance type predicted VSG. Readmission was less likely for VSG (OR 0.72, 95 % CI 0.65–0.81), male sex (OR 0.83, 95 % CI 0.72–0.95), and more likely for black race (OR Black vs White 1.2, 95 % CI 1.1–1.4).
Conclusions
Discharge year strongly predicted surgery type. Females, whites, and Medicaid recipients received RYGB more than referents. Conversely, males, non-whites, and insured patients were more likely to receive VSG. Underinsured, regardless of surgery type, were more likely to be readmitted. These findings have important implications for health policy and cost-containment strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The growing obesity epidemic and its costly health sequelae demand a definitive approach to treatment. In the early 2000s, laparoscopic Roux-en-Y gastric bypass (RYGB) had become the most commonly performed bariatric procedure [1–3] closely followed by the laparoscopic adjustable gastric band (LAGB) [4]. Post-operative complications requiring reoperations and readmissions [5–15] and weight regain [16] have deterred insurance companies from offering standard coverage for weight loss surgery [17]. The early experience with sleeve gastrectomy (VSG) suggests a more favorable outcome profile [18]. It has become an increasingly offered intervention [19, 20], recently recognized by the American Society for Metabolic and Bariatric Surgery as a safe and effective primary bariatric procedure. However, the demographic profile, readmission rates, and revision rates after VSG have not been fully quantified.
Regardless of procedure, reducing readmissions and revisional surgeries [21] are critical to reducing health care costs. There are limited data, however, quantifying national surgical trends, post-operative readmissions, and revisional surgeries for bariatric procedures. In addition, factors associated with choosing RYGB, LAGB, or VSG are largely unreported, as are the factors predictive of readmission.
We hypothesized that there is a growing trend away from RYGB and LAGB in favor of VSG for weight loss. We further suspected that VSG would be associated with fewer revisions and readmissions than RYGB or LAGB. We also hypothesized that certain demographics and comorbidities would be associated with receipt of RYGB, LAGB, and VSG.
In order to test these aims, we sought to
-
1.
Identify time trends in the incidence of RYGB, LAGB, and VSG
-
2.
Quantify the occurrence of revisional surgeries and readmissions after each of these procedures
-
3.
Identify predictors of receipt of each of the procedures and predictors of readmissions
Methods
Data Source
We used the Premier database from years 2008–2013 and 2014, first and second quarter. Readmissions data were available for 2012–2013 cases. Premier collects data from a variety of institutions including non-profits, non-government systems, community facilities, teaching hospitals, and large health systems. The information collected is hospital-based, service-level data, and includes data on utilization of resources, costs, diagnoses linked to those costs, and procedures linked to the encounter. The data is extracted from discharge forms of a representative 20 % subset of the US hospital discharges in a de-identified format. It is the largest inpatient drug utilization database in the USA. It captures complete billing and coding data based on the International Classification of Disease (ICD) and Current Procedural Terminology (CPT) codes, for more than 45 million hospital inpatient discharges and more than 210 million hospital outpatient visits. It also captures discharge information on inpatient length of stay, 30-day readmissions, morbidity, and mortality. These data and analysis are exempt from Institutional Review Board approval requirements.
Case Acquisition
CPT and ICD-9 codes corresponding to VSG, RYGB, and LAGB were used to acquire cases. Outpatient, in this dataset, was defined as hospital outpatient designations (i.e., same day surgical procedures) and did not include ambulatory center procedures.
From the set above, cases with ICD-9 codes corresponding to a primary diagnosis of obesity and morbid obesity were selected by including codes 278.00 or 278.01. Additionally, patients with a primary diagnosis indicating complication of a prior bariatric procedure (539.01, 539.09, 539.89) were included.
We excluded cases with missing variables, cases of gastrectomy, or bypass performed for purposes other than weight loss (stricture, malignancy, ulcer disease) and cases coded as non-elective. Open procedures were also excluded as the number of open procedures represented less than 4 % of cases acquired.
Independent Variables
Age was examined both continuously and as <65 versus ≥65. Race was examined as Black, White, and Other. Patients were classified as either independent or partially/totally dependent functional status. Insurance status included Medicare, Medicaid Managed Care, Uninsured, and Other. Medicaid is a state and federally funded, medical insurance coverage offered based on low-income and/or certain disabilities in the USA. We studied hospitals as teaching or community facilities. Comorbidities examined included myocardial infarction, congestive heart failure, peripheral vascular disease, coronary disease, chronic obstructive pulmonary disease, rheumatoid arthritis, peptic ulcers, mild and moderate to severe hepatic disease, diabetes with or without sequelae, renal dysfunction, malignancy with and without metastasis, paralysis, AIDS, and HTN. These were collapsed into the Charlson Comorbidity Index.
Dependent Variables
Independent variables in our predictive models were (1) receipt of RYGB, (2) receipt of VSG, (3) predictors of readmissions for the entire cohort, (4) predictors of readmission for RYGB, and (5) predictors of readmission for VSG.
Statistical Analyses
For the entire cohort, we compared the unadjusted associations of demographic and comorbid conditions with receipt of specific surgeries using Chi-square tests. Multivariate logistic regression analysis was used to predict surgery type, revisions, and readmissions after adjusting for demographics and comorbidities. Statistical significance was considered at the alpha = .05 level. All tests were two-sided, when applicable. The data analysis for this paper was generated using SAS software, version 9.2. copyright, SAS Institute Inc. SAS and all other SAS Institute Inc. product or service names are registered trademarks or trademarks of SAS Institute Inc., Cary, NC, USA.
Results
Cohort Characteristics and Bivariate Results
Table 1 summarizes our total cohort’s demographics and comorbidities. Between 2008 and the first half of 2014, Premier captured 53,365 patients who underwent RYGB, 30,601 who had VSG, and 27,960 who had LAGB. During this time period, there were a minimal number of duodenal switch operations captured, and these were not included in our analyses. Mean age at operation was 45. Mean age among those undergoing RYGB was 45 years, VSG, 44 years, and, LAGB, 45 years. The cohort was 78 % female and 67 % white. Thirty-two percent of captured cases were diabetic. Over the time period examined, persons over the age of 65 represented an increasing proportion of the cohort, growing from 4 to 6 % in the 5.5 year period. Table 2 summarizes readmission characteristics.
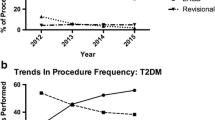
The proportion of VSG captured over time increased from 3.0 % of all cases to 54 % of all cases performed by the first half of 2014. Inversely, the proportion of inpatient bands performed over time fell from 29 % in 2008 to 1 % in 2014. This trend was similar in all captured outpatient LAGB. Whereas, 96 % of outpatient bariatric surgeries were LAGB in 2008, LAGB represented only 32 % of outpatient bariatric procedures by 2014. RYGB accounted for 52 % of inpatient bariatric procedures in 2008 and fell to 32 % by 2014. Among diabetic patients, the incidence of LAGB procedures also decreased with time from 28 % in 2008 to 1 % by 2014. VSG in diabetics increased from 4 to 48 % in the same time frame. Figure 1 shows the distribution of RYGB, LAGB, and VSG over time.
Multivariate Analysis
Predictors of RYGB
After adjusting for age, sex, race, year of discharge, hospital region and size, insurance status, and comorbidities, we found that earlier year of discharge, female sex, white race, western (versus southern) region of the country, and Medicaid insurance predicted receipt of RYGB. Cases captured in 2014 were 16 % less likely to have received RYGB than those in 2008. Females were 7 % as likely to have received RYGB as males and whites were 15 % as likely to have received RYGB as blacks. Figure 2 demonstrates odds ratios and confidence intervals for these variables.
Predictors of VSG
After adjusting for age, sex, race, year of discharge, hospital region and size, insurance status, and comorbidities, we found that later year of discharge, male sex, nonwhite race, northeast or western (versus southern) regions of the country, and insurance status predicted receipt of VSG. Patients with managed care insurance plans were more than twice as likely as Medicare patients to receive VSG. Figure 3 demonstrates odds ratios and confidence intervals for these variables.
Predictors of Readmission
After adjusting for surgery type, age, sex, race, year of discharge, hospital region and size, insurance status, surgeon specialty and comorbidities, we found that readmission was less likely for VSG (OR 0.72, 95 % CI 0.65–0.81), male sex (OR 0.83, 95 % CI 0.72–0.95), and more likely for black race (OR black vs white 1.2, 95 % CI 1.1–1.4).
For RYGB, more recent year of procedure (OR 0.79, 95 % CI 0.67-0.90) and enrollment in managed care (OR 0.70, 95 % CI 0.57–0.85) were associated with lower likelihood of readmission. For VSG, blacks (OR Black vs White 1.3, 95 % CI 1.1–1.6) were more likely to be readmitted, males (OR 0.78, 95 % CI 0.63–0.96), those enrolled in managed care (OR 0.67, 95 % CI 0.51–0.88), and uninsured patients (OR 0.49, 95 % CI 0.28–0.85) were less likely to be admitted.
Sensitivity Analysis
We conducted a number of additional analyses to test whether our results were the consequences of our modeling choices. First, since year of discharge was the independent variable of interest, we examined the behavior of other variables in the model when year was removed from the models entirely. In order to study whether inpatient versus outpatient status affected readmission rates, we added this variable to the models and found no meaningful difference. We also ran additional models excluding outpatient cases to evaluate for differences in inpatient and outpatient populations. In each of these models, estimates changed minimally.
We also examined whether the associations between calendar year and surgery type varied by sex, race, insurance status, hospitals, or surgeon type. Here, we found that the effect of time is more profound for women than men. That is, the likelihood of receiving VSG with increasing year was much higher for women than men and for non-whites.
We also examined the associations of any diagnosis of diabetes mellitus (type 1 or type 2) and of preoperative gastroesophageal reflux disease (GERD) with type of surgery performed. In these models, we found that the presence of diabetes increased the odds of receiving RYGB (OR 1.6, 95 % CI 1.58–1.67) and decreased the odds of receiving VSG (OR 0.76, 95 % CI 0.73–0.79). The presence of pre-procedural GERD nearly doubled the odds of receiving RYGB (OR 1.89, 95 % CI 1.89–1.94), but was not a significant predictor of VSG (OR 1.02, 95 % CI 0.99–1.06).
Discussion
In this risk-adjusted analysis, year of discharge was the foremost predictor of surgery type. With increasing year of discharge, the adjusted odds of RYGB decreased while the adjusted odds of VSG increased. We suspected that the readmission profile for the latter was more favorable and our data support this. Females, whites, diabetics, patients with GERD, and Medicaid recipients received RYGB more frequently than their referents. In contrast, males, non-whites, non-diabetics, and insured patients were more likely than their referents to receive VSG. Patients with GERD were no less likely to receive VSG than those without GERD. Our data suggest that underinsured patients, regardless of surgery type, were more likely to be readmitted. These findings have important implications for health policy and cost-containment strategies.
While the Premier database affords an incomparable means of examining inpatient procedures, it does not contain information on patient BMI status. This variable is likely to be associated with choice of surgery type. Also, Premier does not allow us to examine surgical history, patient preferences, preoperative weight loss, or crossover events (LAGB converted to RYGB or LAGB to VSG). Also, VSG became a Medicare-approved procedure for obesity only in 2012. Therefore, the lower prevalence of VSG prior to 2012 could in part be explained by this coverage issue rather than a change in practice trends. Finally, the database affords us a retrospective analysis of surgical trends and, therefore, is open to confounding.
In spite of its limitations, these data offer several contributions to the literature. There has been an unequivocal shift in practice pattern from RYGB and LAGB to VSG. Though readmissions play a major role in consumption of health care dollars, there is limited literature describing readmissions after bariatric surgery. Our data offers important insights into this problem. Our data demonstrate a 30 % lower readmission rate for VSG than RYGB. This important advantage may help hospitals in reducing costly and unnecessary readmissions. Higher readmissions seen in females and non-whites are also important findings that should be used to direct hospital-based efforts to reduce readmission rates.
In conclusion, our findings provide insight into national procedural trends regarding bariatric surgeries, factors informing which surgery is performed, and which patients are readmitted. The implications of our study findings are multiple. Regarding its surgical anatomy, the VSG offers no anastomoses, no risk of internal herniation, and a reduced risk of vitamin deficiency due to lack of malabsorptive component. Patients undergoing VSG also have lower readmission rates than patients undergoing other bariatric procedures. This advantage may inform the rising rates of VSG in the United States. Long-term durability of weight loss and comorbid disease control are not completely understood. Also, long-term post-operative complication rates after VSG have not yet been quantified. Moving forward, longitudinal data should address these questions.
References
Lazzati A, Guy-Lachuer R, Delaunay V, et al. Bariatric surgery trends in France: 2005–2011. Surg Obes Relat Dis. 2014;10(2):328–34. doi:10.1016/j.soard.2013.07.015.
Memarian E, Calling S, Sundquist K, et al. Sociodemographic differences and time trends of bariatric surgery in Sweden 1990–2010. Obes Surg. 2014;24(12):2109–16.
Reames BN, Finks JF, Bacal D, et al. Changes in bariatric surgery procedure use in Michigan, 2006–2013. JAMA. 2014;312(9):959–61.
Hinojosa MW, Varela JE, Parikh D, et al. National trends in use and outcome of laparoscopic adjustable gastric banding. Surg Obes Relat Dis. 2009;5(2):150–5. doi:10.1016/j.soard.2008.08.006.
Arterburn D, Powers JD, Toh S, et al. Comparative effectiveness of laparoscopic adjustable gastric banding vs laparoscopic gastric bypass. JAMA Surg. 2014;149(12):1279–87.
Anita P. Courcoulas, Susan Z. Yanovski, Denise Bonds, Thomas L. Eggerman, Mary Horlick, Myrlene A. Staten, David E. Arterburn, Long-term Outcomes of Bariatric Surgery: A National Institutes of Health Symposium.
Jafari MD, Jafari F, Young MT, et al. Volume and outcome relationship in bariatric surgery in the laparoscopic era. Surg Endosc. 2013;12:4539–46.
Kellogg TA, Swan T, Leslie DA, et al. Patterns of readmission and reoperation within 90 days after Roux-en-Y gastric bypass. Surg Obes Relat Dis. 2009;5(4):416–23. doi:10.1016/j.soard.2009.01.008.
Krell RW, Finks JF, English WJ, et al. Profiling hospitals on bariatric surgery quality: which outcomes are most reliable? J Am Coll Surg. 2014;219(4):725–34. e3.
Kruger RS, Pricolo VE, Streeter TT, et al. A bariatric surgery center of excellence: operative trends and long-term outcomes. J Am Coll Surg. 2014;218(6):1163–74. doi:10.1016/j.jamcollsurg.2014.01.056.
Saunders JK, Ballantyne GH, Belsley S, et al. 30-day readmission rates at a high volume bariatric surgery center: laparoscopic adjustable gastric banding, laparoscopic gastric bypass, and vertical banded gastroplasty-Roux-en-Y gastric bypass. Obes Surg. 2007;17(9):1171–7.
Saunders J, Ballantyne GH, Belsley S, et al. One-year readmission rates at a high volume bariatric surgery center: laparoscopic adjustable gastric banding, laparoscopic gastric bypass, and vertical banded gastroplasty-Roux-en-Y gastric bypass. Obes Surg. 2008;18(10):1233–40. doi:10.1007/s11695-008-9517-8.
Tee MC, Cao Y, Warnock GL, et al. Effect of bariatric surgery on oncologic outcomes: a systematic review and meta-analysis. Surg Endosc. 2013;27(12):4449–56.
Weiner R, El-Sayes I, Manger T, et al. Obesity Surgery Working Group, Competence Network Obesity. Antidiabetic efficacy of obesity surgery in Germany: a quality assurance nationwide survey. Surg Obes Relat Dis. 2014;10(2):322–7.
Wolfe BM, Belle SH. Long-term risks and benefits of bariatric surgery: a research challenge. JAMA. 2014;312(17):1792–3.
Cooper TC1, Simmons EB, Webb K, Burns JL, Kushner RF. Trends in Weight Regain Following Roux-en-Y Gastric Bypass (RYGB) Bariatric Surgery. Obes Surg. 2015 Jan 17.
Henteleff HJ, Birch DW. Hallowell PT; CAGS/ACS Evidence Based Reviews in Surgery Group. Cost-effectiveness of bariatric surgery for severely obese adults with diabetes. Can J Surg. 2013;56(5):353–5.
Willson T, Gomberawalla A, Mahoney K, et al. Factors influencing 30-day emergency visits and readmissions after sleeve gastrectomy: results from a community bariatric center. Obes Surg. 2014;21.
Nguyen NT, Nguyen B, Gebhart A, et al. Changes in the makeup of bariatric surgery: a national increase in use of laparoscopic sleeve gastrectomy. J Am Coll Surg. 2013;216(2):252–7.
Germanova D, Loi P, van Vyve E, et al. Coelio Club; Previous bariatric surgery increases postoperative morbidity after sleeve gastrectomy for morbid obesity. Acta Chir Belg. 2013;113(4):254–7.
Stefanidis D, Malireddy K, Kuwada T, et al. Revisional bariatric surgery: perioperative morbidity is determined by type of procedure. Surg Endosc. 2013;27(12):4504–10.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Abraham, A., Ikramuddin, S., Jahansouz, C. et al. Trends in Bariatric Surgery: Procedure Selection, Revisional Surgeries, and Readmissions. OBES SURG 26, 1371–1377 (2016). https://doi.org/10.1007/s11695-015-1974-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-015-1974-2