Abstract
Background
The main clinical consequence of sliding hiatal hernia (SHH) is gastroesophageal reflux disease (GERD). Endoscopy and barium swallow X-ray are commonly used to diagnose SHH. We aimed to assess the clinical utility of endoscopy and X-ray in the diagnosis of SHH in morbidly obese patients before and after gastric bypass (GBP).
Methods
Ninety-two patients underwent reflux symptoms evaluation, upper gastrointestinal endoscopy, and barium swallow X-ray before and 6 months after banded GBP. The performance of endoscopy in diagnosing SHH was assessed, taking X-ray as reference. Endoscopy and X-ray were tested as predictors of SHH with GERD.
Results
SHH was more prevalent when characterized by X-ray than endoscopy either before (33% vs. 17%; P = 0.017) or after GBP (26% vs. 7%; P = 0.001). Endoscopy showed low sensitivity (≤40%) and high specificity (≥94%) in diagnosing SHH. Before GBP, more patients with SHH had GERD compared to patients without SHH using either X-ray (83% vs. 58%; P = 0.016) or endoscopy (94% vs. 61%; P = 0.009). After GBP, only patients with radiologic evidence of SHH showed higher prevalence of GERD compared to patients without SHH (50% vs. 26%; P = 0.037). SHH patients also reported weekly or daily vomit more often than patients without SHH (59% vs. 32%; P = 0.026).
Conclusions
In morbidly obese patients, X-ray is superior to endoscopy in diagnosing SHH either before or after banded GBP. In patients treated with this technique, the utilization of X-ray may help in the management of reflux symptoms and frequent vomit.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sliding hiatal hernia (SHH) refers to a condition in which the proximal stomach herniates through the esophageal hiatus of the diaphragm into the mediastinum. The main clinical consequence of SHH is gastroesophageal reflux disease (GERD) [1,2], clinically manifested either by reflux symptoms such as heartburn and acid regurgitation or by reflux esophagitis [3]. In the scenario of GERD, SHH may contribute to the failure of the antireflux barrier and compromising of the esophageal clearance, resulting in increased exposition of the esophagus to reflux of gastric contents [4–6].
Diagnostic techniques used to identify SHH include upper gastrointestinal endoscopy, barium swallow X-ray, and esophageal manometry [1,7–9]. In the clinical practice, the most employed technique is endoscopy, followed by X-ray and manometry. SHH has been characterized at endoscopy when the diaphragmatic indentation is seen 2 cm or more distal to the Z-line and the top of the stomach mucosal folds [10,11]. Radiologic diagnosis depends on characterization of the landmarks of the esophagogastric junction (EGJ) extending above the diaphragmatic hiatus. Classic criteria include herniation of at least 2 cm of gastric cardia above the hiatus [12]. Among these two techniques, X-ray seems to be superior to identify and measure SHH although with limited evidence [1,9].
Obese patients have presented a higher prevalence of GERD compared to lean individuals [13–16]. A hypothesis to explain this association is an increased stress over the diaphragm leading to disruption of the EGJ, resulting in SHH [17]. Theoretically, patients with morbid obesity are at increased risk to develop SHH. In this population, endoscopy has been routinely used either to assess reflux symptoms or as part of the evaluation for bariatric surgery [18–20]. However, the utility of endoscopy as well as X-ray in the diagnosis of SHH in morbidly obese patients has not been formerly investigated. Furthermore, the performance of these techniques in diagnosing SHH after gastric bypass (GBP) is lacking. Therefore, the aim of this study was to assess the clinical utility of endoscopy and X-ray in the diagnosis of SHH in morbidly obese patients evaluated before and after banded GBP.
Methods
Patients
Ninety-two patients with morbid obesity (64 female, aging 38 ± 11.5 years, BMI 45.5 ± 7.6 kg/m2) were consecutively investigated between March and October of 2007 before GBP and 6 months after the surgery. The recruitment was performed irrespective of GERD symptoms. Patients were considered eligible for the study in the presence of the following criteria: (1) age between 18 and 70 years; (2) BMI ≥ 40 or ≥35 kg/m2 combined with significant comorbidities; (3) absence of gastroesophageal surgery; (4) agreement to participate in the study.
Patients underwent GERD symptoms evaluation, upper gastrointestinal endoscopy, and barium swallow X-ray before and after GBP. All procedures were performed prospectively following a standardized protocol. Patients were first classified as having SHH or not based on radiologic criteria. We then tested the performance of endoscopy in identifying SHH, followed by evaluation of X-ray and endoscopy in predicting SHH with esophageal syndrome of GERD. This was defined according to the Montreal Consensus in the presence of troublesome typical symptoms and/or reflux esophagitis [3]. A possible relation between SHH and vomit was also assessed 6 months after the surgery.
The study was approved by the Ethical Committee of the Hospital de Clinicas de Porto Alegre. Informed consent was obtained for every participant.
Clinical Assessment
Typical reflux symptoms were assessed after application of a GERD symptoms questionnaire (GERD-SQ) validated to the native language [21], either before or 6 months after the surgery. GERD-SQ scored heartburn with the question “How bad is the heartburn?” whereas regurgitation was scored with the question “Do you feel return of bitter material from the stomach to the throat?” Symptoms were graded as follows: (0) no symptoms; (1) symptoms noticeable but not troublesome; (2) symptoms troublesome but not every day; (3) symptoms troublesome every day; (4) symptoms affect daily life; (5) symptoms are incapacitating. Symptoms were classified as troublesome when the score was 2 or more. The questionnaire was applied by a nurse blinded to complementary tests. In the evaluation after GBP, patients were asked to classify vomit as absent, monthly, weekly, or daily. Vomit was considered frequent when weekly or daily. Body weight and height were measured using a scale (Filizola, São Paulo, Brazil) and a stadiometer, and BMI was calculated by dividing the weight (kg) by the square of height (m2).
Endoscopy
Endoscopy was performed after 8-h fasting, using a videoendoscope (Olympus GIF-130, Tokyo, Japan). Reflux esophagitis was described according with Los Angeles (LA) classification [22]. SHH was searched at the end of the procedure after deflation of the stomach. SHH was characterized when gastric folds were located at least 2 cm above the diaphragmatic indentation on inspiration [10,11]. Size of SHH was measured using the hash marks on the endoscope, spaced 5 cm apart, relative to the incisors. Endoscopy was conducted by two experienced endoscopists blinded to patient's symptoms and X-ray findings.
Barium Swallow X-Ray
X-ray was performed with patients in fasting conditions, following a standardized protocol. Patients were studied after drinking 200 ml of diluted barium. Determination of the presence of SHH was based on the appearance of the EGJ on the prone horizontal radiographs of the lower esophagus. A skilled radiologist blinded to patient’s symptoms and endoscopy findings analyzed the radiographs. SHH was characterized either in the presence of B ring or gastric folds located at least 2 cm above the level of the diaphragmatic hiatus [12]. B ring was defined as a smooth, symmetric ring-like indentation at the EGJ. The axial length of SHH was measured in centimeters.
Gastric Bypass
Roux-en-Y banded GBP was performed through an upper midline incision. A gastric pouch was created by dividing the stomach with a 10-cm stapler from the lesser curvature (7 cm vertically from the cardia) to 1 cm to the left of the Hiss angle. The estimated volume of the gastric pouch was 20 to 30 ml. The gastric pouch was banded in its middle portion by using a silastic ring (6.5 cm long in circumference). A gastro-jejunal anastomosis was created distal to the ring, keeping an alimentary limb with 100 cm in length, and a biliopancreatic limb ranging 60 to 80 cm. SHH was not repaired during the procedure, except in case of very large hernia (>6 cm).
Statistical Analysis
Categorical data are presented as percentage, which were tested using χ 2 test or Fisher test. Quantitative data are presented as mean (±SD) or when otherwise stated. Parametric (Student's t) or nonparametric tests (Mann–Whitney) were used to compare values between groups depending on the variable distribution by using GraphPad Prism 4 (GraphPad Software, Inc., San Diego, CA, USA). Statistical significance was considered if P < 0.05.
The performance of endoscopy was assessed using WinPEPI version 4.8, including calculation of sensitivity, specificity, predictive values, and accuracy, which were classified as follows: very high (100–81%), high (80–61%), intermediate (60–41%), low (40–21%), and very low (≤20%).
Results
Patients
Out of 92 patients who underwent GBP, eight refused to perform the evaluations after the procedure, including the only patient in whom a very large SHH (11 cm) was surgically repaired. Thus, the study population evaluated after GBP was composed of 84 patients. Demographic and anthropometric data of patients evaluated before and after GBP are presented in Table 1. Most patients were young adults (59% at ≤40 years old) and women (70%). As expected, a significant decrease was observed in BMI after 6 months of the surgery, from 45.5 ± 7.6 to 33.1 ± 6 kg/m2 (P < 0.0001).
Characterization of SHH
The overall prevalence of SHH using either X-ray or endoscopy was 37% preoperatively and 30% following the surgery (Table 2). The agreement between X-ray and endoscopy in indicating the presence of SHH was poor either before (Fig. 1) or after GBP (Fig. 2) but was significantly higher before GBP (35% vs. 12%; P = 0.036).
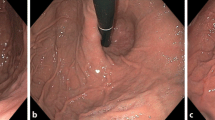
Agreement between X-ray and endoscopy in the characterization of SHH before GBP. In the same patient, radiographic picture (a) shows a SHH interpreted as measuring 5 cm in axial length (traced lines indicate the diaphragmatic impression). On endoscopy, SHH was described as measuring 2 cm in length (b), combined with hiatus enlargement (arrow) in the gastric retroview (c)
Agreement between X-ray and endoscopy in the characterization of SHH after GBP. a (X-ray), b (endoscopy) Pictures of the same patient in whom a SHH was characterized in both techniques. Note that a silastic ring (arrow in a) is found above the diaphragmatic impression, suggesting herniation of the gastric pouch. c, d Pictures of the same patient in whom a SHH was characterized by X-ray (c) but not by endoscopy (d). In this case, a silastic ring (arrow in c) is seen below the diaphragmatic impression
SHH was significantly more prevalent in patients investigated by X-ray than by endoscopy either before (33% vs. 17%; P = 0.017) or after GBP (26% vs. 7%; P = 0.001). The axial length of SHH was larger when measured by X-ray, either before [median (range) = 4 cm (2–11) vs. 2 cm (2–4); P = 0.0001] or after GBP [3.5 cm (2–6) vs. 2 cm (2–2); P = 0.002]. In the radiologic analysis of 84 patients who performed pre- and postoperative evaluations, SHH was found in both evaluations in 13 patients, disappeared in 14 patients, and appeared in nine patients following the surgery.
Performance of Endoscopy in Diagnosing SHH
Data are presented in Table 3. In patients evaluated before GBP, endoscopy showed low sensitivity, very high specificity, and high predictive values in diagnosing SHH. After the surgery, endoscopy showed very low sensitivity and very high specificity to indicate the presence of SHH. Positive predictive value was intermediate, whereas negative predictive value was high. The accuracy of endoscopy in diagnosing SHH was high either before or after GBP.
Prediction of SHH with GERD
In the evaluation before GBP, patients with radiologic evidence of SHH had higher prevalence of GERD compared to patients without SHH (83% vs. 58%; P = 0.016). When using endoscopy, patients with evidence of SHH also showed higher prevalence of GERD compared to patients without SHH (94% vs. 61%; P = 0.009). The percentage of patients with SHH and GERD did not differ in the comparison of X-ray with endoscopy (83% vs. 94%; P = 0.648).
In the evaluation after GBP, the prevalence of GERD in patients with radiologic evidence of SHH decreased in relation to preoperative assessment (50% vs. 83%; P = 0.010) but was higher when compared to patients without SHH following the surgery (50% vs. 26%; P = 0.037). By applying the endoscopic criteria, patients with SHH after GBP also showed a lower prevalence of GERD than patients with SHH in the preoperative evaluation (50% vs. 94%; P = 0.045). After the surgery, GERD prevalence did not differ between patients with endoscopic evidence of SHH and those without SHH (50% vs. 31%; P = 0.380).
In the postoperative evaluation, patients with radiologic evidence of SHH reported weekly or daily vomit more often than patients without SHH (59% vs. 32%; P = 0.026).
Discussion
The aim of this study was to assess the clinical utility of endoscopy and X-ray in the diagnosis of SHH in morbidly obese patients before and after banded GBP. Our main findings were: (1) SHH was more prevalent and larger when characterized by X-ray than endoscopy, either before or after GBP; (2) as a result, the agreement between X-ray and endoscopy was poor, and the sensitivity of endoscopy in diagnosing SHH was low; (3) in the preoperative evaluation, more patients with SHH had GERD compared to patients without SHH, irrespective of the diagnostic technique used to characterize SHH. However, the identification of SHH before GBP rarely modified the surgical approach; and (4) in the postoperative evaluation, X-ray was useful to demonstrate a relation of SHH with GERD and frequent vomit.
To the best of our knowledge, this is the first study comparing endoscopy and X-ray in the diagnosis of SHH before and after GBP. We found that X-ray identified at least a double of cases of SHH compared to endoscopy in both pre- and postoperative evaluations. Furthermore, the axial length of SHH was significantly larger when measured radiologically. According to experts, X-ray has been considered superior to endoscopy in measuring SHH [1,9], but only endoscopy has been routinely indicated in the evaluation prior to bariatric surgery [19,20,23]. A possible explanation for the superiority of X-ray in diagnosing and measuring SHH is that radiographs of the EGJ are taken at the moment of barium deglutition, which is accompanied by esophageal shortening and cranial dislocation of the EGJ [8]. In contrast, the search of SHH during endoscopy is performed at the end of the procedure, when the patient is usually calm with sparser deglutition. These technical peculiarities might explain at least in part the poor agreement between X-ray and endoscopy, as well as the low sensitivity of endoscopy in detecting SHH.
It is well known that SHH may compromise the antireflux barrier and the esophageal clearance, resulting in GERD [1,24]. In the evaluation before GBP, most patients with SHH showed GERD irrespective of the diagnostic technique employed to characterize SHH. Additionally, more patients with SHH had GERD compared to patients without evidence of SHH. These findings reinforce the role of SHH on the pathophysiology of GERD in the scenario of obesity [17,25]. However, the identification of SHH before GBP did not modify our surgical approach, with exception of one patient in whom a very large SHH was repaired. Nevertheless, SHH characterized by X-ray disappeared in approximately half of patients who showed SHH before GBP and appeared in a minority of patients without SHH preoperatively, suggesting that either surgical modifications or factors related with weight loss may contribute to restoration of the EGJ integrity in some patients. The appearance of SHH following GBP might be explained by surgical rupture of the phrenoesophageal ligament or misdiagnosis in the preoperative evaluation. These findings raise the question whether SHH should be repaired in patients undergoing bariatric surgery.
In the postoperative evaluation, the discordance between X-ray and endoscopy in showing SHH was even more evident than before the surgery. By assuming X-ray as reference, the sensitivity of endoscopy was very low in detecting SHH. In consequence, only X-ray was able to indicate a relation between SHH and GERD. In addition, a radiologic evidence of SHH was also related with weekly or daily vomit, suggesting a clinical utility for X-ray in patients who report GERD symptoms or frequent vomit after GBP. Noteworthy, all patients had a banded gastric bypass, which potentially interferes with the transit in the gastric pouch [26]. Whether the avoidance of a silastic ring in this procedure attenuates the finding of SHH and its related symptoms must be addressed.
In conclusion, we assessed the clinical utility of endoscopy and X-ray in the diagnosis of SHH either before or after banded GBP. In the preoperative evaluation, X-ray showed more and larger SHH compared to endoscopy, resulting in low sensitivity of endoscopy in diagnosing SHH. According to our study, the preoperative identification of SHH either by X-ray or endoscopy rarely modifies the surgical technique in patients operated with banded GBP. However, the use of X-ray to identify SHH after this surgery may help in the management of patients with GERD symptoms or frequent vomit.
References
Gordon C, Kang JY, Neild PJ, et al. The role of the hiatus hernia in gastro-oesophageal reflux disease. Aliment Pharmacol Ther. 2004;20:719–32.
Zhu H, Pace F, Trapè E, et al. Prevalence of hiatal hernia and its influence of gastro-oesophageal reflux. Eur J Gastroenterol Hepatol. 1994;6:393–7.
Vakil N, Van Zanten SV, Kahrilas P, et al. The Montreal definition and classification of gastroesophageal reflux disease: a global evidence-based consensus. Am J Gastroenterol. 2006;101:1900–20.
Kahrilas PJ, Lin S, Chen J, et al. The effect of hiatus hernia on gastro-oesophageal junction pressure. Gut. 1999;44:476–82.
Mittal RK, Lange RC, McCallum RW. Identification and mechanism of delayed esophageal acid clearance in subjects with hiatus hernia. Gastroenterology. 1987;92:130–5.
Sloan S, Rademaker AW, Kahrilas PJ. Determinants of gastroesophageal junction incompetence: hiatal hernia, lower esophageal sphincter, or both? Ann Intern Med. 1992;117:977–82.
Fornari F, Fucilini LM, Risson C, et al. Contribution of standard oesophageal manometry in sliding hiatal hernia: From the gastro-oesophageal pressure gradient to the diagnosis. Dig Liver Dis 2009; doi:10.1016/j.dld.2009.04.012.
Kahrilas PJ, Kim HC, Pandolfino JE. Approaches to the diagnosis and grading of hiatal hernia. Best Pract Res Clin Gastroenterol. 2008;22:601–16.
Mittal RK. Hiatal hernia: myth or reality? Am J Med. 1997;103:33S–9S.
Boyce HW. Endoscopic definitions of esophagogastric junction regional anatomy. Gastrointest Endosc. 2000;51:586–92.
Trujillo NP, Slaughter RL, Boyce HW Jr. Endoscopic diagnosis of sliding-type diaphragmatic hiatal hernias. Am J Dig Dis. 1968;13:855–67.
Canon CL, Morgan DE, Einstein DM, et al. Surgical approach to gastroesophageal reflux disease: what the radiologist needs to know. Radiographics. 2005;25:1485–99.
El-Serag HB, Graham DY, Satia JA, et al. Obesity is an independent risk factor for GERD symptoms and erosive esophagitis. Am J Gastroenterol. 2005;100:1243–50.
Fisichella PM, Patti MG. Gastroesophageal reflux disease and morbid obesity: Is there a relation? World J Surg 2009; doi:10.1007/s00268-009-0045-z.
Hampel H, Abraham NS, El-Serag HB. Meta-analysis: obesity and the risk for gastroesophageal reflux disease and its complications. Ann Intern Med. 2005;143:199–211.
Madalosso CA, Fornari F, Callegari-Jacques SM, et al. Performance of the Montreal Consensus in the diagnosis of gastroesophageal reflux disease in morbidly obese patients. Obes Surg. 2008;18:668–74.
Pandolfino JE, El-Serag HB, Zhang Q, et al. Obesity: a challenge to esophagogastric junction integrity. Gastroenterology. 2006;130:639–49.
Fornari F, Madalosso CA, Callegari-Jacques SM, et al. Heartburn during sleep: a clinical marker of gastro-oesophageal reflux disease in morbidly obese patients. Neurogastroenterol Motil. 2009;21:136–42.
Munoz R, Ibanez L, Salinas J, et al. Importance of routine preoperative upper GI endoscopy: why all patients should be evaluated? Obes Surg. 2009;19:427–31.
Sharaf RN, Weinshel EH, Bini EJ, et al. endoscopy plays an important preoperative role in bariatric surgery. Obes Surg. 2004;14:1367–72.
Fornari F, Gruber AC, Lopes AB, et al. Symptom's questionnaire for gastroesophageal reflux disease. Arq Gastroenterol. 2004;41:263–7.
Lundell LR, Dent J, Bennett JR, et al. Endoscopic assessment of oesophagitis: clinical and functional correlates and further validation of the Los Angeles classification. Gut. 1999;45:172–80.
Sharaf RN, Weinshel EH, Bini EJ, et al. Radiologic assessment of the upper gastrointestinal tract: does it play an important preoperative role in bariatric surgery? Obes Surg. 2004;14:313–7.
Penagini R, Carmagnola S, Cantu P. Review article: gastro-oesophageal reflux disease–pathophysiological issues of clinical relevance. Aliment Pharmacol Ther. 2002;16:65–71.
Fass R. The pathophysiological mechanisms of GERD in the obese patient. Dig Dis Sci. 2008;53:2300–6.
Arasaki CH, Del Grande JC, Yanagita ET, et al. Incidence of regurgitation after the banded gastric bypass. Obes Surg. 2005;15:1408–17.
Conflict of Interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fornari, F., Gurski, R.R., Navarini, D. et al. Clinical Utility of Endoscopy and Barium Swallow X-Ray in the Diagnosis of Sliding Hiatal Hernia in Morbidly Obese Patients: A Study Before and After Gastric Bypass. OBES SURG 20, 702–708 (2010). https://doi.org/10.1007/s11695-009-9971-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-009-9971-y