Abstract
Background
Gastroesophageal reflux disease (GERD) has been increasingly recognized in patients with morbid obesity. A recent global evidence-based consensus on GERD has been proposed, but its performance in patients with morbid obesity is unknown. The aim of this study was to assess the performance of the Montreal Consensus in the diagnosis of GERD in morbidly obese patients.
Methods
Seventy-five consecutive morbidly obese patients underwent GERD symptoms assessment, upper gastrointestinal endoscopy, and ambulatory esophageal pH monitoring “off PPI”. The performance of the Montreal Consensus was determined by comparing two diagnostic algorithms: 1. a gold standard approach in which any GERD symptom and findings from both endoscopy and pH monitoring were taken into account, and 2. the approach with the Montreal Consensus, in which troublesome GERD symptoms and endoscopic findings were considered.
Results
GERD was found present in 57 patients by applying the gold standard approach. The Montreal Consensus identified 41 of these patients, whereas the remaining 34 patients were classified as “no GERD”. Of these, 16 (47%) showed reflux esophagitis and/or abnormal pH-metry. The Montreal Consensus had an accuracy of 78.7%, sensitivity of 72% (95% CI 59–82%), specificity of 100% (95% CI 82–100%) and negative predictive value of 47% (95% CI 37–57%).
Conclusions
In morbidly obese patients, the approach with the Montreal Consensus has high specificity and suboptimal sensitivity in the diagnosis of GERD. Its intermediate negative predictive value suggests that complementary investigation might be routine in these patients, particularly in those who do not present with troublesome GERD symptoms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obesity and gastroesophageal reflux disease (GERD) have been increasingly recognized worldwide [1, 2]. Both diseases are associated with negative impact on quality of life and expressive burden in the public health systems [3, 4]. A link between obesity and GERD has been suggested, since studies have recognized obesity as an independent risk factor for reflux symptoms, erosive esophagitis, Barrett’s metaplasia, and esophageal adenocarcinoma [5–9].
Crescent rates of morbid obesity have emerged in the last decades, particularly in the western world [10, 11]. Among several comorbidities, GERD has been frequently found in patients with morbid obesity [12, 13]. Due to the extreme overweight, these patients are exposed to increased rates of transient lower esophageal sphincter relaxations (TLESRs) and gastroesophageal junction disruption, as well as augmented gastroesophageal pressure gradient, favoring the occurrence of reflux [14, 15]. Although crucial for both research and clinical practice, the diagnostic approach for GERD has not been made uniform in these patients.
A recent global consensus on the definition and classification of GERD (Montreal Consensus) has been suggested to standardize data collection in research and clinical practice [16]. Briefly, troublesome GERD symptoms and reflux esophagitis have been considered the “pounds” in the diagnosis of GERD. In spite of this, validated GERD specific questionnaires [17, 18] and traditional complementary techniques, such as a 24-h esophageal pH monitoring, remain as an alternative approach [19].
The increasing prevalence of both morbid obesity and GERD, and the recent description of decreased visceral sensitivity in morbidly obese patients [20, 21] raise the question whether the Montreal Consensus is able to properly recognize GERD in this population. Therefore, the aim of this study was to assess the performance of the Montreal Consensus in the diagnosis of GERD in morbidly obese patients.
Methods
Patients
Seventy-five unselected morbidly obese patients were prospectively studied before bariatric surgery between March and August of 2007. Morbid obesity was characterized by the presence of body mass index (BMI) ≥40 kg/m2 or BMI between 35 and 40 kg/m2 with significant comorbidity. After agreeing to participate in the study, patients underwent clinical evaluation, upper gastrointestinal endoscopy, esophageal manometry, and a 24-h esophageal pH monitoring.
Patients were excluded if they had any of the following conditions: achalasia, gastroesophageal surgery, or use of proton pump inhibitors (PPIs) prior (<7 days) to esophageal function tests. The study was approved by the ethical committee of the Hospital de Clinicas de Porto Alegre. Informed consent was obtained for every participant.
Clinical Evaluation
A standardized procedure was carried out to assess clinical data previously to esophageal function tests. Patients were asked to fill in a validated GERD symptom’s questionnaire [17], in which troublesome heartburn and acid regurgitation were characterized. Briefly, this questionnaire consists of 11 questions about GERD symptoms, including six for heartburn and one for acid regurgitation. Each question grades the symptom in three levels of severity: 0—no symptoms; 1—symptoms noticeable but not troublesome; 2–5—symptoms noticeable and troublesome.
Subsequently, patients were asked about the presence of troublesome chest pain and extraesophageal symptoms possibly related to GERD, including chronic cough, asthma, and chronic laryngitis symptoms (hoarseness, sore throat, and throat clearing). In the presence of any GERD symptoms, patients were asked to elect their chief complaint. Prior gastroesophageal surgery and use of PPIs were also registered. Anthropometric measures (weight, height, and BMI) and demographic data, including race and school level, were taken by a trained nurse.
Upper Gastrointestinal Endoscopy
Endoscopy was carried out by two endoscopists following a standardized protocol, previously to esophageal function tests. Patients were examined after an 8-hour fasting, using a videoendoscope (Olympus GIF-130, Tokyo, Japan). Reflux esophagitis was described according to Los Angeles classification [22]. In case of esophagitis grade A, a consensus about the presence of mucosal breaks was reached after analysis of endoscopic pictures by a third endoscopist, blinded of the patient’s symptoms and pH-metry results.
Ambulatory Esophageal pH Monitoring
Esophageal pH monitoring was performed with a portable pH data recorder (Sandhill Scientific, Inc.; Highlands Ranch, CO, USA) and a pH catheter containing an antimony sensor. After calibration in pH 4.0 and 7.0 solutions, the catheter was inserted transnasally and positioned 5 cm above the LES, as previously determined by manometry testing. The use of PPIs was discontinued at least 7 days before the study. Patients were instructed to keep their habitual daily activities and record symptoms, food or fluid consumption, and posture changes on a diary card. On the following day, the catheter was removed and the data downloaded and analyzed (software GERDcheck, Sandhill Scientific Inc.).
All meal periods were excluded from pH analysis [23]. Patients were considered to have increased esophageal acid exposure if the fraction time with pH <4 was ≥4.4% during the total study period, ≥8.4% in the upright period or ≥3.5% in the supine period [24]. A composite score analysis (DeMeester) was performed, with a value ≥14.7 considered abnormal. Symptom index was calculated as the number of symptom episodes associated with acid reflux divided by the total number of symptom episodes, multiplied by 100%. It was assumed as positive (R+S+) if ≥50% [25]. Esophageal pH monitoring was considered abnormal in the presence of increased acid exposure, increased DeMeester composite score and/or R+S+.
Definition of GERD
The presence of GERD was assessed by two simulated approaches. The gold standard approach defined GERD in the presence of troublesome typical GERD symptoms, reflux esophagitis, and/or abnormal pH monitoring. Typical GERD symptoms included heartburn and acid regurgitation with intensity and frequency enough to be judged troublesome by the patient. Patients with non-troublesome typical GERD symptoms were also considered to have GERD if reflux esophagitis and/or abnormal pH monitoring were found at complementary investigation.
The approach with the Montreal Consensus defined GERD depending on symptom presentation and endoscopic findings [16]. The disease was recognized in the presence of troublesome GERD symptoms. Patients were further classified as having esophageal or extraesophageal syndromes according to the pattern of symptoms, i.e., troublesome heartburn, acid regurgitation, and chest pain, or troublesome cough, laryngitis, and asthma. According to the Montreal Consensus, the presence of GERD symptoms with frequency and intensity not sufficient to be troublesome does not characterize GERD and therefore these patients should not deserve further investigation. Complementary evaluation with endoscopy was carried out in patients with troublesome GERD symptoms to identify the presence of esophageal mucosa injury.
Statistical Analysis
Data are expressed as mean ± SD. Quantitative variables were first tested for Gaussian distribution. Unpaired t test or Mann–Whitney test was employed when appropriate. Categorical variables were analyzed using chi-square test. The performance of the Montreal Consensus in the diagnosis of GERD was assessed by the calculation of test sensitivity, specificity, and predictive values, taking the gold standard approach as referential. The Consensus performance was determined assuming GERD prevalence values of the own study population and from reported data in morbidly obese patients [21]. Test performance was assessed using WinPEPI version 4.1 [26]. A P value of <0.05 was accepted as indicating statistical significance.
Results
Patients
All enrolled patients completed the study. There were 23 men and 52 women, with mean age of 37 years (range 18–61 years) and average BMI of 45.1 kg/m2 (range 35.3–67.7 kg/m2). Most patients (96%) were white Brazilians, while three patients were multiracial Brazilians. Out of the 75 patients, 64 (85%) had low education level, whereas 11 (15%) reported to have graduated from high school.
GERD According to the Gold Standard Approach
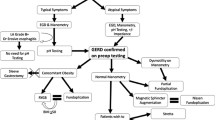
The diagnostic algorithm is presented in Fig. 1. Patients were first classified depending on the clinical presentation: 41 patients had troublesome GERD symptoms (Group 1), seven had non-troublesome GERD symptoms (Group 2), and 27 denied any reflux symptoms (Group 3).
GERD according with the gold standard approach. Patients were first classified depending on the clinical presentation. Those with troublesome GERD symptoms (group 1) were considered to have GERD independently of the complementary investigation. Patients with non-troublesome GERD symptoms (group 2) or without symptoms (group 3) were considered to have GERD in the presence of objective GERD parameters seen at endoscopy or pH-metry
Complementary investigation of Group 1 revealed objective GERD parameters in 33 patients (80%). Esophageal pH monitoring was abnormal in 29 patients: 27 had increased acid exposure (10.3 ± 5.2%) and increased DeMeester score (36.4 ± 17.5), of which 11 had R+S+ for heartburn, one patient had increased DeMeester score (19.4) with normal acid exposure, and one patient had R+S+ for heartburn with normal acid levels. Endoscopy revealed reflux esophagitis in 27 patients: grade A (n = 14), grade B (n = 9), and grade C (n = 4). Eight patients showed no pathological findings at complementary investigation. These patients were considered to have GERD by the presence of troublesome GERD symptoms.
Group 2 had six patients (86%) with objective GERD parameters: five with increased acid exposure (17.1% ± 10.9%) and increased DeMeester score (57.6 ± 32.9), of which one had R+S+ for heartburn, and one patient with R+S+ for heartburn and normal acid levels. Reflux esophagitis was found in five patients: grade A (n = 3) and grade B (n = 2). One patient with non-troublesome GERD symptoms was classified as “no GERD” by lack of pathological findings.
Complementary investigation of Group 3 showed objective GERD parameters in 10 patients (37%): nine had increased acid levels (6.7% ± 2.8%) at pH monitoring, of which seven showed increased DeMeester score (28.4 ± 11.2). Reflux esophagitis was found in three patients (grade A). Seventeen patients had no findings compatible with GERD.
In total, 18 out of 75 morbidly obese patients were considered as “no GERD” after clinical and complementary evaluation. The comparisons between patients with and without GERD showed no significant differences regarding age (38.7 ± 9.6 vs. 33.9 ± 13.3 years; P = 0.097) and sex distribution (male, 35% vs. 17%; P = 0.239). Regular use of PPI was reported by eight patients. Of these patients, seven had abnormal pH monitoring and six showed reflux esophagitis at endoscopy.
GERD According to the Montreal Consensus
The diagnostic algorithm is shown in Fig. 2. Out of the 75 patients, 41 had troublesome typical GERD symptoms by clinical evaluation. In these patients, upper endoscopy revealed normal esophageal mucosa in 14 (34%) and reflux esophagitis in 27 patients (66%).
The remaining 34 patients were classified as “no GERD” by the absence of troublesome reflux symptoms. The comparisons between patients with and without GERD revealed no significant differences regarding age (39.3 ± 9.8 vs. 35.4 ± 11.4 years; P = 0.118) and sex distribution (male, 27% vs. 35%; P = 0.428).
Chest pain was reported by four patients in whom heartburn was the main symptom. Atypical GERD symptoms were reported by 14 patients: five with chronic cough, five with chronic laryngeal symptoms, and four patients with both chronic cough and laryngeal symptoms. These patients had heartburn or acid regurgitation as the chief complaint.
Performance of the Montreal Consensus in the Diagnosis of GERD
The Montreal Consensus showed high specificity, suboptimal sensitivity, high positive predictive value, and intermediate negative predictive value in the diagnosis of GERD after comparison with the gold standard approach (Table 1). Diagnostic accuracy of the Montreal Consensus was 78.7%. This performance was calculated applying the prevalence of GERD found in the study population (76%). By calculating test performance with a reported GERD prevalence of 60% [21], a deterioration was observed in its negative predictive value (70% [95% CI: 61–78%]), while test specificity and sensitivity remained unchanged.
Discussion
Gastroesophageal reflux disease and obesity have gained particular attention by its significant impact on global public health [2, 3, 11, 27]. Both diseases have been associated with lethal conditions, such as esophageal adenocarcinoma and morbid obesity [9, 28, 29]. This phenomenon claims for diagnostic tools of widespread application, able to standardize patient selection in both research and clinical practice. A global evidence-based consensus on GERD has been recently proposed to fulfill the above-mentioned gap [16]. Based on this, we assessed the performance of the Montreal Consensus in the diagnosis of GERD in morbidly obese patients. Test performance was determined after comparison with a gold standard approach for GERD.
The application of the Montreal Consensus had an accuracy of 78.7%, with high specificity and suboptimal sensitivity in the diagnosis of GERD. Posttest probability of the disease was high after a positive result, following its optimal specificity. However, posttest probability of GERD was relatively high (47%) after a negative result, since nearly half of patients labeled as “no GERD” actually showed objective GERD parameters at the gold standard approach.
The high specificity of the Montreal Consensus is linked to the fact that troublesome typical GERD symptoms are synonymous with the disease, irrespective of additional investigation [16]. By using a properly approach to characterize these symptoms, the specificity of the Montreal Consensus is expected to be high, independent of the study population. The key point of this phenomenon is the accuracy of the method applied to characterize symptoms. We believe that our approach was satisfactory based on the application of a structured GERD symptom’s questionnaire, validated to our native language and able to differentiate between troublesome and non-troublesome typical reflux symptoms [17].
By comparing with the gold standard approach, the Montreal Consensus reached suboptimal sensitivity in the diagnosis of GERD. Missing patients were those who reported non-troublesome symptoms or denied reflux symptoms. Complementary investigation with endoscopy and pH monitoring revealed objective GERD parameters in approximately half of these patients. Consequently, the negative predictive value of the Montreal Consensus for GERD recognition was relatively high in our study population. The adoption of a lower GERD prevalence in the analytical model [21] did not change test sensitivity and specificity, but deteriorated its negative predictive value. This finding may be due to the effect of local disease prevalence and should be considered for clinical decisions in different populations investigated with the Montreal Consensus [30].
GERD was highly prevalent in our study population, raising methodological concerns. Participants were candidates for bariatric surgery who fulfilled the criteria for characterization of morbid obesity. After agreement to participate, patients were consecutively included in the study, independent of the presence of GERD symptoms. The high rate of objective GERD parameters found at both endoscopy and pH monitoring was obtained after careful analysis of complementary data, including revision of endoscopic images of patients with small (<5 mm) mucosal breaks, a condition potentially exposed to misinterpretation [31]. In addition, we adopted higher referential values [24] for classification of acid exposure than those routinely employed in our service [32].
Several studies have described an association between obesity and GERD, including crescent rates of reflux esophagitis [5, 6, 8, 33, 34], increased esophageal acid exposure [35], and abnormal visceral sensation [20, 21]. Racial influences and socioeconomic factors may also contribute to GERD in obese individuals. It has been recently shown that an increased obesity may disproportionately increase GERD symptoms in white populations [36]. In addition, a link has been suggested between low socioeconomic status and reflux symptoms [37]. Moreover, the mechanisms by which severe overweight provokes more reflux and consequent esophageal mucosa damage is not clear, but the combination of gastroesophageal junction overstress and abnormal esophageal sensitivity is likely involved [14, 21].
In summary, we assessed the performance of the Montreal Consensus in the diagnosis of GERD in morbidly obese patients. The Consensus showed high specificity and suboptimal sensitivity in the diagnosis of GERD. A relatively high negative predictive value was demonstrated by the lack of recognition of objective GERD parameters in patients without troublesome reflux symptoms. These findings indicate that the Montreal consensus is not sufficient to diagnose all patients with GERD, and that further investigations should be performed routinely in patients who do not present with troublesome typical GERD symptoms. This is particularly important as bariatric surgery may interfere with the gastroesophageal barrier, and promote GERD, especially when surgical techniques such as gastric banding, sleeve gastrectomy, or vertical banded gastroplasty are employed.
References
Bray GA, Bellanger T. Epidemiology, trends, and morbidities of obesity and the metabolic syndrome. Endocrine. 2006;29:109–17.
Dent J, El-Serag HB, Wallander MA, et al. Epidemiology of gastro-oesophageal reflux disease: a systematic review. Gut. 2005;54:710–7.
Shaheen NJ, Hansen RA, Morgan DR, et al. The burden of gastrointestinal and liver diseases, 2006. Am J Gastroenterol. 2006;101:2128–38.
Sichieri R, Do NS, Coutinho W. The burden of hospitalization due to overweight and obesity in Brazil. Cad Saude Publica. 2007;23:1721–7.
Corley DA, Kubo A, Levin TR, et al. Abdominal obesity and body mass index as risk factors for Barrett’s esophagus. Gastroenterology. 2007;133:34–41.
El-Serag HB, Graham DY, Satia JA, et al. Obesity is an independent risk factor for GERD symptoms and erosive esophagitis. Am J Gastroenterol. 2005;100:1243–50.
El-Serag HB, Kvapil P, Hacken-Bitar J, et al. Abdominal obesity and the risk of Barrett’s esophagus. Am J Gastroenterol. 2005;100:2151–6.
Hampel H, Abraham NS, El-Serag HB. Meta-analysis: obesity and the risk for gastroesophageal reflux disease and its complications. Ann Intern Med. 2005;143:199–211.
Lagergren J, Bergstrom R, Lindgren A, et al. Symptomatic gastroesophageal reflux as a risk factor for esophageal adenocarcinoma. N Engl J Med. 1999;340:825–31.
Deitel M. Overweight and obesity worldwide now estimated to involve 1.7 billion people. Obes Surg. 2003;13:329–30.
Deitel M. The obesity epidemic. Obes Surg. 2006;16:377–8.
Cawley J, Prinz T, Beane S. Health insurance claims data as a means of assessing reduction in co-morbidities 6 months after bariatric surgery. Obes Surg. 2006;16:852–8.
Suter M, Dorta G, Giusti V, et al. Gastro-esophageal reflux and esophageal motility disorders in morbidly obese patients. Obes Surg. 2004;14:959–66.
Pandolfino JE, El-Serag HB, Zhang Q, et al. Obesity: a challenge to esophagogastric junction integrity. Gastroenterology. 2006;130:639–49.
Wu JC, Mui LM, Cheung CM, et al. Obesity is associated with increased transient lower esophageal sphincter relaxation. Gastroenterology. 2007;132:883–9.
Vakil N, van Zanten SV, Kahrilas P, et al. The Montreal definition and classification of gastroesophageal reflux disease: a global evidence-based consensus. Am J Gastroenterol. 2006;101:1900–20.
Fornari F, Gruber AC, Lopes AB, et al. Symptom’s questionnaire for gastroesophageal reflux disease. Arq Gastroenterol. 2004;41:263–7.
Velanovich V, Vallance SR, Gusz JR, et al. Quality of life scale for gastroesophageal reflux disease. J Am Coll Surg. 1996;183:217–24.
Hirano I, Richter JE. ACG practice guidelines: esophageal reflux testing. Am J Gastroenterol. 2007;102:668–85.
Jaffin BW, Knoepflmacher P, Greenstein R. High prevalence of asymptomatic esophageal motility disorders among morbidly obese patients. Obes Surg. 1999;9:390–5.
Ortiz V, Ponce M, Fernandez A, et al. Value of heartburn for diagnosing gastroesophageal reflux disease in severely obese patients. Obesity (Silver Spring). 2006;14:696–700.
Lundell LR, Dent J, Bennett JR, et al. Endoscopic assessment of oesophagitis: clinical and functional correlates and further validation of the Los Angeles classification. Gut. 1999;45:172–80.
Wo JM, Castell DO. Exclusion of meal periods from ambulatory 24-hour pH monitoring may improve diagnosis of esophageal acid reflux. Dig Dis Sci. 1994;39:1601–7.
Jamieson JR, Stein HJ, DeMeester TR, et al. Ambulatory 24-h esophageal pH monitoring: normal values, optimal thresholds, specificity, sensitivity, and reproducibility. Am J Gastroenterol. 1992;87:1102–11.
Wiener GJ, Richter JE, Copper JB, et al. The symptom index: a clinically important parameter of ambulatory 24-hour esophageal pH monitoring. Am J Gastroenterol. 1988;83:358–61.
Abramson JH. WINPEPI (PEPI-for-Windows): computer programs for epidemiologists. Epidemiol Perspect Innov. 2004;1:6.
Bray GA. Obesity: a time bomb to be defused. Lancet. 1998;352:160–1.
Adams KF, Schatzkin A, Harris TB, et al. Overweight, obesity, and mortality in a large prospective cohort of persons 50 to 71 years old. N Engl J Med. 2006;355:763–78.
Lagergren J, Bergstrom R, Nyren O. Association between body mass and adenocarcinoma of the esophagus and gastric cardia. Ann Intern Med. 1999;130:883–90.
Linn S, Grunau PD. New patient-oriented summary measure of net total gain in certainty for dichotomous diagnostic tests. Epidemiol Perspect Innov. 2006;3:11.
Lee YC, Lin JT, Chiu HM, et al. Intraobserver and interobserver consistency for grading esophagitis with narrow-band imaging. Gastrointest Endosc. 2007;66:230–6.
Johnson LF, DeMeester TR. Twenty-four-hour pH monitoring of the distal esophagus. A quantitative measure of gastroesophageal reflux. Am J Gastroenterol. 1974;62:325–32.
Aro P, Ronkainen J, Talley NJ, et al. Body mass index and chronic unexplained gastrointestinal symptoms: an adult endoscopic population based study. Gut. 2005;54:1377–83.
Edelstein ZR, Farrow DC, Bronner MP, et al. Central adiposity and risk of Barrett’s esophagus. Gastroenterology. 2007;133:403–11.
El-Serag HB, Ergun GA, Pandolfino J, et al. Obesity increases oesophageal acid exposure. Gut. 2007;56:749–55.
Corley DA, Kubo A, Zhao W. Abdominal obesity, ethnicity and gastro-oesophageal reflux symptoms. Gut. 2007;56:756–62.
Jansson C, Nordenstedt H, Johansson S, et al. Relation between gastroesophageal reflux symptoms and socioeconomic factors: a population-based study (the HUNT Study). Clin Gastroenterol Hepatol. 2007;5:1029–34.
Author information
Authors and Affiliations
Corresponding author
Additional information
Madalosso and Fornari contributed equally to the study.
Rights and permissions
About this article
Cite this article
Madalosso, C.A.S., Fornari, F., Callegari-Jacques, S.M. et al. Performance of the Montreal Consensus in the Diagnosis of Gastroesophageal Reflux Disease in Morbidly Obese Patients. OBES SURG 18, 668–674 (2008). https://doi.org/10.1007/s11695-008-9462-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-008-9462-6