Abstract
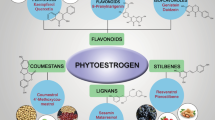
Pueraria mirifica Airy Shaw et Suvatabandhu is a medicinal plant endemic to Thailand. It has been used in Thai folklore medicine for its rejuvenating qualities in aged women and men for nearly one hundred years. Indeed, it has been claimed that P. mirifica contains active phytoestrogens (plant substances with estrogen-like activity). Using high performance liquid chromatography, at least 17 phytoestrogens, mainly isoflavones, have been isolated. Thus, fairly considerable scientific researches, both in vitro in cell lines and in vivo in various species of animals including humans, have been conducted to date to address its estrogenic activity on the reproductive organs, bones, cardiovascular diseases and other climacteric related symptoms. The antioxidative capacity and antiproliferative effect on tumor cell lines have also been assessed. In general, P. mirifica could be applicable for preventing, or as a therapeutic for, the symptoms related to estrogen deficiency in menopausal women as well as in andropausal men. However, the optimal doses for each desirable effect and the balance to avoid undesired side effects need to be calculated before use.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ingham JL, Tahara S, Pope GS. Chemical components and pharmacology of the rejuvenating plant Pueraria mirifica. In: Keung WM, ed. Pueraria: The genus Pueraria. London: Taylor & Francis, 2002: 97–118
Bodner CC, Hymowitz T. Ethnobotany of Pueraria species. In: Keung WM, ed. Pueraria: The genus Pueraria. London: Taylor & Francis, 2002: 29–58
Cherdshewasart W, Kitsamai Y, Malaivijitnond S. Evaluation of the estrogenic activity of the wild Pueraria mirifica by vaginal cornification assay. J Reprod Dev 2007; 53(2): 385–393
Van der Maesen LJG. Pueraria: botanical characteristics. In: Keung WM, ed. Pueraria: The genus Pueraria. London: Taylor &Francis, 2002: 1–28
Malaivijitnond S, Chansri K, Kijkuokul P, Urasopon N, Cherdshewasart W. Using vaginal cytology to assess the estrogenic activity of phytoestrogen-rich herb. J Ethnopharmacol 2006; 107(3): 354–360
Trisomboon H, Malaivijitnond S, Watanabe G, Taya K. Ovulation block by Pueraria mirifica: a study of its endocrinological effect in female monkeys. Endocrine 2005; 26(1): 33–39
Trisomboon H, Malaivijitnond S, Watanabe G, Cherdshewasart W, Taya K. The estrogenic effect of Pueraria mirifica on gonadotrophin levels in aged monkeys. Endocrine 2006; 29(1): 129–134
Lonuchit T, Malaivijitnond S. The determination of Pueraria mirifica samples in the laboratory. Thai Science Journal 2011; 65(6): 76–81
Cherdshewasart W, Sriwatcharakul S. Major isoflavonoid contents of the 1-year-cultivated phytoestrogen-rich herb, Pueraria mirifica. Biosci Biotechnol Biochem 2007; 71(10): 2527–2533
Suwanvijitr T, Kaewmuangmoon J, Cherdshewasart W, Chanchao C. Morphometric and genetic variation in Pueraria mirifica cultivars across Thailand. Pak J Bot 2010; 42(1): 97–109
Suntara A. The Remedy pamphlet of Kwao Krua tuber of Luang Anusarnsuntarakromkarnphiset. Chiang Mai, Thailand: Chiang Mai Upatipongsa Press, 1931
Kerr A. A reputed rejuvenator. J Siam Soc (Natural History Suppl) 1932; 8: 336–338
Sukhavachana D. Ostrogenic principle of Butea superb. J Med Assoc Thai 1941; 24: 83–94
Bounds DG, Pope GS. Light-absorption and chemical properties of miroestrol, the oestrogenic substance of Pueraria mirifica. J Chem Soc 1960; 3696–3704
Kashemsanta L, Suvatabandhu K, Airy Shaw HK. A new species of Pueraria (Leguminosae) from Thailand, yielding an oestrogenic principle. Kew Bull 1952; 7(4): 549–552
Yusakul G, Putalun W, Udomsin O, Juengwatanatrakul T, Chaichantipyuth C. Comparative analysis of the chemical constituents of two varieties of Pueraria candollei. Fitoterapia 2011; 82(2): 203–207
Pope GS, Grundy HM, Jones HEH, Tait SAS. The estrogenic substance (miroestrol) from the tuberous roots of Pueraria mirifica. J Endocrinol 1958; 17(1): 15–16
Wanadorn W. A reputed rejuvenator. J Siam Soc Nat His 1933; Suppl9: 145–147 (in Thai)
Cain JC. Miroestrol: an oestrogen from the plant Pueraria mirifica. Nature 1960; 188(4753): 774–777
Jones HEH, Pope GS. A study of the action of miroestrol and other oestrogens on the reproductive tract of the immature female mouse. J Endocrinol 1960; 20(3): 229–235
Jones HEH, Pope GS. A method for the isolation of miroestrol from Pueraria mirifica. J Endocrinol 1961; 22(3): 303–312
Chansakaow S, Ishikawa T, Seki H, Sekine K, Okada M, Chaichantipyuth C. Identification of deoxymiroestrol as the actual rejuvenating principle of “Kwao Keur,” Pueraria mirifica. The known miroestrol may be an artifact. J Nat Prod 2000; 63(2): 173–175
Okamura S, Sawada Y, Satoh T, Sakamoto H, Saito Y, Sumino H, Takizawa T, Kogure T, Chaichantipyuth C, Higuchi Y, Ishikawa T, Sakamaki T. Pueraria mirifica phytoestrogens improve dyslipidemia in postmenopausal women probably by activating estrogen receptor subtypes. Tohoku J Exp Med 2008; 216(4): 341–351
Jones HEH, Waynforth HB, Pope GS. The effect of miroestrol on vaginal cornification, pituitary function and pregnancy in the rat. J Endocrinol 1961; 22(3): 293–302
Ingham JL, Tahara S, Dziedzic SZ. A chemical investigation of Pueraria mirifica roots. Z Naturforsch Ser C 1986; 41: 403–408
Ingham JL, Tahara S, Dziedzic SZ. Coumestans from the roots of Pueraria mirifica. Z Naturforsch. Ser C 1988; 43: 5–10
Ingham JL, Tahara S, Dziedzic SZ. Minor isoflavones from the roots of Pueraria mirifica. Z Naturforsch Ser C 1989; 44: 724–726
Chansakaow S, Ishikawa T, Sekine K, Okada M, Higuchi Y, Kudo M, Chaichantipyuth C. Isoflavonoids from Pueraria mirifica and their estrogenic activity. Planta Med 2000; 66(6): 572–575
Cherdshewasart W, Subtang S, Dahlan W. Major isoflavonoid contents of the phytoestrogen rich-herb Pueraria mirifica in comparison with Pueraria lobata. J Pharm Biomed Anal 2007; 43(2): 428–434
Urasopon N, Hamada Y, Asaoka K, Poungmali U, Malaivijitnond S. Isoflavone content of rodent diets and its estrogenic effect on vaginal cornification in Pueraria mirifica-treated rats. Sci Asia 2008; 34(4): 371–376
Barnes S, Coward L, Kirk M, Sfakianos J. HPLC-mass spectrometry analysis of isoflavones. Proc Soc Exp Biol Med 1998; 217(3): 254–262
Murkies AL, Wilcox G, Davis SR. Clinical review 92: Phytoestrogens. J Clin Endocrinol Metab 1998; 83(2): 297–303
Ito F, Iwasaki M, Watanabe T, Ishikawa T, Higuchi Y. The first total synthesis of kwakhurin, a characteristic component of a rejuvenating plant, “kwao keur”: toward an efficient synthetic route to phytoestrogenic isoflavones. Org Biomol Chem 2005; 3(4): 674–681
Malaivijitnond S, Kiatthaipipat P, Cherdshewasart W, Watanabe G, Taya K. Different effects of Pueraria mirifica, a herb containing phytoestrogens, on LH and FSH secretion in gonadectomized female and male rats. J Pharmacol Sci 2004; 96(4): 428–435
Yasuda T, Kano Y, Saito K, Ohsawa K. Urinary and biliary metabolites of puerarin in rats. Biol Pharm Bull 1995; 18(2): 300–303
Park EK, Shin J, Bae EA, Lee YC, Kim DH. Intestinal bacteria activate estrogenic effect of main constituents puerarin and daidzin of Pueraria thunbergiana. Biol Pharm Bull 2006; 29(12): 2432–2435
Jin JS, Nishihata T, Kakiuchi N, Hattori M. Biotransformation of C-glucosylisoflavone puerarin to estrogenic (3S)-equol in coculture of two human intestinal bacteria. Biol Pharm Bull 2008; 31(8): 1621–1625
Cherdshewasart W, Traisup V, Picha P. Determination of the estrogenic activity of wild phytoestrogen-rich Pueraria mirifica by MCF-7 proliferation assay. J Reprod Dev 2008; 54(1): 63–67
Chivapat S, Chavalittumrong P, Rattanajarasroj S, Chuthaputti A, Panyamang S. Toxicity study of Pueraria mirifica Airy Shaw et Suvatabandhu. Bull Med Sci 2000; 42(3): 202–223
Cherdshewasart W. Toxicity tests of a phytoestrogen-rich herb; Pueraria mirifica. J Sci Res Chula Univ 2003; 28(1): 1–12
Manosroi A, Saowakhon S, Manosroi J. Preliminary chronic toxicity study of herbal formulations containing red Kwao Krua (Butea superba Roxb.) or white Kwao Krua (Pueraria mirifica Airy Shaw and Suvatabandhu) in wistar rats. SWU J Pharm Sci 2004; 9(1): 1–12
Chivapat S, Chavalittumrong P, Rattanajarasroj S, Panyamang S. Chronic toxicity of Pueraria mirifica in rats. Thai J Pharm 2005; 27(2–3): 76–90
Saenphet K, Kantaoop P, Saenphet S, Aritajat S. Mutagenicity of Pueraria mirifica Airy Shaw & Suvatabandhu and antimutagenicity of Thunbergia laurifolia Linn. Southeast Asian J Trop Med Public Health 2005; 36(Suppl 4): 238–241
Cherdshewasart W, Sutjit W, Pulcharoen K, Chulasiri M. The mutagenic and antimutagenic effects of the traditional phytoestrogen-rich herbs, Pueraria mirifica and Pueraria lobata. Braz J Med Biol Res 2009; 42(9): 816–823
Muangman V, Cherdshewasart W. Clinical trial of the phytoestrogen-rich herb, Pueraria mirifica as a crude drug in the treatment of symptoms in menopausal women. Siriraj Hosp Gaz 2001; 53(5): 300–310
Chandeying V, Lamlertkittikul S. Challenges in the conduct of Thai herbal scientific study: efficacy and safety of phytoestrogen, Pueraria mirifica (Kwao Keur Kao), phase I, in the alleviation of climacteric symptoms in perimenopausal women. J Med Assoc Thai 2007; 90(7): 1274–1280
Manonai J, Chittacharoen A, Udomsubpayakul U, Theppisai H, Theppisai U. Effects and safety of Pueraria mirifica on lipid profiles and biochemical markers of bone turnover rates in healthy postmenopausal women. Menopause 2008; 15(3): 530–535
Cherdshewasart W, Cheewasopit W, Picha P. The differential antiproliferation effect of white (Pueraria mirifica), red (Butea superba), and black (Mucuna collettii) Kwao Krua plants on the growth of MCF-7 cells. J Ethnopharmacol 2004; 93(2–3): 255–260
Sookvanichsilp N, Soonthornchareonnon N, Boonleang C. Estrogenic activity of the dichloromethane extract from Pueraria mirifica. Fitoterapia 2008; 79(7–8): 509–514
Furuya Y, Kohno N, Fujiwara Y, Saitoh Y. Mechanisms of estrogen action on the proliferation of MCF-7 human breast cancer cells in an improved culture medium. Cancer Res 1989; 49(23): 6670–6674
Cherdshewasart W, Cheewasopit W, Picha P. Anti-proliferative effects of the white (Pueraria mirifica), red (Butea superba) and black (Mucuna collettii) Kwao Krua plants on the growth of Hela cells. J Sci Res Chula Univ 2004; 29(1): 27–32
Boonchird C, Mahapanichkul T, Cherdshewasart W. Differential binding with ERalpha and ERbeta of the phytoestrogen-rich plant Pueraria mirifica. Braz J Med Biol Res 2010; 43(2): 195–200
Lee YS, Park JS, Cho SD, Son JK, Cherdshewasart W, Kang KS. Requirement of metabolic activation for estrogenic activity of Pueraria mirifica. J Vet Sci 2002; 3(4): 273–277
Cherdshewasart W, Sriwatcharakul S, Malaivijitnond S. Variance of estrogenic activity of the phytoestrogen-rich plant. Maturitas 2008; 61(4): 350–357
Udomsuk L, Jarukamjorn K, Putalun W, Sakuma T, Kawasaki Y, Nemoto N. Modified expression of aryl hydrocarbon receptorrelated genes by deoxymiroestrol, a phytoestrogen, in mouse hepatocytes in primary culture. J Ethnopharmacol 2011; 137(1): 902–908
Udomsuk L, Juengwatanatrakul T, Putalun W, Jarukamjorn K. Down regulation of gene related sex hormone synthesis pathway in mouse testes by miroestrol and deoxymiroestrol. Fitoterapia 2011; 82(8): 1185–1189
Siangcham T, Saenphet S, Saenphet K. Estrogen bioassay of Pueraria mirifica Airy Shaw & Suvatabandhu. J Med Plant Res 2010; 4(9): 741–744
Urasopon N, Hamada Y, Cherdshewasart W, Malaivijitnond S. Preventive effects of Pueraria mirifica on bone loss in ovariectomized rats. Maturitas 2008; 59(2): 137–148
Malaivijitnond S, Tungmunnithum D, Gittarasanee S, Kawin K, Limjunyawong N. Puerarin exhibits weak estrogenic activity in female rats. Fitoterapia 2010; 81(6): 569–576
Kuiper GGJM, Carlsson B, Grandien K, Enmark E, Häggblad J, Nilsson S, Gustafsson JA. Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors α and β. Endocrinology 1997; 138(3): 863–870
Kuiper GGJM, Lemmen JG, Carlsson B, Corton JC, Safe SH, van der Saag PT, van der Burg B, Gustafsson JA. Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor β. Endocrinology 1998; 139(10): 4252–4263
Jaroenporn S, Malaivijitnond S, Wattanasirmkit K, Trisomboon H, Watanabe G, Taya K, Cherdshewasart W. Effects of Pueraria mirifica, an herb containing phytoestrogens, on reproductive organs and fertility of adult male mice. Endocrine 2006; 30(1): 93–101
Jaroenporn S, Malaivijitnond S, Wattanasirmkit K, Watanabe G, Taya K, Cherdshewasart W. Assessment of fertility and reproductive toxicity in adult female mice after long-term exposure to Pueraria mirifica herb. J Reprod Dev 2007; 53(5): 995–1005
Trisomboon H, Malaivijitnond S, Watanabe G, Taya K. Estrogenic effect of Pueraria mirifica on the menstrual cycle and hormones related ovarian function in cyclic female cynomolgus monkeys. J Pharmacol Sci 2004; 94(1): 51–59
Trisomboon H, Malaivijitnond S, Cherdshewasart W, Watanabe G, Taya K. The influence of Pueraria mirifica herb containing phytoestrogens on the urinary gonadotropin and estradiol levels in aged menopausal monkeys. Anim Sci J 2007; 78(4): 378–386
Trisomboon H, Malaivijitnond S, Cherdshewasart W, Watanabe G, Taya K. Assessment of urinary gonadotropin and steroid hormone profiles of female cynomolgus monkeys after treatment with Pueraria mirifica. J Reprod Dev 2007; 53(2): 395–403
Urasopon N, Hamada Y, Asaoka K, Cherdshewasart W, Malaivijitnond S. Pueraria mirifica, a phytoestrogen-rich herb, prevents bone loss in orchidectomized rats. Maturitas 2007; 56(3): 322–331
Trisomboon H, Malaivijitnond S, CherdshewasartW,Watanabe G, Taya K. Effect of Pueraria mirifica on the sexual skin coloration of aged menopausal cynomolgus monkeys. J Reprod Dev 2006; 52(4): 537–542
Malaivijitnond S, Hamada Y, Suryobroto B, Takenaka O. Female long-tailed macaques with scrotum-like structure. Am J Primatol 2007; 69(7): 721–735
Onouchi T, Kato J. Estrogen receptors and estrogen-inducible progestin receptors in the sexual skin of the monkey. J Steroid Biochem 1983; 18(2): 145–151
Langkalichan Y, Smitasiri Y. Effect of White Cwow (Pueraria mirifica) on reproduction in male Albino rat. Master’s Thesis, Chiang Mai University, Thailand, 1984
Muangdet N, Anuntalabhochai S. Effects of low doses of white gwow (Pueraria mirifica) on female Japanese quails. J Sci Fac CMU 1986; 12(1): 28–40
Smitasiri Y, Junyatum U, Songjitsawad A, Sripromma P, Trisrilp S, Snuntalabhochai S. Post-coital antifertility effects of Pueraria mirifica in rat. J Sci Fac CMU 1987; 13(1): 19–28
Kanis JA, Melton LJ 3rd, Christiansen C, Johnston CC, Khaltaev N. The diagnosis of osteoporosis. J Bone Miner Res 1994; 9(8): 1137–1141
Limpaphayom KK, Taechakraichana N, Jaisamrarn U, Bunyavejchevin S, Chaikittisilpa S, Poshyachinda M, Taechamahachai C, Havanond P, Onthuam Y, Lumbiganon P, Kamolratanakul P. Prevalence of osteopenia and osteoporosis in Thai women. Menopause 2001; 8(1): 65–69
Compston JE. Sex steroids and bone. Physiol Rev 2001; 81(1): 419–447
Weitzmann MN, Pacifici R. Estrogen deficiency and bone loss: an inflammatory tale. J Clin Invest 2006; 116(5): 1186–1194
Riggs BL, Wahner HW, Seeman E, Offord KP, Dunn WL, Mazess RB, Johnson KA, Melton LJ 3rd. Changes in bone mineral density of the proximal femur and spine with aging. Differences between the postmenopausal and senile osteoporosis syndromes. J Clin Invest 1982; 70(4): 716–723
Ohta H, Makita K, Komukai S, Nozawa S. Bone resorption versus estrogen loss following oophorectomy and menopause. Maturitas 2002; 43(1): 27–33
Fontanges E, Fontana A, Delmas P. Osteoporosis and breast cancer. Joint Bone Spine 2004; 71(2): 102–110
Sulak PJ. Endometrial cancer and hormone replacement therapy. Appropriate use of progestins to oppose endogenous and exogenous estrogen. Endocrinol Metab Clin North Am 1997; 26(2): 399–412
Canavan TP, Doshi NR. Endometrial cancer. Am Fam Physician 1999; 59(11): 3069–3077
Lissin LW, Cooke JP. Phytoestrogens and cardiovascular health. J Am Coll Cardiol 2000; 35(6): 1403–1410
Wang X, Wu J, Chiba H, Umegaki K, Yamada K, Ishimi Y. Puerariae radix prevents bone loss in ovariectomized mice. J Bone Miner Metab 2003; 21(5): 268–275
Wang X, Wu J, Chiba H, Yamada K, Ishimi Y. Puerariae radix prevents bone loss in castrated male mice. Metabolism 2005; 54(11): 1536–1541
Onoe Y, Miyaura C, Ohta H, Nozawa S, Suda T. Expression of estrogen receptor β in rat bone. Endocrinology 1997; 138(10): 4509–4512
Trisomboon H, Malaivijitnond S, Suzuki J, Hamada Y, Watanabe G, Taya K. Long-term treatment effects of Pueraria mirifica phytoestrogens on parathyroid hormone and calcium levels in aged menopausal cynomolgus monkeys. J Reprod Dev 2004; 50(6): 639–645
Yamaguchi M, Gao YH. Inhibitory effect of genistein on bone resorption in tissue culture. Biochem Pharmacol 1998; 55(1): 71–76
Gao YH, Yamaguchi M. Anabolic effect of daidzein on cortical bone in tissue culture: comparison with genistein effect. Mol Cell Biochem 1999; 194(1–2): 93–97
Sugimoto E, Yamaguchi M. Stimulatory effect of daidzein in osteoblastic MC3T3-E1 cells. Biochem Pharmacol 2000; 59(5): 471–475
Gao YH, Yamaguchi M. Suppressive effect of genistein on rat bone osteoclasts: apoptosis is induced through Ca2+ signaling. Biol Pharm Bull 1999; 22(8): 805–809
Chandeying V, Sangthawan M. Efficacy comparison of Pueraria mirifica (PM) against conjugated equine estrogen (CEE) with/ without medroxyprogesterone acetate (MPA) in the treatment of climacteric symptoms in perimenopausal women: phase III study. J Med Assoc Thai 2007; 90(9): 1720–1726
Lamlertkittikul S, Chandeying V. Efficacy and safety of Pueraria mirifica (Kwao Kruea Khao) for the treatment of vasomotor symptoms in perimenopausal women: phase II Study. J Med Assoc Thai 2004; 87(1): 33–40
Wattanapitayakul SK, Chularojmontri L, Srichirat S. Effects of Pueraria mirifica on vascular function of ovariectomized rabbits. J Med Assoc Thai 2005; 88(Suppl 1): S21–S29
Manonai J, Chittacharoen A, Theppisai U, Theppisai H. Effect of Pueraria mirifica on vaginal health. Menopause 2007; 14(5): 919–924
Manonai J, Seif C, Böhler G, Jünemann KP. The effect of Pueraria mirifica on cytologic and urodynamic findings in ovariectomized rats. Menopause 2009; 16(2): 350–356
Xu H, Gouras GK, Greenfield JP, Vincent B, Naslund J, Mazzarelli L, Fried G, Jovanovic JN, Seeger M, Relkin NR, Liao F, Checler F, Buxbaum JD, Chait BT, Thinakaran G, Sisodia SS, Wang R, Greengard P, Gandy S. Estrogen reduces neuronal generation of Alzheimer β-amyloid peptides. Nat Med 1998; 4(4): 447–451
McEwen BS. Clinical review 108: The molecular and neuroanatomical basis for estrogen effects in the central nervous system. J Clin Endocrinol Metab 1999; 84(6): 1790–1797
Frick KM, Fernandez SM, Bulinski SC. Estrogen replacement improves spatial reference memory and increases hippocampal synaptophysin in aged female mice. Neuroscience 2002; 115(2): 547–558
Sucontphunt A, De-Eknamkul W, Nimmannit U, Dan Dimitrijevich S, Gracy RW. Protection of HT22 neuronal cells against glutamate toxicity mediated by the antioxidant activity of Pueraria candollei var. mirifica extracts. J Nat Med 2011; 65(1): 1–8
Chindewa R, Lapanantasin S, Sanvarinda Y, Chongthammakun S. Pueraria mirifica, phytoestrogen-induced change in synaptophysin expression via estrogen receptor in rat hippocampal neuron. J Med Assoc Thai 2008; 91(2): 208–214
Cherdshewasart W, Panriansaen R, Picha P. Pretreatment with phytoestrogen-rich plant decreases breast tumor incidence and exhibits lower profile of mammary ERalpha and ERbeta. Maturitas 2007; 58(2): 174–181
Phansawan B, Poungbangpho S. Antioxidant capacities of Pueraria mirifica, Stevia rebaudiana Bertoni, Curcuma longa Linn., Andrographis paniculata (Burm.f.) Nees. and Cassia alata Linn. for the development of dietary supplement. Kasetsart J (Nat Sci) 2007; 41(3): 548–554
Cherdshewasart W, Sutjit W. Correlation of antioxidant activity and major isoflavonoid contents of the phytoestrogen-rich Pueraria mirifica and Pueraria lobata tubers. Phytomedicine 2008; 15(1–2): 38–43
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Malaivijitnond, S. Medical applications of phytoestrogens from the Thai herb Pueraria mirifica . Front. Med. 6, 8–21 (2012). https://doi.org/10.1007/s11684-012-0184-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11684-012-0184-8