Summary
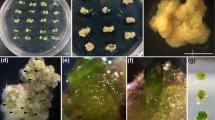
An efficient regeneration system was developed by culturing immature cotyledons and embryo axes of Arachis hypogaea L. cv. Georgia Green on Murashige and Skoog basal medium (MS) supplemented with various concentrations of thidiazuron (TDZ; 1, 5, 10, and 15 µM). Highly morphogenic callus was produced from 100% of the explants comprising the cotyledon with attached embryo axis when cultured in the dark on 10 µM TDZ. Upon excision and continued culture in the dark on 10 µM TDZ, morphogenic callus grew repetitively during monthly subcultures and retained its regeneration potential. For organogenesis, a gradual reduction in TDZ concentration and exposure to light were necessary before transfer to MS basal medium. Inclusion of indole-3-butyric acid in liquid MS medium favored rooting of recovered shoots. A distinct feature of this investigation is the induction of highly morphogenic callus by TDZ and regeneration of morphologically normal, fertile peanut plants after 8 months of callus subculture.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arndt, F. R.; Rusch, R.; Stillfried, H. V. SN 49537, a new cotton defoliant. Plant Physiol. 57s:99; 1976.
Baker, C. M.; Durham, R. E.; Burns, J. A.; Parrott, W. A.; Wetzstein, H. Y. High frequency somatic embryogenesis in peanut (Arachis hypogaea L.) using mature, dry seed. Plant Cell Rep. 15:38–42; 1995.
Baker, C. M.; Wetzstein, H. Y. Influence of auxin type and concentration on peanut somatic embryogenesis. Plant Cell Tissue Organ Cult. 36:361–368; 1994.
Bates, S.; Preece, J. E.; Navarrete, N. E.; Van Sambeek, J. W.; Gaffney, G. R. Thidiazuron stimulates shoot organogenesis and somatic embryogenesis in white ash (Fraxinus americana L.). Plant Cell Tissue Organ Cult. 31:21–29; 1992.
Bohmer, P.; Meyer, B.; Jacobsen, H. J. Thidiazuron-induced high frequency of shoot induction and plant regeneration in protoplast derived pea callus. Plant Cell Rep. 15:26–29; 1995.
Capelle, S. C.; Mok, D. W. S.; Kirchner, S. C.; Mok, M. C. Effects of thidiazuron on cytokinin autonomy and the metabolism of N6-(delta 2-isopentenyl) (8-14C) adenosine in callus tissues of Phaseolus lunatus L. Plant Physiol. 73:796–780; 1983.
Durham, R. E.; Parrott, W. A. Repetitive embryogenesis from peanut cultures in liquid medium. Plant Cell Rep. 11:122–125; 1992.
Gill, R.; Gerrath, J. M.; Saxena, P. K. High-frequency direct embryogenesis in thin layer cultures of hybrid seed geranium (Pelargonium × hortorum). Can. J. Bot. 71:408–413; 1993.
Gill, R.; Saxena, P. K. Direct somatic embryogenesis and regeneration of plants from seedlings of peanut (Arachis hypogaea): promotive role of thidiazuron. Can. J. Bot. 70:1186–1192; 1992.
Gill, R.; Saxena, P. K. Somatic embryogenesis in Nicotiana tabacum: induction by thidiazuron of direct embryo differentiation from leaf disc. Plant Cell Rep. 12:154–159; 1993.
Hazra, S. S.; Sathaye, S. S.; Mascarenhas, A. F. Direct somatic embryogenesis in peanut (Arachis hypogaea). Bio/Technology 7:949–951; 1989.
Hinchee, M. A. W.; Corbin, D. R.; Armstrong, C. L.; Fry, J. E.; Sato, S. S.; Deboer, D. L.; Petersen, W. L.; Armstrong, T. A.; Connor-Ward, D. V.; Layton, J. G.; Horsch, R. D. Plant transformation. In: Vasil, I. K.; Thorpe, T. A., eds. Plant cell and tissue culture. Dordrecht, The Netherlands: Kluwer Academic Publishers; 1994:231–270.
Huetteman, C. A.; Preece, J. E. Thidiazuron: a potent cytokinin for woody plant tissue culture. Plant Cell Tissue Organ Cult. 33:105–119; 1993.
Kaneda, Y.; Tabei, Y.; Nishimura, S.; Harada, K.; Akihama, T.; Kitamura, K. Combination of thidiazuron and basal media with low salt concentrations increases the frequency of shoot organogenesis in soybeans (Glycine max L. Merr.). Plant Cell Rep. 17:8–12; 1997.
Kanyand, M.; Dessai, A. P.; Prakash, C. S. Thidiazuron promotes high frequency regeneration of peanut (Arachis hypogaea) plants in vitro. Plant Cell Rep. 14:1–5; 1994.
Kartha, K. K.; Pahl, K.; Leung, N. L.; Mroginski, L. A. Plant regeneration from meristems of grain legumes: soybean, cowpea, peanut, chickpea, and bean. Can. J. Bot. 59:1671–1679; 1981.
Knauft, D. A.; Ozias-Akins, P. Recent methodologies for germplasm enhancement and breeding. In: Pattee, H. E.; Stalker, H. T., ed. Advances in peanut science. Stillwater, OK: American Peanut Research and Education Society, Inc.; 1996:54–94.
Li, Z.; Jarret, R. L.; Pittman, R. N.; Demski, J. W. Shoot organogenesis from cultured seed explants of peanut (Arachis hypogaea L.) using thidiazuron. In Vitro Cell. Dev. Biol. 30P:187–191; 1994.
Livingstone, D. M.; Birch, R. G. Plant regeneration and microprojectile-mediated gene transfer in embryogenic leaflets of peanut (Arachis hypogaea L.). Aust. J. Plant Physiol. 22:585–591; 1995.
McKently, A. H. Direct somatic embryogenesis from axes of mature peanut embryos. In Vitro Cell. Dev. Biol. 27P:197–200; 1991.
McKently, A. H. Effect of genotype on somatic embryogenesis from axes of mature peanut embryos. Plant Cell Tissue Organ Cult. 42:251–254; 1995.
Mok, M. C.; Mok, D. W. S.; Turner, J. E.; Mujer, C. V. Biological and biochemical effects of cytokinin-active phenylurea derivatives in tissue culture system. HortScience 22:1194–1196; 1987.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol. Plant. 15:473–497; 1962.
Murthy, B. N. S.; Murch, S. J.; KrishnaRaj, S.; Saxena, P. K. Thidiazuron: a potent regulator of in vitro plant morphogenesis. In Vitro Cell. Dev. Biol. Plant 34:267–275; 1998.
Murthy, B. N.; Murch, S. J.; Saxena, P. K. Thidiazuron-induced somatic embryogenesis in intact seedlings of peanut (Arachis hypogaea): endogenous growth regulator levels and significance of cotyledons. Physiol. Plant. 94:268–276; 1995.
Ozias-Akins, P. Plant regeneration from immature embryos of peanut. Plant Cell Rep. 8:217–218; 1989.
Ozias-Akins, P.; Anderson, W. F.; Holbrook, C. C. Somatic embryogenesis in Arachis hypogaea L.: genotype comparison. Plant Sci. 83:103–111; 1992a.
Ozias-Akins, P.; Schnall, J. A.; Anderson, W. F.; Singsit, C.; Clemente, T. E.; Adang, M. J.; Weissinger, A. K. Regeneration of transgenic peanut plants from stably transformed embryogenic callus. Plant Sci. 93:185–194; 1993.
Ozias-Akins, P.; Singsit, C.; Branch, W. D. Interspecific hybrid inviability in crosses of Arachis hypogaea × A. stenosperma can be overcome by in vitro embryo maturation or somatic embryogenesis. J. Plant Physiol. 140:207–212; 1992b.
SAS Institute Inc. SAS Technical Report P-229, SAS/STAT Software: changes and enhancements, Release 6.07. Cary, NC: SAS Institute; 1992.
Saxena, P. K.; Malik, K. A.; Gill, R. Induction by thidiazuron of somatic embryogenesis in intact seedlings of peanut. Planta 187:421–424; 1992.
Sellars, R. M.; Southward, G. M.; Phillips, G. C. Adventitious somatic embryogenesis from cultured immature zygotic embryos of peanut and soybean. Crop Sci. 30:408–414; 1990.
Singsit, C.; Adang, M. J.; Lynch, R. E.; Anderson, W. F.; Wang, A.; Cardineau, G.; Ozias-Akins, P. Expression of a Bacillus thuringiensis crylA(c) gene in transgenic peanut plants and its efficacy against lesser cornstalk borer. Transgenic Res. 6:169–176; 1997.
Thomas, J. C.; Katterman, F. R. Cytokinin activity induced by thidiazuron. Plant Physiol. 81:681–683; 1986.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gill, R., Ozias-Akins, P. Thidiazuron-induced highly morphogenic callus and high frequency regeneration of fertile peanut (Arachis hypogaea L.) plants. In Vitro Cell.Dev.Biol.-Plant 35, 445–450 (1999). https://doi.org/10.1007/s11627-999-0066-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11627-999-0066-1