Abstract
Anaphyllum wightii Schott. is an ethnomedicinally significant plant endemic to the southern region of Western Ghats. The present study aimed to develop an efficient protocol for the in vitro propagation of the plant through somatic embryogenesis. Fresh petioles were selected as explants for the experiment. The medium used was half-strength Murashige and Skoog (MS) medium supplemented with different concentrations of 2,4-dichlorophenoxyacetic acid (2,4-D) and naphthaleneacetic acid (NAA). Both 2,4-D and NAA showed embryogenic callus induction from petiole explants after up to 4 wk of culture. NAA at 2 mg L−1 showed the highest percentage of embryogenic callus induction (83.33 ± 8.80%). Histological and stereomicroscopic observations of the embryogenic callus revealed various stages of somatic embryos indicating an asynchronous type of embryogenesis. Full-strength MS medium containing 3 mg L−1 6-benzylaminopurine induced the maximum number of shoots per callus (6.00 ± 0.58) after 30 d of culture for NAA-induced calluses. The highest rooting response was obtained with half-strength MS medium fortified with 0.5 mg L−1 indole-3-butyric acid. The in vitro rooted plantlets were hardened by transferring to small plastic pots containing sand and garden soil (1:1) and showed a 76% survival rate after 4 wk. Thus, the present work developed an efficient in vitro protocol for the conservation of this plant species and also contributes to the study of the embryonic development.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The plant Anaphyllum wightii Schott., commonly known as ‘Wight’s twisted arum’ (‘Keerikizhangu’ in Malayalam), is an endemic and threatened genus of South India (Ahmedullah and Nayar 1986). It is included in the ‘Vulnerable’ category of IUCN (Rajasekharan and Wani 2020). The plant is a tall herb belonging to the family ‘Araceae’ and is usually found in the Western Ghats region at an altitude of 650 to 1000 m. The plant has a rhizomatous stem and pinnately compound leaves. The inflorescence is a spadix with a once or twice twisted spathe.
The tribal communities in Kerala, including the Kani tribes of Kottoor reserve forest and the Agasthyavanam, Thiruvananthapuram, and Malapandaram tribes of Achankovil forest, Kollam, use fresh tubers of A. wightii as an antidote against snake bite and also as food (Ramachandran 2007; Mathew and George 2013). The rhizome of this plant is used by the Kanikkar tribal community of Tamil Nadu to treat skin diseases like eczema and scabies (Kunjumon et al. 2016). Reports suggest that the rhizome of A. wightii possesses antimicrobial, antioxidant, hepatoprotective, anthelmintic, anti-inflammatory, and antidiabetic pharmacological activities (Udayan et al. 2007).
Even though vegetative propagation through tuber is an effective plant regeneration method, the overexploitation of tubers for their medicinal and food value limits their use as a propagule. The seeds of this plant can also germinate into plantlets; however, flowering and seed setting usually occur once a year, and the propagation through seeds alone is not sufficient for the conservation of such an endemic plant species. Hence, it is necessary to develop an efficient protocol for the in vitro propagation of this ethnomedicinally significant plant. Somatic embryogenesis is one of the efficient methods of in vitro plant propagation where a bipolar structure, similar to a zygotic embryo, develops from a non-zygotic cell having independent vasculature without connection to that of the original tissue (Arnold et al. 2002). Somatic embryogenesis is a powerful tool for the study of plant developmental processes since it involves most of the developmental stages, the same as that of zygotic embryos (Elhiti et al. 2013). Since there are no previous reports regarding somatic embryogenesis in this endemic plant, the present study aimed to develop a protocol for the in vitro propagation of the plant through somatic embryogenesis.
Materials and Methods
Explant source
The plant materials of A. wightii Schott. were collected from the Kallar region of Thiruvananthapuram District, Kerala, and maintained in the garden of the Department of Botany, University of Kerala, Thiruvananthapuram. Fresh and healthy petiole explants, obtained from approximately 6- to 8-mo-old field-grown plants, were used for the in vitro culture experiments.
Explant preparation and sterilization
The petiole explants were cut into pieces and washed under running tap water for 15 min. The explants were then washed with diluted Tween™ 20 detergent solution (1 drop per 100 mL) (HiMedia Laboratories Pvt. Ltd., Mumbai, India) for 10 min, followed by washing under running tap water for about 30 min. The explants were subsequently rinsed with sterile distilled water three times and surface sterilized by immersing in 70% ethanol for 30 s, followed by washing with 0.1% (w/v) mercuric chloride (HiMedia Laboratories Pvt. Ltd.) for 4 min. Again, the explants were vigorously rinsed with sterile distilled water 3 to 5 times to remove the traces of mercuric chloride solution.
Embryogenic callus induction and proliferation
After surface sterilization, the petiole explants were cut into pieces of smaller size (0.5 to 1 cm) and inoculated onto half-strength basal Murashige and Skoog (MS; Murashige and Skoog 1962) medium (HiMedia Laboratories Pvt. Ltd.) (pH 5.8) supplemented with 0.5 mg L−1, 1.0 mg L−1, 2.0 mg L−1, 3.0 mg L−1, 4.0 mg L−1, or 5.0 mg L−1 2,4-D or 0.5 mg L−1, 1.0 mg L−1, 2.0 mg L−1, 4.0 mg L−1, 6.0 mg L−1, or 8.0 mg L−1 naphthaleneacetic acid (NAA) (Sigma-Aldrich, Bangalore, India). The experimental design used was a completely randomized block design. Each treatment consisted of 10 explants, and the experiment was conducted in triplicate. The cultures were maintained at 25 ± 1 °C with alternate light (12-h) and dark (12-h) conditions, and the percentage of embryogenic callus induction was recorded after 45 d of incubation. Subculturing was done at an interval of 30 d on the same medium composition, in which they induced, for the proliferation of embryogenic callus cultures.
Shoot regeneration from embryogenic callus cultures
The embryogenic calluses were inoculated on full-strength MS medium fortified with 1 to 5 mg L−1 6-benzylaminopurine (BAP) (Sigma-Aldrich) with regular subculturing after 30 d of incubation. The percentage of shoot induction, shoot number, and shoot length were recorded separately for 2,4-D- and NAA-induced calluses.
Histological analysis
The embryogenic calluses and embryoids were subjected to free-hand sectioning followed by staining with safranin (HiMedia Laboratories Pvt. Ltd.) and were observed under a Leica EZ4E stereomicroscope and image analyzer (Leica DM2000 microscope) (Leica Microsystems; Schweiz AG, Heerbrugg, Switzerland) to study the anatomical features of somatic embryos.
In vitro root induction
The individual shoots formed from embryogenic callus cultures were transferred to half-strength MS medium supplemented with 0.2 to 2.0 mg L−1 indole-3-acetic acid (IAA), indole-3-butyric acid (IBA), or NAA (Sigma-Aldrich) for in vitro root induction. The percentage of rooting response, the number of roots per shoot, and root length were recorded for all three hormones after 45 d of incubation.
Hardening and acclimatization
After 45 d, the in vitro rooted plantlets were removed from the culture bottles (400-mL glass bottles with plastic screw caps) (Abroach Exim Pvt. Ltd., Ahmedabad, India) containing medium, washed in tap water to remove traces of medium, and planted in small pots containing sand and garden soil (1:1) and covered with a transparent polythene bag with small pores to maintain humidity. The plantlets were watered once a week. After 4 wk, the polythene bags were removed and transferred to greenhouse conditions.
Statistical analysis
Statistical analysis was performed using the software SPSS/PC version 22 (SPSS Inc., Chicago, IL). Mean values and standard error were calculated by one-way ANOVA, and the means were compared by Duncan’s multiple range test at the level of P ≤ 0.05 (Duncan 1955).
Results
Embryogenic callus induction
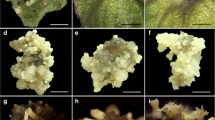
Properly sterilized fresh petiole explants were used for the induction of somatic embryogenesis. The half-strength MS medium fortified with 0.5 to 5.0 mg L−1 2,4-D or 0.5 to 8.0 mg L−1 NAA was used for the experiment. Off-white or cream-colored compact calluses were obtained from the cut edges of horizontally inoculated petiole explants after about 4 wk of culture in the half-strength MS medium supplemented with 1 mg L−1 2,4-D and 2 mg L−1 NAA (Fig. 1).
Embryogenic callus induction from petiole explants of Anaphyllum wightii Schott. (A) Embryogenic callus (naphthaleneacetic acid 2 mg L.−1). (B, C) Stereomicroscopic images of callus with globular and scutellar embryos (bar = 2 mm and 0.5 mm, respectively). (D) Isolated globular embryo (bar = 2 mm). (E) Scutellar embryo with scutellum (S), coleoptile (C), and root pole (Rp) (bar = 0.5 mm). (F) Coleoptilar embryo (bar = 1 mm).
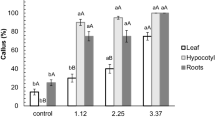
The calluses were subcultured on the same medium composition in which they were induced and were turned embryogenic after 2 wk of subculturing. The percentage of embryogenic callus induction from petiole explants was recorded for 2,4-D and NAA–supplemented medium after 45 d of culture (Table 1). The results showed that petiole explants exhibited the highest percentage of embryogenic callus induction (83.33 ± 8.80%) on half-strength MS medium containing 2 mg L−1 NAA. In the case of 2,4-D, used in half-strength MS medium, the highest percentage of embryogenic callus induction was observed at 1 mg L−1 (76.67 ± 8.82%). The embryogenic calluses obtained were repeatedly subcultured every 30 d for proliferation and maintenance of embryogenic cultures. Thus, the results indicated that NAA is superior to 2,4-D for embryogenic callus induction and maintenance. The various developmental stages of somatic embryos, such as globular, scutellar, and coleoptilar embryos, were observed under a stereomicroscope (Fig. 1).
Shoot regeneration from embryogenic callus cultures
After proliferation, the NAA- and 2,4-D-induced embryogenic calluses were transferred to full-strength MS medium containing 1 to 5 mg L−1 BAP separately for the maturation of embryos and shoot regeneration from the embryogenic calluses. The embryogenic callus cultures exhibited a good percentage of multiple shoot induction from the calluses within 2 wk (Fig. 2). The shooting response shown by BAP on 2,4-D- and NAA-induced embryogenic calluses are represented in Tables 2 and 3, respectively.
Multiple shoot regeneration from embryogenic callus of Anaphyllum wightii Schott. (A) Embryogenic callus. (B) Multiple shoot regeneration after 45 d on Murashige and Skoog medium (MS) containing 3 mg L−1 6-benzylaminopurine. (C) After 90 d. (D) In vitro root induction from shoot explants on half-strength MS medium containing 0.5 mg L−1 indole-3-butyric acid after 30 d. (E) In vitro rooted plantlet after 45 d. (F) Acclimatized plants after 4 wk.
For the 2,4-D-induced callus cultures, the maximum percentage of regeneration (100%), the highest number of shoots per callus, and maximum shoot length were recorded on full-strength MS medium containing 3 mg L−1 BAP after 90 d of culture. The maximum shoot number and shoot length obtained were 7.33 ± 0.33 and 3.66 ± 0.05 cm, respectively.
Full-strength MS medium containing 3 mg L−1 BAP gave the highest number of shoots per callus (6.00 ± 0.58), and 5 mg L−1 BAP showed a maximum shoot length of 5.34 ± 0.67 cm after 30 d of culture for NAA-induced calluses. The NAA-induced calluses were capable of 100% regeneration on all five concentrations of BAP. While comparing the responses obtained from 2,4-D- and NAA-induced callus cultures on medium supplemented with 1 to 5 mg L−1 BAP, it can be seen that NAA-induced calluses showed a higher number of shoots per callus and maximum shoot length in a short period of culture. However, the shoots that arose from 2,4-D-induced calluses were seen healthier with comparatively thicker petioles and wider leaf blades than those from NAA-induced callus cultures.
Histological analysis of embryogenic callus and embryos
Histological analysis of embryogenic callus revealed the presence of globular and coleoptilar embryos. Globular embryo consisted of an epidermis that differentiated it from the callus tissue and an independent vascular system without any connection to that of the callus. The coleoptilar stage of the embryo consisted of scutellum, coleoptile, shoot meristem, and root meristem (Fig. 3).
Histological observations of embryogenic callus and somatic embryos of Anaphyllum wightii Schott. (A) Longitudinal section of callus with globular embryos. (B, C) Longitudinal section of a globular embryo with the epidermis (Ep) and independent vascular system (Vs) viewed under a stereomicroscope (bar = 0.5 mm) and image analyzer. (D) Longitudinal section of coleoptilar embryo with scutellum (Sc), coleoptile (Cl), shoot meristem (Sm), and root meristem (Rm) (bar = 1 mm).
In vitro root induction
In vitro raised individual shoot explants were inoculated on half-strength MS medium containing 0.2 to 2 mg L−1 IAA, IBA, or NAA for root induction. The results obtained from in vitro rooting experiments are mentioned in Table 4.
Among the three auxins, IAA and IBA showed root induction after about 3 wk of culture, whereas NAA showed no response. IBA at 0.5 mg L−1 induced a better rooting response, after 45 d, in terms of the percentage of root induction (83.30 ± 3.30%), root number per shoot (4.33 ± 0.31), and root length (5.83 ± 0.93 cm) compared to other hormone concentrations used (Fig. 2).
Hardening and acclimatization
The hardening stage is an important step involved in every micropropagation experiment before introducing the in vitro regenerated plants to the natural environment. Since the tissue culture plants are grown under controlled temperature and humidity conditions in an aseptic environment, they will have a tendency of rapid water loss and wilting outside the culture room. Hence, the in vitro regenerated plantlets were initially covered with transparent polythene bags in order to maintain optimum humidity and watered once a week. After 4 wk, the polythene bags were removed, and the plants were transferred to greenhouse conditions. The plants were watered three times a week and were acclimatized with a survival rate of 76% (Fig. 2).
Discussion
Somatic embryogenesis is an efficient method for the mass propagation of many plant species and is also suitable for the regeneration of transgenic plants (Bhattacharyya et al. 2016). Plant regeneration through somatic embryogenesis is affected by various factors such as the genetic composition of plant material, the treatment of primary explants, and culture conditions (Mikuła and Rybczyński 2001). The culture medium has been reported to exert a more substantial effect on embryogenesis than other factors (Pavlović et al. 2013). Somatic embryos may arise either directly from the epidermal or sub-epidermal cells of explants (Moradi et al. 2017) or indirectly from an intervening callus phase induced from explants (Niknejad et al. 2011). The plant growth regulators and their concentration also influence the induction of somatic embryogenesis. Higher concentrations of auxins, especially 2,4-D, are used for inducing somatic embryos in some plant species (Raghavan 2004; Chen et al. 2010), whereas cytokinins at high concentrations are effective in other species (Kim et al. 2003).
Among the aroid species, plant regeneration through somatic embryogenesis was first reported in Anthurium andraeanum (Kuehnle et al. 1992). Later, several other aroids, including Colocasia esculenta (Deo et al. 2009), Dieffenbachia sp. (Shen and Lee 2009), and Spathiphyllum sp. (Zhao et al. 2012), were reported to be competent for induction of somatic embryogenesis. However, there were no previous reports of somatic embryogenesis induction in the genus Anaphyllum. Thus, the present work developed an easy and efficient protocol for inducing somatic embryogenesis in A. wightii for the first time. Embryogenic callus cultures possess the potential to proliferate for many generations and to develop somatic embryos (Hu et al. 2017; Thorat et al. 2017).
Blessy et al. (2021) reported the use of nodal portions of the rhizome as the explants for micropropagation of A. wightii. Since the overharvesting of the rhizome of this vulnerable species for its edible and medicinal purposes limits its use as planting material or explant, hence in the present study, the fresh petiole explants were used for the mass propagation of A. wightii through somatic embryogenesis. Hu et al. (2005) also reported the use of petiole explants for the induction of embryogenic calluses in the aroid Amorphophallus konjac. Even though the petiole explants exhibited embryogenic callus induction on half-strength MS medium containing both 2,4-D and NAA, the latter was observed to be superior for embryogenic callus induction and maintenance. Here, the type of embryogenesis observed is indirect somatic embryogenesis since an intervening callus phase is involved in the process. Similar results were obtained in the case of Anthurium andraeanum, where NAA was found to be more effective than other auxins, including 2,4-D, for the induction of embryogenic callus cultures from petiole explants of in vitro established plantlets (Pinheiro et al. 2014).
In the present work, the embryogenic callus was observed to contain developing somatic embryos at the globular, scutellar, and coleoptilar stages. This agrees with the general pattern of somatic embryogenesis in monocots, where the process of somatic embryo differentiation involves three major stages, namely globular, scutellar, and coleoptilar embryos (Burrieza et al. 2012). These stages of somatic embryos are also observed in other monocot species like Crocus sativus (Devi et al. 2014), Hordeum vulgare (Kachhwaha et al. 1997), and Phalaenopsis amabilis (Mose et al. 2017). Here, the formation of globular proembryos was observed as embryonic masses consisting of small, deeply stained meristematic cells resulting from repeated cell division. The proembryos later differentiated into large globular-shaped embryos with a protoderm differentiating it from the callus tissue which is a characteristic feature of somatic embryo development (Yeung 1995; Arnold et al. 2002).
In the next stage of development, the globular embryo forms a notch at the terminal position, similar to that reported in ginger (Lincy et al. 2009), leading to the scutellar stage (de Alcantara et al. 2014). The scutellar embryo consisted of two protrusions at the apical part and a notch between them. The shoot meristem develops from the scutellar notch and becomes enclosed by the coleoptile. The root meristem will also be formed at this stage in the basal part of the embryo (Mose et al. 2017). The matured coleoptile embryo was characterized by the development of a scutellum, coleoptile, and shoot and root meristems. Here, the scutellum was found to be slightly greenish color, which was similar to that in the case of Hordeum vulgare (Kachhwaha et al. 1997).
The cytokinin BAP was found to be effective for multiple shoot induction, as reported previously in the same species. IBA at 0.5 mg L−1 induced a better rooting response compared to other auxins used, which also agrees with the findings of Blessy et al. (2021). The in vitro regenerated plants were successfully acclimatized with a good survival rate of 76%, which was better than that obtained in a previous study (65%) on the same species (Blessy et al. 2021).
Conclusions
Somatic embryogenesis serves as a tool for the large-scale propagation of plants and is also an effective method for studying the regulation of embryo development. The present study developed a successful protocol for the in vitro plant regeneration of the endemic and ethnomedicinal plant Anaphyllum wightii. Here, the somatic embryos were obtained by the process of indirect somatic embryogenesis due to the presence of an intervening callus phase. In A. wightii, the process of embryo development was asynchronous since the embryogenic callus consisted of embryos at various stages of development adjacent to each other. Hence, it can be concluded that the auxins 2,4-D and NAA are effective in the induction of somatic embryogenesis using the petiole explants of A. wightii, which may lead to the conservation of this endemic plant. The present study may also be helpful in studying the pattern of embryo development in this species since the developmental stages of somatic embryos are often similar to that of zygotic embryos.
References
Ahmedullah M, Nayar M (1986) Flora of India series IV. In: Sharma BD, Modak PD, Agarwal VS, Pal SC, Balakrishnan NP (eds) Endemic plants of the Indian region, Peninsular India, volume I. Botanical Survey of India, Calcutta, pp 205–208
Arnold SV, Sabala I, Bozhkov P, Daychok FL (2002) Developmental pathways of somatic embryogenesis. Plant Cell Tiss Org Cult 69:233–249
Bhattacharyya P, Kumaria S, Job N, Tandon P (2016) En-masse production of elite clones of Dendrobium crepidatum: a threatened, medical orchid used in traditional Chinese medicine (TCM). J Appl Res Med Aromat Plants 3:168–176
Blessy R, Anami Augustrus Arul A, Ani Besant S, Catherine Sheeja V, Alexander HJ, Jancy Rani G (2021) Micropropagation of an endangered medicinal plant Anaphyllum wightii Schott. through nodal culture. Int J Bot Stud 6:210–214
Burrieza HP, Lopez-Fernandez MP, Chiquieri TB, Silveira V, Maldonado S (2012) Accumulation pattern of dehydrins during sugarcane (Var. SP80.3280) somatic embryogenesis. Plant Cell Rep 31:2139–2149
Chen AH, Yang JL, Da Niu Y, Yang CP, Liu GF, Yu CY, Li CH (2010) High-frequency somatic embryogenesis from germinated zygotic embryos of Schisandra chinensis and evaluation of the effects of medium strength, sucrose, GA3, and BA on somatic embryo development. Plant Cell Tiss Org Cult 102:357–364
de Alcantara GB, Dibax R, Oliveira RAD, Filho JCB, Daros E (2014) Plant regeneration and histological study of the somatic embryogenesis of sugarcane (Saccharum spp.) cultivars RB855156 e RB72454. Acta Sci Agron 36:63–72
Deo PC, Harding RM, Taylor M, Tyagi AP, Becker DK (2009) Somatic embryogenesis, organogenesis and plant regeneration in taro (Colocasia esculenta var. esculenta). Plant Cell Tiss Org Cult 99:61–71
Devi K, Sharma M, Ahuja PS (2014) Direct somatic embryogenesis with high frequency plantlet regeneration and successive cormlet production in saffron (Crocus sativus L.). S Afr J Bot 93:207–216
Elhiti M, Stasolla C, Wang A (2013) Plant molecular regulation of plant somatic embryogenesis. In Vitro Cell Dev Biol – Plant 49:631–642
Hu JB, Liu J, Yan HB, Xie CH (2005) Histological observations of morphogenesis in petiole derived callus of Amorphophallus rivieri Durieu in vitro. Plant Cell Rep 24:642–648
Hu R, Sun Y, Wu B, Duan H, Zheng H, Hu D, Lin H, Tong Z, Xu J, Li Y (2017) Somatic embryogenesis of immature Cunninghamia lanceolata (Lamb.) hook zygotic embryos. Sci Rep 7:1–14
Kachhwaha S, Varshney A, Kothari SL (1997) Somatic embryogenesis and long-term high plant regeneration from barley (Hordeum vulgare L.) using picloram. Cereal Res Commun 25:117–126
Kim SW, Oh SC, Liu JR (2003) Control of direct and indirect somatic embryogenesis by exogenous growth regulators in immature zygotic embryo cultures of rose. Plant Cell Tiss Org Cult 74:61–66
Kuehnle AR, Chen F-C, Sugii N (1992) Somatic embryogenesis and plant regeneration in Anthurium andraeanum hybrids. Plant Cell Rep 11:438–442
Kunjumon M, Thomas S, George RE, Thankamani VI (2016) Phytochemical, antibacterial, and antifungal activity of rhizome from Anaphyllum wightii. Schott against clinical isolates and plant pathogens. Int J Phytomed 7:459–467
Lincy AK, Remashree AB, Sasikumar B (2009) Indirect and direct somatic embryogenesis from aerial stem explants of ginger (Zingiber officinale Rosc.). Acta Bot Croat 68:93–103
Mathew J, George KV (2013) Medicinal plants used by Malampadaram tribals in Achankovil forests of Kerala to cure snake bite, wound, and bone fracture. Life Sci Leafl 8:50–54
Mikuła A, Rybczyński JJ (2001) Somatic embryogenesis of Gentiana genus I. The effect of the preculture treatment and primary explant origin on somatic embryogenesis of Gentiana cruciata (L.), G. pannonica (Scop.), and G. tibetica (King). Acta Physiol Plant 23:15–25
Moradi S, Daylami SD, Arab M, Vahdati K (2017) Direct somatic embryogenesis in Epipactis veratrifolia, a temperate terrestrial orchid. J Hortic Sci Biotechnol 92:88–97
Mose W, Indrianto A, Purwantoro A, Semiarti E (2017) The influence of thidiazuron on direct somatic embryo formation from various types of explant in Phalaenopsis amabilis (L.) blume orchid. Hayati J Biosci 4:201–205
Murashige T, Skoog FA (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Niknejad A, Kadir MA, Kadzimin SB (2011) In vitro plant regeneration protocorms like bodies (PLBs) and callus of Phalaenopsis gigantea (Epidendroideae: Orchidaceae). Afr J Biotechnol 10:11808–11816
Pavlović S, Vinterhalter B, Zdravković-Korać S, Vinterhalter D, Zdravković J, Ćosić T, Mitić N, Cingel A, Savić J, Bohanec B, Ninković S (2013) In vitro plant regeneration from immature zygotic embryos and repetitive somatic embryogenesis in kohlrabi (Brassica oleracea var. gongylodes). In Vitro Cell Dev Biol - Plant 49:294–303
Pinheiro MVM, Martins FB, Cruz ACFD, Carvalho ACPPD, Oliveira EJD, Otoni WC (2014) Somatic embryogenesis in anthurium (Anthurium andraeanum cv. Eidibel) as affected by different explants. Acta Sci Agron 36:87–98
Raghavan V (2004) Role of 2,4-dichlorophenoxyacetic acid (2,4-D) in somatic embryogenesis on cultured zygotic embryos of Arabidopsis: cell expansion, cell cycling, and morphogenesis during continuous exposure of embryos to 2,4-D. Am J Bot 91:1743–1756
Rajasekharan PE, Wani SH (Eds.) (2020) Conservation and utilization of threatened medicinal plants. Springer International Publishing, Switzerland
Ramachandran VS (2007) Wild edible plants of the Anamalais, Coimbatore District, Western Ghats, Tamil Nadu. Indian J Tradit Knowl 6:173–176
Shen R, Lee N (2009) Cytokinins stimulate somatic embryogenesis and plant regeneration from the male inflorescence of Dieffenbachia ‘Tiki.’ J Agri Assoc Taiwan 10:380–388
Thorat AS, Sonone NA, Choudhari VV, Devarumath RM, Babu KH (2017) Plant regeneration from cell suspension culture in Saccharum officinarum L. ascertaining of genetic fidelity through RAPD and ISSR markers. Biotech 7:16
Udayan PS, George S, Tushar KV, Indira B (2007) Ethnomedicine of Malampandaram tribes of Achankovil forest of Kollam District, Kerala. Indian J Tradit Knowl 6:569
Yeung EC (1995) Structural and developmental patterns in somatic embryogenesis. In: Thorpe TA (ed) In vitro embryogenesis in plants. Kluwer Publishers, Dordrecht, Netherlands, pp 205–247
Zhao J, Cui J, Liu J, Liao F, Henny RJ, Chen J (2012) Direct somatic embryogenesis from leaf and petiole explants of Spathiphyllum ‘Supreme’ and analysis of regenerants using flow cytometry. Plant Cell Tiss Org Cult 110:239–249
Funding
This study received financial support from the Council of Scientific and Industrial Research (CSIR).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lekshmi, S., Swapna, T.S. In vitro plant regeneration through somatic embryogenesis in Anaphyllum wightii Schott. In Vitro Cell.Dev.Biol.-Plant 58, 1099–1106 (2022). https://doi.org/10.1007/s11627-022-10308-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-022-10308-2