Abstract
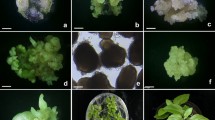
Long term high plant regeneration was obtained from embryogenic callus induced from immature embryo explants of Hordeum vulgare cv RD-57 cultured on MSB medium supplemented with Picloram (2 and 4mg I-1). In the subsequent subcultures different types of calli were recognised which could be distinguished from each other on the basis of their morphology, growth and plant regeneration ability. Best growth and maintenance of embryogenic state of callus was achieved on MSB medium supplemented with Picloram (2 mgl-1). For plant regeneration, embryogenic callus was transferred to hormone free MSB medium. Regenerated plantlets were rooted either on hormone free MSB medium or MSB medium supplemented with IAA (0.5 mgl-1). The regenerated plants were transferred to field conditions where they matured and set seeds. Histological studies of embryogenic callus revealed all the stages of typical somatic embryogenesis. In addition, atypical somatic embryos and secondary somatic embryogenesis was also observed and occasionally de novo shoot morphogenesis was seen in the same cultures.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bhaskaran S & Smith RH (1990) Regeneration in cereal tissue culture: A review. Crop Sci. 30: 1328–1337
Bhattacharya S (1991) Plant regeneration from mature embryo derived callus of certain barley cultivars. Phytomorphol. 41: 121–127
Botti C & Vasil IK (1983) Plant regeneration by somatic embryogenesis from parts of cultured mature embryos of Pennisetum americanum (L.) K. Schum. Z. Pflanzenphysiol. 111: 319–325
Bregitzer P (1991) Plant regeneration and callus type in barley: effects of genotypes and culture medium. Crop Sci. 32: 1108–1112
Cheng, TV & Smith RH (1975) Organogenesis from callus culture of Hordeum vulgare. Planta 123: 307–310
Dunstan DI, Short KC & Thomas E (1978) The anatomy of secondary morphogenesis in cultured scutellum tissues of Sorghum bicolor. Protoplasma 97: 251–260
Dunstan DI, Short KC, Dhaliwal H & Thomas E (1979) Further studies on plantlet production from cultured tissues of Sorghum bicolor. Protoplasma 101: 355–361
Eapen S & George L (1989) High frequency plant regeneration through somatic embryogenesis in finger millet (Eleusine coracana (L.) Gaertn). Plant Sci. 61: 127–130
Gamborg OL, Miller RA & Ozima K (1968) Culture methods and detection of glucanases in suspension cultures of wheat and barley. Can. J. Biochem. 46: 417–421.
Goldstein CS & Kronstad WE (1986) Tissue culture and plant regeneration from immature embryo explants of barley, Hordeum vulgare. Theor. Appl. Genet. 71: 631–636.
He DG, Yang YM, Bertram J & Scott KJ (1990) The histological development of the regenerative tissue derived from cultured immature embryos of wheat (Triticum aestivum L). Plant Sci. 68: 103–111
Ho WJ & Vasil IK (1983) Somatic embryogenesis in sugarcane (Saccharum officinarum L). The morphology and physiology of callus formation and the ontogeny of somatic embryos. Protoplasma 118: 169–180.
Jahne A, Lazzeri PA, Jager-Gussen, M & Lorz H (1991) Plant regeneration from embryogenic cell suspensions derived from anther cultures of barley (Hordeum vulgare L). Theor. Appl. Genet. 82: 74–80.
Jelaska S, Renzel Z & Cesar V (1984) Plant regeneration from mesocotyl callus of Hordeum vulgare L. Plant Cell Rep. 3: 125–129.
Johansen DA (1940) Plant Microtechnique, McGraw-Hill Book Company Inc., New York
Jones T & Rost TL (1989) The developmental anatomy and ultrastructure of somatic embryos from Rice (Oryza sativa L.) scutellum epithelial cells. Bot. Gaz. 150: 41–49
Kachhwaha S & Kothari SL (1994a) Plant regeneration in barley through microtillering and multiple shoot differentiation. Curr. Sci. 67: 116–118.
Kachhwaha S & Kothari SL (1994b) Mode of plant regeneration from immature embryo cultures of Hordeum vulgare L. Acta Bot. Indica 22: 232–235.
Kachhwaha S, & Kothari SL (1996) Plant regeneration from immature embryo explants of kordeum spontaneum and Hordeum vulgare. Cereal Res. Comm. 24: 27–32.
Kothari SL & Chandra N (1988) Somatic embryogenesis and plant regeneration from seed callus of barley (Hordeum vulgare L.). Curr. Sci. 57: 1351–1352.
Kothari SL & Chandra N (1995) Advances in tissue culture and genetic transformation of cereals. J. Ind. Bot. Soc. 74: 323–342.
Kothari SL, Sharma V & Chandra N (1994a) Variation in morphogenetic patterns of plant regeneration in seed callus of Guinea grass (Panicum maximum Jacq.). J. Ind. Bot. Soc. 73: 203–208.
Kothari SL, Sivadas P & Chandra N (1994b) Differentiation and development of somatic embryos in suspension cultures and plantlet formation in Pennisetum americanum. J. Ind. Bot. Soc. 73: 233–236.
Kott LS & Kasha KJ (1984) Initiation and morphological development of somatic embryoids from barley cell cultures. Can. J. Bot. 62: 1245–1249
Lu CY & Vasil IK (1985) Histology of somatic embryogenesis in Panicum maximum (Guinea grass). Amer. J. Bot. 72: 1908–1913
Luhrs R & Lorz H (1987) Plant regeneration in vitro from embryogenic cultures of spring and winter type barley (Hordeum vulgare L.) varieties. Theor. Appl. Genet. 75: 16–25.
Luhrs R & Lorz H (1988) Initiation of morphogenic cell suspension and protoplast cultures of barley (Hordeum vulgare L.). Planta 175: 71–81
Lupotto E (1984) Callus induction and plant regeneration from barley mature embryos. Ann. Bot. 54: 523–529.
McCain JW & Hodges TK (1986) Anatomy of somatic embryos from maize embryo cultures. Bot. Gaz. 147: 453–460.
Murashige T & Skoog F (1962) A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol. Plant. 15: 473–497.
Oinam GS & Kothari SL (1995) Totipotency of coleoptile tissue in indica rice (Oryza sativa L. cv. CH-1039) Plant Cell Rep. 14: 245–248.
Orton J (1979) A quantitative analysis of growth and regeneration from tissue cultures of Hordeum vulgare, H. jubatum and their interspecific hybrid. Env. Exp. Bot. 19: 319–335.
Ozias-Akins P & Vasil IK (1982) Plant regeneration from cultured immature embryos and ai]inflorescences of Triticum aestivum L. (wheat):Evidence for somatic embryogenesis. Protoplasma 110: 95–105.
Seguin-Swartz G, Kott L & Kasha KJ (1984) Development of haploid cell lines from immature barley, Hordeum vulgare, embryos. Plant Cell Rep. 3: 95–97.
Sharma V, Kothari SL & Chandra N (1989) In vitro regeneration field transfer of plantlets and growth to maturity of plants of Sorghum bicolor (L.) Moench. Curr. Sci. 58: 586–588.
Sharma VK, Rao A, Varshney A & Kothari SL (1995) Comparison of developmental stages of inflorescence for high frequency plant regeneration in Triticum aestivum L. and T. durum Desf. Plant Cell Rep. 15: 227–231.
Sivadas P, Chandra N & Kothari SL (1992) Histology of somatic embryogenesis and shoot bud formation in callus cultures of finger millet, Eleusine coracana (L.). Gaertn. Phytomorphol. 42: 203–207.
Thomas MR & Scott KJ (1985) Plant regeneration by somatic embryogenesis from callus initiated from immature embryos and immature inflorescence of Hordeum vulgare L. J Plant Physiol 121: 159–169.
Vasil IK (1994) Molecular improvement of cereals. Plant Mol. Biol. 25: 925–937.
Vasil V & Vasil IK (1982) The ontogeny of somatic embryos of Pennisetum americanum (L) K. Schum. In cultured immature embryos. Bot. Gaz. 143: 454–465.
Vasil V, Lu CY & Vasil IK (1985) Histology of somatic embryogenesis in cultured immature embryos of maize (Zea mays L). Protoplasma 127: 1–8.
Wakizuka T & Yamaguchi T (1987) The induction of enlarged apical domes in vitro and multi-shoot formation from finger millet (Eleusine coracana). Ann. Bot. 60: 331–336.
Weighel RC & Hughes KW (1985) Long term regeneration by somatic embryogenesis in barley (Hordeum vulgare L) tissue cultures derived from apical meristem explants. Plant Cell Tissue Org. Cult. 5: 151–162.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kachhwaha, S., Varshney, A. & Kothari, S.L. Somatic Embryogenesis and Long Term High Plant Regeneration from Barley (Hordeum Vulgare L.) Using Picloram. CEREAL RESEARCH COMMUNICATIONS 25, 117–126 (1997). https://doi.org/10.1007/BF03543446
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03543446