Abstract
Purpose
This study aims to compare the effectiveness of Billroth-II with Braun and Roux-en-Y reconstruction after laparoscopic distal gastrectomy.
Methods
From April 2010 to August 2012, 66 patients underwent laparoscopic distal gastrectomy (Billroth-II with Braun reconstruction, 26; Roux-en-Y, 40). The patients’ data were collected prospectively and reviewed retrospectively.
Results
The mean operation and reconstruction times were statistically shorter for Billroth-II with Braun reconstruction than Roux-en-Y (198.1 ± 33.0 vs. 242.3 ± 58.1 min, p = 0.001). One case of postoperative stricture was observed in each group. One case each of intra-abdominal abscess and delayed gastric emptying occurred in the Billroth-II with Braun group. At 1 year postoperatively, gastric residue and reflux esophagitis were not significantly different between the groups. Gastritis and bile reflux were more frequently observed in the Billroth-II with Braun group (p = 0.004 and p < 0.001, respectively). At 2 years postoperatively, gastric residue was not significantly different, but gastritis, bile reflux, and esophagitis were more frequent in the Billroth-II with Braun group (p = 0.029, p < 0.001, and p = 0.036, respectively).
Conclusion
The postoperative effectiveness of Roux-en-Y reconstruction may be superior to Billroth-II with Braun reconstruction after laparoscopic distal gastrectomy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
From the results of several prospective and retrospective studies, laparoscopic gastrectomy with D1+ lymph node dissection has been accepted as a standard treatment option for early gastric cancer.1 – 4 Recent advances in laparoscopic surgical instruments and skills have extended the surgical range; it is even possible to perform more than D2 lymph node dissection. As the 5-year survival rate of patients with early gastric cancer is >90 %, consequently, the interests of surgeons and patients have shifted to quality of life (QoL) beyond the curability of cancer.5 , 6 In accordance with these trends, minimally invasive or function-preserving surgery such as sentinel lymph node navigation surgery and pylorus preserving gastrectomy is now being investigated. These surgeries are expected to improve QoL postoperatively by preserving gastric function and obtaining a safe oncologic specimen.7 , 8
After distal gastrectomy, which is performed for tumors located in the lower two thirds of the stomach, mostly Billroth-I (B-I), Billroth-II (B-II), and Roux-en-Y (REY) reconstruction are used to maintain bowel continuity.9 Duodenogastric bile reflux due to removal of the pylorus is one of the major factors that affect patient’s QoL.10 In addition, this bile reflux is a potential risk factor for malignant changes in the remnant stomach and lower esophagus.11 – 16
Although B-I reconstruction is commonly used because of its simplicity and physiologic advantages, surgeons have to choose B-II or REY reconstruction if the remnant stomach or duodenum becomes short due to extensive resection to ensure the safety margin. REY reconstruction has been reported as a procedure in which bile reflux rarely occurs, and it has become more popular in Korean and Japan.17 Nevertheless, B-II reconstruction is still popular as an alternative procedure because REY reconstruction is regarded as the time-consuming and more complicated procedure with a high morbidity. However, bile reflux occurs frequently after B-II due to its structural drawback. For this reason, Braun anastomosis was designed to decrease this bile inflow to the stomach. Braun anastomosis will be a good substitute for REY if it plays a dependable role in bile passage.
Even though some studies have compared B-I with B-II or REY in terms of postoperative outcome such as morbidity or the nutritional result, there are few comparative reports directly comparing B-II with Braun (B-IIB) and REY reconstruction. In the present study, we evaluated which reconstruction method is more superior in regard to improving patients’ QoL, especially in terms of bile reflux and reflux esophagitis according to postoperative endoscopic findings.
Materials and Methods
Patients
This study was designed retrospectively to compare B-IIB to REY reconstruction after laparoscopic distal gastrectomy in terms of patients’ QoL. Between March 2010 and October 2012, 485 patients underwent laparoscopic distal gastrectomy for gastric cancer at the Department of Gastrointestinal Surgery at Pusan National University Hospital. The preoperative clinical stage was evaluated for all patients with endoscopic ultrasonography and abdominal computed tomography. Preoperatively, all patients were histologically diagnosed with gastric adenocarcinoma by endoscopic biopsy. Endoscopic clipping was performed preoperatively to identify the lesion during the operation in all patients.
Two surgeons participated in this study, and laparoscopic gastrectomy was performed for patients with clinical Stage IA or IB gastric cancer as defined by the seventh edition of the American Joint Commission on Cancer Staging System. Of these patients, 413 who underwent B-I reconstruction and 6 who underwent B-II reconstruction were excluded from this study. Of the remaining 66 patients, 26 underwent B-IIB and 40 underwent REY reconstruction. All data were retrieved prospectively, and we analyzed the following variables retrospectively: (1) patients’ demographics (i.e., age, sex, co-morbidities, body mass index (BMI), American Society of Anesthesiologists (ASA) score); (2) perioperative and clinicopathologic data (i.e., the operative time, bleeding loss, tumor size, proximal/distal margin, invasion depth, nodal status, hospital stay, and morbidity); and (3) endoscopic findings (preoperatively and postoperatively at 1 and 2 years).
Operative Procedure
Laparoscopic Distal Gastrectomy with Lymph Node Dissection
Conventional laparoscopic distal gastrectomy was performed by two skilled surgeons who have performed laparoscopic gastrectomy on >200 cases. All patients underwent D2 lymph node dissection (1, 3, 4sb, 4d, 5, 6, 7, 8a, 9, 11p, 12a, and ±14v), which was defined by the Japanese Gastric Cancer Association,18 and extracorporeal anastomosis after distal gastrectomy was routinely performed. Four vessels of major gastric blood supply (i.e., the right/left gastroepiploic and right/left gastric vessels) were ligated if the tumor was located in the mid-third of the stomach, and the first short gastric branch was ligated. The duodenum was divided by a laparoscopic linear stapler, and a reinforcement suture was not used at the duodenal stump. All reconstructions were performed extracorporeally.
Gastric Division
In all patients, the endoscopic clipping was performed preoperatively for identification of the lesion during the operation. In the B-II with Braun anastomosis, a gastrotomy was made first at anterior gastric wall. Through this gastrotomy, the circular stapler was introduced and gastrojejunostomy was performed with the jejunum at which an anvil was inserted. Then, stomach was divided linear stapler extracorporeally. In the REY reconstruction, the stomach was divided first with linear stapler.
B-II with Braun Reconstruction
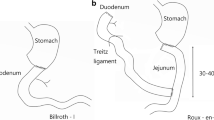
After inserting the anvil into the jejunum (50 cm distal from the Treitz ligament), a gastrotomy was performed in the anterior gastric wall. Through this gastrotomy, iso-peristaltic gastrojejunostomy was performed with a circular stapler (Premium Plus CEEA 25; US Surgical, Norwalk, CT). Then, the stomach was divided with a linear stapler. Additionally, jejunojejunostomy was conducted 35 cm from where the gastrojejunostomy was performed by manual extracorporeal suturing with absorbable monofilament fiber. The diameter of jejunojejunostomy was about 2 cm (Fig. 1a). And Petersen’s space was closed by intracorporeal suturing.
REY Reconstruction
The stomach was first divided upon identifying the lesion. And the anvil was inserted into the greater curvature tip of the remnant stomach. The Roux limb was made by dividing the jejunum 15 cm from the Treitz ligament. Then iso-peristaltic gastrojejunostomy was performed with a circular stapler (Premium Plus CEEA 25; US Surgical, Norwalk, CT) through the anterocolic route, and the jejunal stump was closed with a linear stapler. Jejunojejunostomy was conducted 35 cm from where the gastrojejunostomy performed by same method with B-IIB reconstruction (Fig. 1b). Petersen’s space was closed by intracorporeal suturing similar to that performed in B-IIB reconstruction. In addition, the potential site of the hernia at the mesentery of jejunojejunostomy was closed by extracorporeal manual suturing.
Interpretation of the Endoscopic Findings
Esophagogastroduodenoscopy was performed and the results were interpreted preoperatively and at 1 and 2 years postoperatively by an experienced endoscopist. The endoscopic findings were analyzed using the RGB classification, which includes the gastric residue, degree of gastritis, and bile reflux (Table 1). Moreover, reflux esophagitis was evaluated using the Los Angeles classification.
Statistical Analysis
All variables were analyzed using SPSS, version 19.0 for Windows (SPSS, Inc., Chicago, IL). Categorical variables were analyzed by the chi-square test and Fisher’s exact test. Continuous variables were analyzed by the Student’s t test and expressed as means ± standard deviations. To analyze the RGB classification, the gastric residue and degree of gastritis were calculated between grade 0 and others to achieve statistical validity of the chi-square test. For the Los Angeles classification of reflux esophagitis, data were calculated between none and others. A p value <0.05 was considered statistically significant.
Results
Patients’ demographic data are presented in Table 1. Overall, the mean age and BMI were 58.2 ± 10.1 years and 23.5 ± 2.2 kg/m2, respectively. There were no significant differences in terms of the patients’ age, sex, and BMI between the B-IIB and REY groups, except the ASA score (Table 2).
Each group did not show significant differences in the pathologic characteristics such as the tumor size, margin, and stage. Blood loss was 161.7 ± 146.6 mL in the B-IIB group compared with 245.0 ± 207.0 mL in the REY group, but this was not statistically significant. In the REY group, the operative time was statistically significantly longer than that in the B-IIB group (198.1 ± 33.0 vs. 242.3 ± 58.1 min, p = 0.001). There was no anastomotic leakage and bleeding, but one case of postoperative stricture was observed in each group. Additionally, one case of intra-abdominal abscess and one case of delayed gastric emptying occurred in the B-IIB group (Table 3).
Comparison of Endoscopic Findings According to the Postoperative Period
All data were calculated with Pearson’s chi-square test. Gastric residue was not significantly different between the B-IIB and REY groups, according to the postoperative period. Preoperatively, there were no significant differences between the two groups in terms of the gastric residue, gastritis, and bile reflux. However, at 1 and 2 years postoperatively, gastritis and bile reflux were more frequently observed in the B-IIB group (Table 4; after 1 year: p = 0.001, p < 0.001; after 2 years: p = 0.002, p < 0.001, respectively).
The modified Los Angeles classification was used to evaluate the grade of reflux esophagitis. There were no significant differences in the incidence preoperatively and at 1 year postoperatively, whereas at 2 years postoperatively, reflux esophagitis was more frequent in the B-IIB group (Table 5, p = 0.017).
Discussion
Gastroesophageal reflux, which prevents the intake of a regular diet, is one of the important factors that significantly decrease QoL during the postoperative period. Thus, many researchers have paid attention to these reflux symptoms. Gastroduodenostomy (B-I), gastrojejunostomy (B-II), and REY anastomosis have been mainly performed as a reconstruction method for intestinal continuity after distal gastrectomy. Although REY reconstruction has recently focused on decreasing bile reflux, the most optimal reconstructive procedures after distal gastrectomy remain unclear.
B-I has been chosen as the primary reconstruction methods after distal gastrectomy, as it has a simple procedure and physiologic feature that does not require the intestine to be bypassed. Other reconstruction methods such as B-II or REY should be considered in cases in which the stomach or duodenum need to be largely resected to obtain a safe surgical margin. In 2009, the Korea Gastric Cancer Association reported in a nationwide survey that B-I, B-II, and REY reconstructions were performed in 63.4, 33.1, and 3.3 % of cases, respectively, after distal gastrectomy.19 Fukuhara et al. reported that REY reconstruction is superior to B-I and B-II in terms of preventing bile reflux and gastritis.20 , 21 Namikawa et al. also recommended REY reconstruction over B-I reconstruction after distal gastrectomy to reduce bile reflux and improve a patient’s QOL.22 Recently, a Japanese research group reported that they prefer using REY reconstruction as alternative methods to substitute for B-I reconstruction.23 However, surgeons still tend to choose B-II reconstruction when B-I reconstruction is not applicable. This may be because of concern for high morbidity rates because REY reconstruction is complicated procedure to require more anastomoses and stump.
B-II reconstruction has an inevitable structural disadvantage, as bile flows into the remnant stomach. Thus, jejunojejunostomy between the afferent (A) and efferent (E) loops, i.e., Braun anastomosis, has been suggested to overcome this limitation. Vogel et al. reported that Braun anastomosis improved bile reflux effectively compared with B-II only.24 It is expected that bile reflux may be decreased through Braun anastomosis by redirecting the outflow of bile to the E-loop. If the function of Braun anastomosis, especially in terms of the bile stream, is associated with an acceptable result, B-IIB can be a good alternative reconstruction method to REY in theory. However, few reports have compared B-IIB with REY reconstruction.
Kubo et al. suggested a new endoscopic grading system to evaluate the remnant stomach, i.e., the RGB classification, which provides scores for gastric residue, gastritis, and bile reflux.25 In the current study, there was no difference in gastric residue at the preoperative evaluation and at 1 and 2 years postoperatively between the B-IIB and REY groups; however, gastritis and bile reflux were more frequently observed at 1 and 2 years postoperatively in the B-IIB group. This result fails to show the effect of bile diversion into the E-loop in B-IIB reconstruction. Consequently, this would cause reflux esophagitis over time. Figure 2 shows 1-year postoperative endoscopic findings in fourteenth patient who underwent B-IIB reconstruction after laparoscopic distal gastrectomy. The patient has complained epigastric soreness and intermittent dyspepsia. He had reflux esophagitis of grade B and severe gastritis with bile reflux. Those findings were not changed at 2-year postoperative endoscopic findings as well.
Postoperative endoscopic findings at 1 year after Billroth-II with Braun reconstruction (fourteenth patient). a Grade B reflux esophagitis showing mucosal break longer than 5 mm (Los Angeles classification system). b Severe redness due to gastritis (G, grade 3). c, d Bile reflux (B, grade 1) into the stomach with gastritis. There was no gastric residue (R, grade 0)
Lee et al. reported that gastritis and bile reflux was significantly more frequently associated with B-IIB reconstruction, and there was no difference in the RGB score between B-I and B-IIB reconstruction in their study that compared 1-year postoperative endoscopic findings among B-I, B-IIB, and REY reconstructions.26 In addition, Park et al. demonstrated that bile reflux had a high incidence (43.3 %) in B-IIB, and REY was a superior reconstruction method to B-IIB after distal gastrectomy.27
Although we were not able to determine the exact reason why the diversion of bile failed, it was considered to be due to the following reasons: (1) some bile still flows into the stomach because the pressure from the A-loop to the remnant gastric cavity is lower than that to the E-loop; and (2) it may be difficult to block the bile flow because the peristaltic direction of the A-loop is toward the remnant stomach. Thus, uncut REY reconstruction, which occludes the jejunogastric pathway, has been suggested as an alternative method by some surgeons. Previous studies have reported that uncut REY shows less frequent Roux stasis, which is one of the main problems of REY reconstruction.28 , 29 However, the exact mechanism of Roux stasis is debatable.
In the perioperative data, REY reconstruction had no specific disadvantage compared with B-IIB reconstruction except a longer operation time and Roux stasis did not occur in all REY cases. Postoperative complications were more frequent in the B-IIB group, although this was not statistically significant. Nevertheless, as aforementioned, some surgeons still may consider REY reconstruction a more complicated procedure than B-I or B-II reconstruction. Thus, they may hesitate to perform REY reconstruction; however, a various reports have suggested that there are no differences in the postoperative complication among each method of reconstruction because of the recent developments and improvements of surgical instruments and skill.9 , 10 , 26 , 30 Some surgeons may prefer B-II reconstruction to REY reconstruction in terms of preventing the dumping syndrome and weight loss. However, nutritional outcomes, including weight loss, are not significantly different between B-IIB and REY reconstruction.26
This was a retrospective study with a relatively short study period. Consequently, the enrolled sample size was not large because of a high performance rate of B-I reconstruction after distal gastrectomy. Additionally, because this study focused on endoscopic findings related to enterogastric bile reflux after distal gastrectomy, we could not obtain more data on the global health status, symptoms, activity, and emotional status of postoperative patients by using the tools for evaluating patient’s QoL such as the European Organization for Research and Treatment of Cancer questionnaire, Gastrointestinal Quality of Life Index, or EuroQol. If correlations between these various additional results related to the patients’ QoL and the RGB score was analyzed, comprehensive results could have been drawn from this study. Although a hepatobiliary scan helps identify enterogastric bile reflux, endoscopy is a more effective modality for evaluating the function of the remnant stomach, including direct postoperative changes such as esophagogastritis. In addition, the present study had a comparative design, and all the medical records were collected prospectively, although it was not a large-scale study. Additionally, many surgeons currently have great interest in totally laparoscopic gastrectomy, and they prefer B-II or REY reconstruction as the intracorporeal anastomosis procedure because B-I reconstruction (delta anastomosis) is technically difficult. Thus, our findings can be another significant reference for re-evaluating B-IIB reconstruction, and for surgeons to choose the appropriate procedure to manage patients after distal gastrectomy in the long-term because there were few reports to directly compared B-IIB with REY reconstruction.
Conclusion
REY reconstruction may be superior to B-IIB reconstruction in terms of the incidence of postoperative gastritis, bile reflux, and reflux esophagitis after laparoscopic distal gastrectomy, and B-IIB reconstruction may not effectively prevent intragastric bile flow.
References
Kitano S, Iso Y, Moriyama M, Sugimachi K. Laparoscopy-assisted Billroth I gastrectomy. Surg Laparosc Endosc. 1994;4(2):146–8.
Kitano S, Shiraishi N, Fujii K, Yasuda K, Inomata M, Adachi Y. A randomized controlled trial comparing open vs laparoscopy-assisted distal gastrectomy for the treatment of early gastric cancer: an interim report. Surgery. 2002;131(1 Suppl):S306-11.
Lee JH, Han HS, Lee JH. A prospective randomized study comparing open vs laparoscopy-assisted distal gastrectomy in early gastric cancer: early results. Surg Endosc. 2005;19(2):168–73.
Kim HH, Hyung WJ, Cho GS, Kim MC, Han SU, Kim W et al. Morbidity and mortality of laparoscopic gastrectomy versus open gastrectomy for gastric cancer: an interim report—a phase III multicenter, prospective, randomized Trial (KLASS Trial). Ann Surg. 2010;251(3):417–20.
Oh SY, Kwon S, Lee KG, Suh YS, Choe HN, Kong SH et al. Outcomes of minimally invasive surgery for early gastric cancer are comparable with those for open surgery: analysis of 1,013 minimally invasive surgeries at a single institution. Surg Endosc. 2014;28(3):789–95.
Yang HK, Suh YS, Lee HJ. Minimally invasive approaches for gastric cancer-Korean experience. J Surg Oncol. 2013;107(3):277–81.
Hiki N, Nunobe S, Kubota T, Jiang X. Function-preserving gastrectomy for early gastric cancer. Ann Surg Oncol. 2013;20(8):2683–92.
Fujimura T, Fushida S, Tsukada T, Kinoshita J, Oyama K, Miyashita T et al. A new stage of sentinel node navigation surgery in early gastric cancer. Gastric Cancer. 2015;18(2):210–7.
Kang KC, Cho GS, Han SU, Kim W, Kim HH, Kim MC et al. Comparison of Billroth I and Billroth II reconstructions after laparoscopy-assisted distal gastrectomy: a retrospective analysis of large-scale multicenter results from Korea. Surg Endosc. 2011;25(6):1953–61.
Inokuchi M, Kojima K, Yamada H, Kato K, Hayashi M, Motoyama K et al. Long-term outcomes of Roux-en-Y and Billroth-I reconstruction after laparoscopic distal gastrectomy. Gastric Cancer. 2013;16(1):67–73.
Taylor PR, Mason RC, Filipe MI, Vaja S, Hanley DC, Murphy GM et al. Gastric carcinogenesis in the rat induced by duodenogastric reflux without carcinogens: morphology, mucin histochemistry, polyamine metabolism, and labelling index. Gut. 1991;32(12):1447–54.
Goldstein SR, Yang GY, Curtis SK, Reuhl KR, Liu BC, Mirvish SS et al. Development of esophageal metaplasia and adenocarcinoma in a rat surgical model without the use of a carcinogen. Carcinogenesis. 1997;18(11):2265–70.
Fein M, Peters JH, Chandrasoma P, Ireland AP, Oberg S, Ritter MP et al. Duodenoesophageal reflux induces esophageal adenocarcinoma without exogenous carcinogen. J Gastrointest Surg. 1998;2(3):260–8.
Osugi H, Fukuhara K, Takada N, Takemura M, Kinoshita H. Reconstructive procedure after distal gastrectomy to prevent remnant gastritis. Hepatogastroenterology. 2004;51(58):1215–8.
Sato T, Miwa K, Sahara H, Segawa M, Hattori T. The sequential model of Barrett’s esophagus and adenocarcinoma induced by duodeno-esophageal reflux without exogenous carcinogens. Anticancer Res. 2002;22(1A):39–44.
Kauer WK, Peters JH, DeMeester TR, Feussner H, Ireland AP, Stein HJ et al. Composition and concentration of bile acid reflux into the esophagus of patients with gastroesophageal reflux disease. Surgery. 1997;122(5):874–81.
Kojima K, Yamada H, Inokuchi M, Kawano T, Sugihara K. A comparison of Roux-en-Y and Billroth-I reconstruction after laparoscopy-assisted distal gastrectomy. Ann Surg. 2008;247(6):962–7.
JGC Association. Japanese gastric cancer treatment guidelines 2010 (ver. 3). Gastric Cancer. 2011;2(14):113–23.
Jeong O, Park YK. Clinicopathological features and surgical treatment of gastric cancer in South Korea: the results of 2009 nationwide survey on surgically treated gastric cancer patients. J Gastric Cancer. 2011;11(2):69–77.
Fukuhara K, Osugi H, Takada N, Takemura M, Higashino M, Kinoshita H. Reconstructive procedure after distal gastrectomy for gastric cancer that best prevents duodenogastroesophageal reflux. World J Surg. 2002;26(12):1452–7.
Fukuhara K, Osugi H, Takada N, Takemura M, Ohmoto Y, Kinoshita H. Quantitative determinations of duodenogastric reflux, prevalence of Helicobacter pylori infection, and concentrations of interleukin-8. World J Surg. 2003;27(5):567–70.
Namikawa T, Kitagawa H, Okabayashi T, Sugimoto T, Kobayashi M, Hanazaki K. Roux-en-Y reconstruction is superior to billroth I reconstruction in reducing reflux esophagitis after distal gastrectomy: special relationship with the angle of his. World J Surg. 2010;34(5):1022–7.
Kumagai K, Shimizu K, Yokoyama N, Aida S, Arima S, Aikou T. Questionnaire survey regarding the current status and controversial issues concerning reconstruction after gastrectomy in Japan. Surg Today. 2012;42(5):411–8.
Vogel SB, Drane WE, Woodward ER. Clinical and radionuclide evaluation of bile diversion by Braun enteroenterostomy: prevention and treatment of alkaline reflux gastritis. An alternative to Roux-en-Y diversion. Ann Surg. 1994;219(5):458–65; discussion 65–6.
Kubo M, Sasako M, Gotoda T, Ono H, Fujishiro M, Saito D et al. Endoscopic evaluation of the remnant stomach after gastrectomy: proposal for a new classification. Gastric Cancer. 2002;5(2):83–9.
Lee MS, Ahn SH, Lee JH, Park do J, Lee HJ, Kim HH et al. What is the best reconstruction method after distal gastrectomy for gastric cancer? Surg Endosc. 2012;26(6):1539–47.
Park JY, Kim YJ. Uncut Roux-en-Y reconstruction after laparoscopic distal gastrectomy can be a favorable method in terms of gastritis, bile reflux, and gastric residue. J Gastric Cancer. 2014;14(4):229–37.
Morrison P, Miedema BW, Kohler L, Kelly KA. Electrical dysrhythmias in the Roux jejunal limb: cause and treatment. The American Journal of Surgery. 1990;160(3):252–6.
Miedema BW, Kelly KA. The Roux stasis syndrome: treatment by pacing and prevention by use of an ‘uncut’ Roux limb. Arch Surg. 1992;127(3):295–300.
Xiong JJ, Altaf K, Javed MA, Nunes QM, Huang W, Mai G et al. Roux-en-Y versus Billroth I reconstruction after distal gastrectomy for gastric cancer: a meta-analysis. World J Gastroenterol. 2013;19(7):1124–34.
Acknowledgments
This work was supported by the 2-year Research Grant from Pusan National University Hospital.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
In Choi, C., Baek, D.H., Lee, S.H. et al. Comparison Between Billroth-II with Braun and Roux-en-Y Reconstruction After Laparoscopic Distal Gastrectomy. J Gastrointest Surg 20, 1083–1090 (2016). https://doi.org/10.1007/s11605-016-3138-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-016-3138-7