Abstract
Purpose
Total pancreatectomy (TP) eliminates the risk and morbidity of pancreatic leak after pancreaticoduodenectomy (PD). However, TP is a more extensive procedure with guaranteed endocrine and exocrine insufficiency. Previous studies conflict on the net benefit of TP.
Methodology
A comparison of patients undergoing non-emergent, curative-intent TP or PD for pancreatic neoplasia using the National Surgical Quality Improvement Project data from 2005–2011 was done. Main outcome measures were mortality and major and minor morbidities.
Results
Of the 6,314 (97 %) who underwent PD and the 198 (3 %) who underwent TP, malignancy was present in 84 % of patients. The two groups were comparable at baseline. Mortality was higher after TP (6.1 %) than DP (3.1 %), p = 0.02. Adjusting for differences on multivariable analysis, TP carried increased mortality (OR 2.64, 95 % CI 1.3–5.2, p = 0.005). TP was also associated with increased rates of major morbidity (38 vs. 30 %, p = 0.02) and blood transfusion (16 vs. 10 %, p = 0.01). Infectious and septic complications occurred equally in both groups.
Conclusion
The morbidity of a pancreatic fistula can be eliminated by TP. However, based on our findings, TP is associated with increased major morbidity and mortality. TP cannot be routinely recommended for to reduce perioperative morbidity when pancreaticoduodenectomy is an appropriate surgical option.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
For pancreatic adenocarcinoma, pancreaticoduodenectomy (PD) remains the most common surgery. While total pancreatectomy (TP) is an option, in the modern era, TP is usually only performed for the treatment of multifocal disease such as multifocal neuro-endocrine tumors and intraductal papillary neoplasm. Per a major surgical text, “Another indication for TP [for head carcinoma] is the presence of an atrophic, soft, friable pancreatic remnant that does not hold sutures.”1 The argument supposes that the elimination of high-risk pancreatic anastomoses significantly improves morbidity, out-weighing any short-term morbidity or long-term endocrinopathy resulting from TP.2,3 Theoretically, elimination of this anastomosis by performing TP could reduce perioperative morbidity. Previous studies comparing the perioperative outcomes of PD and TP have conflicting results.
Historically, disappointment with oncologic results of PD for carcinoma enticed surgeons to attempt more extensive operations. However, studies of TP for pancreatic neoplasms failed to demonstrate improved benefit over less aggressive resections. Indeed in 1995, Yeo and associates4 demonstrated similar median survival for patients who had TP compared to PD (10 vs. 16 months). Likewise, researchers at Memorial Sloan-Kettering Cancer Center have shown that patients with TP had a significantly lower overall survival compared with a contemporary cohort of patients who underwent PD and distal pancreatectomy.5 There is no proven oncologic benefit for routine TP over PD, though specific clinical situations may be exceptions.
Advocates of TP to reduce morbidity rely on studies showing that “perioperative …outcome are comparable to standard pancreatic resections.”1 Early attempts with TP were fraught with complications, including mortality rates up to 20 %.6 Modern reports demonstrating more acceptable morbidity and mortality combined with better insulin and pancreatic enzyme preparations have reinvigorated interest in TP. Two single-institution studies show similar perioperative outcomes for TP vs. PD.2,3 Several other studies have shown increased morbidity, mortality, or both.6–9 The conflicting evidence is confounded by the sample sizes, populations, and designs of these studies.
To compare perioperative morbidity between patients undergoing TP and PD, we examined nationwide, prospective, clinical data. We have attempted to improve on previous studies by limiting analysis to a relatively homogenous group of patients and employing a large outcomes database.
Materials and Methods
Data Source and Inclusion Criteria
Our observational study used the National Surgical Quality Improvement Project (NSQIP) data from 2005–2011. The NSQIP database is a surgical outcomes research tool of the American College of Surgeons (ACS). On randomly selected surgical patients, the ACS NSQIP collects pre-operative co-morbidities, intraoperative variables, and 30-day post-operative outcomes. Specially trained nurses record the data which are audited semi-annually. The database is de-identified, and therefore, this study was exempt from the Institutional Review Board process.
ACS NSQIP and the hospitals participating in the ACS NSQIP are the source of the data used herein; they have not verified and are not responsible for the statistical validity of the data analysis or the conclusions derived by the authors.
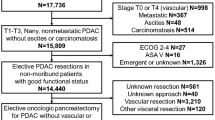
All patients who underwent PD or TP were identified by the Current Procedural Terminology codes 48150–48155. Patients with concurrent islet cell transplantation were not eligible. All NSQIP variables with procedure codes were examined, including primary, concurrent, and other procedure fields. We excluded cases that were emergent or where disseminated cancer was found. Only patients with a post-operative diagnosis of pancreatic neoplasms were included, international classification of diseases (ICD-9) codes: 221.5−211.9, 239, 239.5, 239.7, 239.8, 239.89, 235, 235.5, and 157–157.9. Patients with the post-operative diagnosis codes 157–157.9 carried the diagnosis of malignancy, while others were considered benign pancreatic neoplasia. Patients undergoing PD was compared those undergoing TP.
Data and Outcomes
We examined demographic data, diagnosis, and the following co-morbidities as potential confounders: (1) diabetes, (2) cardiac disease (hypertension, angina, previous myocardial infarction, cardiac angioplasty, or bypass surgery), (3) active pulmonary disease (pneumonia and severe chronic obstructive pulmonary disease (COPD)), (4) chemo- or radiotherapy within 30 days of surgery, and (5) weight loss.
The primary outcome was mortality at 30 days. Nurse reviewers conduct thorough follow-up to assess outcomes; follow-up is not limited by inpatient or outpatient status. The secondary outcome was morbidity, major and minor. We defined major morbidity as: re-intubation, prolonged intubation, pneumonia, cardiac arrest, myocardial infarction, renal failure requiring dialysis, deep or organ space infections, pulmonary embolus, the need for perioperative transfusion, sepsis, shock, stroke, or coma. We defined minor morbidity as urinary tract or superficial wound infections, deep vein thrombosis, or creatinine elevated >2 mg/dl above baseline without the need for dialysis.13
Statistical Analysis
Mean and median values were used to describe continuous data. Discrete variables were displayed as frequencies. For bivariable analyses, two-tailed t tests and Mann–Whitney U tests were used to compare continuous data, while Fisher’s exact or chi-squared tests were used for categorical variables.
For our multivariable regression, we derived separate logistic regression models for the effect of TP on mortality and overall, major, and minor morbidities. A priori, age and ASA class >2 were included as co-variables due to the known association with morbidity and mortality. Additional co-variables were chosen using stepwise selection to include variables with a p < 0.1. This model was checked against a model comprised of variables with (1) perceived importance or (2) association to the outcome on simple logistic regression. The models were compared using the likelihood ratio test and the Akaike information criteria. In each instance, the stepwise selection of variables returned a superior and more efficient regression model. The Hosmer–Lemeshow test was used to check goodness-of-fit. To check our models, bi-variable analysis was used to assess pre-operative variables as possible confounders. Neither diagnosis nor other pre-operative variables, other than those already included in the regression models, were associated with the outcomes. Statistical analyses were performed using Stata version 12 (College Station, TX).
Results
Population Characteristics
Of the 10,633 eligible pancreatectomies, we excluded 106 (1 %) emergencies and 261 (2.5 %) patients with disseminated cancer. Another 3,754 (37 %) cases with diagnoses other than neoplasia were excluded. The final populations consisted of 6,512 patients with non-emergent, curative intent surgery for pancreatic neoplasia of which 6,314 (97 %) underwent PD and 198 (3 %) underwent TP. Overall the population was 51 % male with a median age of 66 years. Malignancy was the most common post-operative diagnosis, present in 84 % of patients.
Overall, nearly two thirds of patients were independently functional before surgery. While 25 % of patients had no co-morbidities, 55 % had one or two medical conditions. The two groups were well matched in pre-operative characteristics. Active smoking was more common in the TP compared to the PD group (27 vs. 20 %, p = 0.02). Pre-existing diabetes was more common before TP (35 %) compared to PD (22 %), p < 0.001 (Table 1). The operative time was similar between groups.
Outcomes
The primary outcome, mortality, occurred in 3 % of patients. Mortality was significantly higher (6.1 vs. 3.1 %, p < 0.02) after TP (Table 2). In this population, we found preoperative alcohol or steroid use, COPD, hypertension, coronary artery disease, peripheral vascular disease, diabetes, ASA class >2, and perioperative transfusion of ≥2 RBCs predictive of mortality on regression. Even after adjusting for these co-variables on multivariable analysis, TP was associated with an odds ratio (OR) of 2.64 for mortality, p = 0.005 when compared to PD alone (Table 3).
Overall morbidity was similar for the TP and PD groups (44 vs. 39 %, respectively, p = 0.09). The rate of major morbidity was significantly higher in the TP group (38 vs. 30 %, p = 0.01) (Table 2). Except for preoperative alcohol use, the same co-variables mentioned above for mortality were statistically associated with major morbidity and included in the multivariable regression. In this adjusted model, TP was still associated with an increased odds of major morbidity (OR 1.52, p = 0.01) (Table 3).
Analyzed individually, the rates of specific morbidities were not significantly different after TP, compared to PD (Table 2). Surgical site infections, including organ-space infections or intra-abdominal abscesses, were not statistically higher after PD than TP (19 vs. 15 %, p = 0.1). More patients required transfusion of at least 2 units of red cells after TP, compared to PD (16 vs. 10 %, p = 0.01). Slightly higher rates of shock after TP, though not statistically different, might explain the increase in transfusion requirements. The hospital length of stay was 10 days and similar between groups.
Discussion
The most common operation for pancreatic neoplasia is pancreaticoduodenectomy. The Achilles heel of this operation is the pancreatic anastomosis, with an incidence of pancreatic leak ranging from 2–29 %.10 Patients’ risk of post-operative pancreatic fistula (POPF) can be estimated using a combination of pre- and intra-operative factors.11 In this study, 88 % of the high risk group had clinically relevant POPF. No universally accepted intervention has reduced the risk of POPF, despite countless maneuvers and attempts. Some pancreatic surgeons advocate total pancreatectomy in high risk cases to avoid the potential of POPF and subsequent morbidity. These surgeons usually cite studies demonstrating equivalent outcomes for TP and PD or partial pancreatectomy. However, there are similar studies showing worse outcomes for TP.
The current study is important because of the dataset used. NSQIP is a unique database, intended and validated for surgical outcomes research. Other databases, such as SEER, contain a depth of information on pathology, disease staging, and long-term outcomes, but are limited on patient comorbidities and perioperative outcomes. The prospective and multi-institutional data in NSQIP overcome some of the biases inherent in single-institution studies. While administrative data attempt to extract clinical information from claims data, NSQIP employs trained nurses to gather clinical data. Though every data source has its limitations, this NSQIP analysis should add a valuable perspective to the debate.
Our study suggests that total pancreatectomy is associated with double the rate of 30-day mortality, 6.1 vs. 3.1 %, compared to PD. These findings persisted in the multivariable regression despite adjusting for multiple patient factors and co-morbidities with a 2.5 higher odds of death. Table 4 contains a summary of other major series examining the outcomes of TP and PD. Our study data are consistent with other previously published series on TP and PD. Thirty-day mortality after TP ranged from 6–9 % for pancreatic tumors in published series.2,3,12,13 In these studies, mortality after elective PD ranged from 1.5–6.6 %. The interpretation of these findings have varied. Muller et al.2 matched 87 TP to 87 PD. Though their mortality rate was almost exactly that of our study, the difference was not significant. While this was interpreted as no different, the study was underpowered, with only a 25 % to power (based on the sample size) to show a difference despite a two-fold higher mortality in the TP group. This power calculation was performed using validated statistical software, PS Power and Sample Size Calculations (Version 3.0, January 2009).14 McPhee et al.12 showed a statistically higher mortality for TP compared to PD, and distal pancreatectomy. While Reddy et al.3 showed a higher mortality for TP overall p = 0.0007, the trend over time was decreased mortality for TP. The latest time period in their analysis showed similar mortality for TP vs. PD (1.9 vs. 1.2 %). However, this period of 2000–2007 included only 53 TP cases. The relatively small number of TPs may make the mortality estimate less reliable; the demonstrated mortality rate is not consistent with the nationwide study by McPhee et al., the other single-institution series by Muller et al., or our nationwide data. This may reflect characteristics specific to the institution. Nathan et al.13 acknowledged that their dataset did not include perioperative mortality and that they used two surrogate measures of death. The mortality rate of PD in this study was 6.3 %, much higher than other series. Our study demonstrates procedure-specific mortality rates consistent with other series with adequate numbers and power to obtain statistical difference.
The individual post-operative complications measured by NSQIP were not substantially different for TP compared to PD. This is likely due to the small number of events and lack of statistical power. The aggregation of severe complications into “major morbidity” improves the statistical power and did show worse outcomes for TP compared with PD.
Because individual complications occurred at similar rates between groups, we cannot explain the clinically and statistically significant difference in mortality based on any single outcome. . The difference in rate of red cell transfusion may have contributed to the increased mortality seen after TP. In other studies, the primary cause of mortality has been sepsis, often related to pancreatic leak and intra-abdominal infections.3 In this study, rates of infection were not statistically or clinically different. However, this was not the primary outcome and study only had a 35 % power to detect the observed 4 % difference in surgical infections. Since our dataset does not describe cause of death, this question cannot be answered. There is certainly the possibility that a morbidity not captured, such as severe hypoglycemia, resulted in the difference in mortality. However, traditional series do not describe high rates or severe episodes of post-operative glycemic-related morbidity. An alternative hypothesis is that a cascade of complications, not a single event which might be treatable, drives mortality. It is also possible that the study was underpowered to detect the morbidity leading to death.
Advances in surgical technique and perioperative care have improved patient outcomes. Similarly, newer studies on glycemic management and pancreatic enzyme supplementation after TP reveal good quality of life and lower than purported rates of endocrinopathy.15,16 This may allow surgeons to employ TP when the morbidity may previously have been prohibitive. This may also explain the resurrection of TP and increased discussion in the surgical literature. However, our study suggests that the more extensive procedure, TP, may be associated with higher mortality and major morbidity. There is a substantial body of literature that supports the common-sense idea that more extensive surgery carries higher morbidity, even in expert hands or high-volume centers. Our group has demonstrated a two-fold mortality rate and higher morbidity when resecting other abdominal viscera at the time of PD.17,18 Overall simultaneous colorectal and minor hepatic resections have similar perioperative outcomes compared to staged resections. However, colectomies with simultaneous major hepatectomies increase perioperative morbidity.19–22 These morbidities may be manageable with critical care support, advanced anti-microbials, parenteral nutrition, and radiologic procedures. However, the surgical insult and inflammatory response remain. The trend in our literature towards evaluating 90-day mortality may be prescient of our ability to abrogate morbidity or sustain patients who succumb later to the sequel of peri-operative complications. These data support existing evidence that more extensive surgery is associated with higher morbidity.
There are limitations in this study. There is no way to determine why TP was performed. PD may not have been a clinically or oncologically appropriate alternative in these cases. This, however, does not change our findings regarding the overall morbidity associated with the procedures. The current configuration of the NSQIP dataset does not provide assessment of surgery-specific outcomes. As such, we were not able to ascertain differences in outcomes of certain common post-PD complications such as pancreatic leak or delayed gastric emptying. While this might underestimate PD morbidity in this study, clinically significant pancreatic leaks generally manifest as organ-space infections (abscess) or sepsis; these outcomes would be reasonable proxies and are included in this study. Additionally, hyper- and hypoglycemia are not captured in NSQIP and so complications due to post-TP endocrinopathy are unmeasured. However, this does not change the conclusions of our study as inclusion of these outcomes would likely worsen outcomes, after TP.
Conclusions
The most common operation for neoplasia of the pancreatic head is pancreaticoduodenectomy. There is controversy regarding whether or not a total pancreatectomy may improve perioperative outcomes, in part, by avoiding a pancreatic leak and sepsis. In this large, prospective, clinical database, total pancreatectomy for neoplasia was associated with almost twice the mortality of PD and higher rates of major morbidity. This may arise from increased operative time, blood loss, the need for transfusion, or slightly higher rates of shock. While total pancreatectomy eliminates the possibility of a POPF, based on our findings, it cannot be routinely recommended for to reduce perioperative morbidity when pancreaticoduodenectomy is an appropriate surgical option.
References
Jarnagin WR, Belghiti J, Buchler MW, Chapman JR, D’Angelica M, DeMatteo RP. Blumbart’s surgery of the liver, biliary tract and pancreas. Vol 5th. New York, NY: W. B. Saunders, 2012.
Muller MW, Friess H, Kleeff J, Dahmen R, Wagner M, Hinz U, Breisch-Girbig D, Ceyhan GO, Büchler MW. Is there still a role for total pancreatectomy? Annals of Surgery. 2007;246(6):966–74; discussion 974–75.
Reddy S, Wolfgang CL, Cameron JL, Eckhauser F, Choti MA, Schulick RD, Edil BH, Pawlik TM. Total pancreatectomy for pancreatic adenocarcinoma: evaluation of morbidity and long-term survival. Annals of Surgery. 2009;250(2):282–87.
Yeo CJ, Cameron JL, Lillemoe KD, Sitzmann JV, Hruban RH, Goodman SN, Dooley WC, Coleman J, Pitt HA. Pancreaticoduodenectomy for cancer of the head of the pancreas. 201 patients. Annals of Surgery. 1995;221(6):721–71; discussion 731–23.
Ross DE. Cancer of the pancreas; a plea for total pancreatectomy. American Journal of Surgery. 1954;87(1):20–33.
Schmidt CM, Glant J, Winter JM, Kennard J, Dixon J, Zhao Q, Howard TJ, Madura JA, Nakeeb A, Pitt HA, Cameron JL, Yeo CJ, Lillemoe KD. Total pancreatectomy (R0 resection) improves survival over subtotal pancreatectomy in isolated neck margin positive pancreatic adenocarcinoma. Surgery. 2007;142(4):572–78; discussion 578–80.
Sakata N, Egawa S, Motoi F, Mikami Y, Ishida M, Aoki T, Ottomo S, Fukuyama S, Rikiyama T, Katayose Y, Goto M, Unno M. Institutional indications for islet transplantation after total pancreatectomy. Journal of Hepatobiliary Pancreatic Surgery. 2008;15(5):488–92.
Simons JP, Shah SA, Ng SC, Whalen GF, Tseng JF. National complication rates after pancreatectomy: beyond mere mortality. Journal of Gastrointestinal Surgery: Official Journal of the Society for Surgery of the Alimentary Tract. 2009;13(10):1798–1805.
Kneuertz PJ, Pitt HA, Bilimoria KY, Smiley JP, Cohen ME, Ko CY, Pawlik TM. Risk of morbidity and mortality following hepato-pancreato-biliary surgery. Journal of Gastrointestinal Surgery: Official Journal of the Society for Surgery of the Alimentary Tract. 2012;16(9):1727–1735.
Bassi C, Dervenis C, Butturini G, Fingerhut A, Yeo C, Izbicki J, Neoptolemos J, Sarr M, Traverso W, Buchler M; International Study Group on Pancreatic Fistula Definition. Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery. 2005;138(1):8–13.
Callery MP, Pratt WB, Kent TS, Chaikof EL, Vollmer CM, Jr. A prospectively validated clinical risk score accurately predicts pancreatic fistula after pancreatoduodenectomy. Journal of the American College of Surgeons. 2013;216(1):1–14.
McPhee JT, Hill JS, Whalen GF, Zayaruzny M, Litwin DE, Sullivan ME, Anderson FA, Tseng JF. Perioperative mortality for pancreatectomy: a national perspective. Annals of Surgery. 2007;246(2):246–53.
Nathan H, Wolfgang CL, Edil BH, Choti MA, Herman JM, Schulick RD, Cameron JL, Pawlik TM. Peri-operative mortality and long-term survival after total pancreatectomy for pancreatic adenocarcinoma: a population-based perspective. Journal of Surgical Oncology. 2009;99(2):87–92.
Dupont WD, Plummer WD, Jr. Power and sample size calculations. A review and computer program. Controlled Clinical Trials. 1990;11(2):116–28.
Jethwa P, Sodergren M, Lala A, Webber J, Buckels JA, Bramhall SR, Mirza DF. Diabetic control after total pancreatectomy. Digestive and Liver Disease: Official Journal of the Italian Society of Gastroenterology and the Italian Association for the Study of the Liver. 2006;38(6):415–19.
Billings BJ, Christein JD, Harmsen WS, Harrington JR, Chari ST, Que FG, Farnell MB, Nagorney DM, Sarr MG. Quality-of-life after total pancreatectomy: is it really that bad on long-term follow-up? Journal of Gastrointestinal Surgery: Official Journal of the Society for Surgery of the Alimentary Tract. 2005;9(8):1059–66; discussion 1066–67.
Nikfarjam M, Sehmbey M, Kimchi ET, Gusani NJ, Shereef S, Avella DM, Staveley-O’Carroll KF. Additional organ resection combined with pancreaticoduodenectomy does not increase postoperative morbidity and mortality. Journal of Gastrointestinal Surgery: Official Journal of the Society for Surgery of the Alimentary Tract. 2009;13(5):915–21.
Bhayani NH, Kimchi ET, Kaifi JT, Staveley-O’Carroll KF, Gusani NJ. Pancreaticoduodenectomy—multivisceral aresection adds morbidity: a NSQIP analysis. Surgery. 2013; in review.
Hamed OH, Bhayani NH, Ortenzi G, Kaifi JT, Kimchi ET, Staveley-O’Carroll KF, Gusani NJ. Simultaneous colorectal and hepatic procedures for colorectal cancer result in increased morbidity but equivalent mortality compared with colorectal or hepatic procedures alone: outcomes from the National Surgical Quality Improvement Program. HPB: The Official Journal of the International Hepato Pancreato Biliary Association. 2013;15(9):695–702.
de Santibanes E, Fernandez D, Vaccaro C, Quintana GO, Bonadeo F, Pekolj J, Bonofiglio C, Molmenti E. Short-term and long-term outcomes after simultaneous resection of colorectal malignancies and synchronous liver metastases. World Journal of Surgery. 2010;34(9):2133–40.
Capussotti L, Ferrero A, Vigano L, Ribero D, Lo Tesoriere R, Polastri R. Major liver resections synchronous with colorectal surgery. Annals of Surgical Oncology. 2007;14(1):195–201.
Reddy SK, Pawlik TM, Zorzi D, Gleisner AL, Ribero D, Assumpcao L, Barbas AS, Abdalla EK, Choti MA, Vauthey JN, Ludwig KA, Mantyh CR, Morse MA, Clary BM. Simultaneous resections of colorectal cancer and synchronous liver metastases: a multi-institutional analysis. Annals of Surgical Oncology. 2007;14(12):3481–91.
Author information
Authors and Affiliations
Corresponding author
Additional information
The paper was an oral presentation at the 2013 American Hepato-Pancreato-Biliary Association.
Rights and permissions
About this article
Cite this article
Bhayani, N.H., Miller, J.L., Ortenzi, G. et al. Perioperative Outcomes of Pancreaticoduodenectomy Compared to Total Pancreatectomy for Neoplasia. J Gastrointest Surg 18, 549–554 (2014). https://doi.org/10.1007/s11605-013-2393-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-013-2393-0