Abstract
Background/Aims
Serum α-fetoprotein (AFP) and total tumor volume (TTV) are important factors linked with post-operative tumor recurrence in hepatocellular carcinoma (HCC) patients. We investigated the role of a new prognostic marker, AFP-to-TTV ratio, in predicting HCC recurrence.
Methods
A total of 655 HCC patients undergoing resection were analyzed.
Results
In the multivariate logistic model, serum AFP level [odds ratio (OR) 32.459, p = 0.012] and TTV (OR 0.006, p = 0.01) were independently associated with a higher AFT/TTV ratio. The 1-, 3-, and 5-year tumor recurrence rates were 29 %, 55 %, and 68 %, respectively. In the Cox proportional hazards model, alcoholism (hazard ratio [HR], 1.354, p = 0.028), international normalized ratio of prothrombin time ≥1.01 (HR, 1.349, p < 0.001), multiple nodules (HR, 1.381, p = 0.004), main tumor diameter ≥4 cm (HR, 1.535, p = 0.001), macrovascular invasion (HR, 1.362, p = 0.016), and AFP/TTV ratio ≥1.5 (HR, 1.49, p < 0.001) were independently associated with tumor recurrence. In subgroup analysis, a higher AFP/TTV ratio was significantly associated with tumor recurrence in patients characterized by macrovascular invasion, TTV ≥ 40 cm3, or main tumor diameter ≥4cm (all p = 0.001).
Conclusion
The AFP/TTV ratio, a newly proposed marker for predicting post-operative tumor recurrence in HCC, is a feasible surrogate and may be useful in selecting super-high-risk patients for tumor recurrence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatocellular carcinoma (HCC) is a common malignancy accounting for 500,000 deaths per year.1 Surgical resection is considered as a curative treatment for HCC as well as the first treatment option for patients with preserved liver function, smaller tumor size, and no evidence of distant metastasis.2,3 For surgical patients, post-operative tumor recurrence is often seen, and approximately half of the patients who undergo partial hepatectomy will develop tumor recurrence within 3 years of surgery.4–6 Notably, tumor recurrence may adversely affect the survival of HCC patients and result in a devastating outcome. Early identification of patients who are at risk of tumor recurrence and providing intensive surveillance program for these patients may improve their survival.
Pre-operative serum α-fetoprotein (AFP) concentration has been shown as an important predictor of tumor recurrence and long-term survival in HCC patients.4,5,7–10 In addition, some cancer staging systems, such as the Cancer of the Liver Italian Program11 and Taipei integrated scoring system,12 showed that incorporation of serum AFP level into the prognostic model may provide additional prognostic benefit in outcome prediction. On the other hand, abundant studies comprehensively reviewed the role of tumor burden in predicting tumor recurrence.4–8,13,14 A recently reported marker, the calculated total tumor volume (TTV), may more accurately describe tumor burden as opposed to the current standard of number and size of tumor nodules of HCC in terms of outcome prediction.12,15,16
Serum AFP level highly correlates with the extent of tumor burden in HCC, and both factors play a crucial role in predicting tumor recurrence after curative treatment. However, the contribution and interaction of these two factors in association with post-operative tumor recurrence is not clear. Very few studies to date have specifically evaluated the influence of serum AFP level in relation to tumor burden on cancer recurrence. Moreover, whether higher serum AFP levels in patients with the same tumor burden pose additional risk in tumor recurrence is unknown. In an attempt to investigate the role of serum AFP concentration after abolishing the confounding effect of tumor burden on tumor recurrence, in this study, we have analyzed serum AFP level per TTV, or AFP-to-TTV ratio, in HCC patients undergoing surgical resection.
Patients and Methods
Patients
Between 2002 and 2012, patients who were newly diagnosed as HCC and received surgical resection as the primary treatment in Taipei Veterans General Hospital, a hospital providing primary to tertiary medical care to the residents of northern Taiwan, were prospectively identified and retrospectively analyzed in this study. The baseline information, including patient demographics, etiology of underlying liver disease, number and size of tumor(s), serum biochemistries, severity and complications of liver cirrhosis, and performance status, was collected at the time of diagnosis. This study conformed with the standards of the Declaration of Helsinki and current ethical guidelines.
Diagnosis and Definitions
We established the pre-operative diagnosis of HCC by histology or based on the findings of typical radiological features in at least two imaging examinations including ultrasound, contrast-enhanced dynamic computed tomography (CT), magnetic resonance imaging (MRI), and hepatic arterial angiography, or confirmed by a single positive imaging technique associated with serum AFP level >400 ng/mL.17,18 Ascites was diagnosed by abdominal sonography or computed tomography. The underlying etiology of HCC was related to hepatitis B virus (HBV) infection if serological detection of hepatitis B surface antigen (HBsAg) was positive (RIA kits, Abbott Laboratories, North Chicago, IL). Hepatitis C virus (HCV) infection was diagnosed if patients were seropositive for antibody against HCV (anti-HCV) by a second-generation enzyme immunoassay (Abbott Laboratories). Alcoholism was diagnosed in subjects with a documented history of alcohol excess of at least 40 g alcohol daily for 5 years or more.19 Performance status was assessed by using the Eastern Cooperative Oncology Group (ECOG) performance scale: 0 (asymptomatic) to 4 (confined to bed). The Child–Turcotte–Pugh (CTP) classification was evaluated based on serum levels of albumin and bilirubin, prothrombin time prolongation, and the severity of ascites and encephalopathy. The MELD score was calculated by using the equation: \( 9.57\times {\log_e}\left[ {\mathrm{creatinine}\left( {{{\mathrm{mg}} \left/ {\mathrm{dL}} \right.}} \right)} \right]+3.78\times {\log_e}\left[ {\mathrm{bilirubin}\left( {{{\mathrm{mg}} \left/ {\mathrm{dL}} \right.}} \right)} \right]+11.2\times {\log_e}\left[ {\mathrm{international}\,\mathrm{normalized}\,\mathrm{ratio}\left( {\mathrm{INR}} \right)} \right]+6.43 \),20 where the minimal values were set to 1.0 for calculation purposes. The value of creatinine was set to 4.0 mg/dL for calculation for patients with serum creatinine level more than 4.0 mg/dL. Macrovascular invasion was defined by the presence of adjacent thrombus to the tumor in portal vein with blurring boundary confirmed by at least two imaging modalities.21 All patients were treatment-naïve, and none had received specific anti-cancer treatment at the time of diagnosis.
Treatment Indication
Patients with HCC selected for surgical resection were (1) patients with tumor involving no more than three Healey’s segments, (2) patients with preserved liver function and had less than 25 % retention of indocyanine green 15 min after injection, and (3) patients who had no main portal vein trunk involvement or distant metastasis. The tumors were completely removed macroscopically in surgical procedure and were sent for gross and microscopic examinations. The number of tumor nodules was documented by histology, and the measurements of the tumor size were based on the largest dimension of the resected specimen. All patients had a surgical margin free of tumor.
Serum AFP Level, TTV, and AFP-to-TTV Ratio (AFP/TTV)
The number and size of tumor nodules were recorded after surgical resection. TTV was calculated as the sum of all tumor nodule volume, and each tumor nodule volume is calculated as \( {4 \left/ {3} \right.}\times 3.14\times {{\left( {\mathrm{maximum}\,\mathrm{radius}\,\mathrm{of}\,\mathrm{the}\,\mathrm{tumor}\,\mathrm{nodule}\,\mathrm{in}\,\mathrm{cm}} \right)}^3} \) as previously described.12 The AFP/TTV ratio was defined as pre-operative serum AFP concentration per unit TTV and was calculated in each study patient.
Tumor Recurrence
The patients received regular follow-up every 3 months after resection until tumor recurrence or dropout from the follow-up program. Post-operative follow-up program included ultrasound scan and measurement of serum AFP every 3 months and contrast-enhanced dynamic CT every 6 months or when necessary. MRI and/or hepatic angiography were done as supplemental imaging studies. Intrahepatic tumor recurrence is considered when a new hepatic lesion was detected by ultrasound and confirmed by contrast enhancement in arterial phase and wash out in venous phase in dynamic CT scan or MRI, or showing high tumor vascularity (“tumor stain”) in hepatic angiography and with progressive elevation of serum AFP levels. Extrahepatic tumor recurrence was defined by development of new lesions not seen in previous image studies. Once tumor recurrence was confirmed, hospitalization for further diagnosis and treatment was arranged.
Statistical Methods
The Chi-squared test or Fisher’s exact test (two-tailed) was used to compare categorical data. The Mann–Whitney U test and Kruskal–Wallis test was used to compare continuous variables between groups of two and of three or more. The possible risk factors associated with a higher AFP/TTV ratio were investigated by using the logistic regression analysis. Factors which were significant (p < 0.05) in the univariate analysis were introduced into multivariate logistic model to determine the adjusted odds ratio (OR). The Kaplan–Meier method with a log-rank test was applied to compare the survival distributions of different groups of patients. For continuous variables, the median of each variable was used as the cutoff to dichotimize patients in the survival analysis. Factors which were significant (p < 0.05) in the univariate analysis were introduced into multivariate Cox proportional hazards model to determine the adjusted hazard ratio (HR). For all comparisons, a p value less than 0.05 was considered statistically significant.
Results
Patient Characteristics
A total of 655 HCC patients who received surgical resection were identified and analyzed. All patients had histologically confirmed HCC. Of the study patients (Table 1), the mean age was 60 years, and 81 % of them were male. The most common etiology of chronic liver disease was chronic hepatitis B (60 %), followed by chronic hepatitis C (18 %), alcoholism (3 %), and dual virus infection (3 %). Most patients had single tumor nodule (72 %) and had a main tumor diameter more than 4 cm (55 %). The mean serum AFP level was 6,804 ng/mL, and the mean TTV was 375 cm3.
Distribution of Serum AFP Concentrations According to TTV
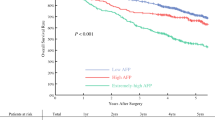
Figure 1a, b demonstrates the distribution of serum AFP level according to different TTV categories in all patients and in patients with AFP/TTV < 1.5 and ≥1.5, respectively. As TTV increased, there was a significant increase in serum AFP level in all study patients (p < 0.001). When the patients were stratified according to the AFP/TTV ratio, an increase in TTV was significantly associated with a higher serum AFP level in both groups of patients with AFP/TTV < 1.5 and ≥1.5 (both p < 0.001).
Box-plot of the distribution of serum AFP level according to TTV in all patients (a), and patients stratified by AFP/TTV (b). The median is identified by a line inside the box. The bottom and top of the box are the 25th and 75th percentile (the lower and upper quartiles, respectively), and the ends of the whiskers indicate the minimum and maximum. There was significant increase in serum AFP level with increasing TTV in all patients (p < 0.001) and in patients with AFP/TTV < 1.5 and ≥1.5 (both p < 0.001)
Factors Associated with Increased AFP/TTV Ratio
The comparison of the baseline demographics in patients with AFP/TTV < 1.5 and ≥1.5 is shown in Table 2. Patients characterized by a higher AFP/TTV ratio were younger (p = 0.001), of female gender (p < 0.001), with underlying viral hepatitis B or C (p = 0.003), and had lower serum BUN level (p < 0.001), lower serum creatinine level (p = 0.001), higher serum AFP level (p < 0.001), single tumor nodule (p < 0.001), and lower TTV (p < 0.001). In the multivariate logistic model, serum AFP level (OR 32.459, p = 0.012) and TTV (OR 0.006, p = 0.010) were the only two independent risk factors associated with a higher AFP/TTV ratio.
Factors Associated with Tumor Recurrence
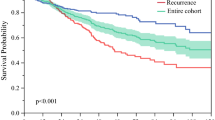
During the follow-up time of 26 ± 23 months, 363 (55 %) patients developed tumor recurrence. The 1-, 3-, and 5-year tumor recurrence rates were 29 %, 55 %, and 68 %, respectively. Factors that were possibly associated with post-operative tumor recurrence are shown in Table 3. Patients with alcoholism (p = 0.009), higher serum INR of prothrombin time (PT) (p = 0.007), multiple tumor nodules (p = 0.004), larger tumor diameter (p < 0.001), worse performance status (p = 0.017), macrovascular invasion (p < 0.001), and higher AFP/TTV ratio (p = 0.035, Fig. 2a) had a significantly increased risk of tumor recurrence in the univariate survival analysis.
The distribution of tumor recurrence-free survival in all patients (a), patients with macrovascular invasion (b), TTV ≥ 40 cm3 (c), and main tumor diameter ≥ 4cm (d). Patients with AFP/TTV < 1.5 had a significantly better recurrence-free survival than patients with AFP/TTV ≥ 1.5 in all patients (p = 0.035; a), and in subgroups of patients with macrovascular invasion, TTV ≥ 40 cm3, and main tumor diameter ≥ 4cm (all p = 0.001; b–d)
In the Cox multivariate model, independent factors associated with a higher risk of tumor recurrence included alcoholism (HR, 1.354, p = 0.028), serum INR of PT ≥ 1.01 (HR, 1.349, p = 0.005), multiple tumor nodules (HR, 1.381, p = 0.004), main tumor diameter ≥4cm (HR, 1.535, p = 0.001), macrovascular invasion (HR, 1.362, p = 0.016), and AFP/TTV ≥ 1.5 (HR, 1.49, p < 0.001).
Tumor Recurrence in Relation to the AFP/TTV Ratio in Different Subgroups of Patients
The mean and median AFP/TTV ratio in patients with and without macrovascular invasion and different groups of TTV, AFP level, size, and number of tumor are demonstrated in Table 4. There was a significant higher AFP/TTV ratio in patients with lower TTV (p < 0.001), higher serum AFP level (p < 0.001), smaller tumor size (p < 0.001), and no macrovascular invasion (p = 0.035).
Further analyses show that, among the subgroups of patients with macrovascular invasion, TTV ≥40 cm3, and main tumor size ≥4 cm, a higher AFP/TTV ratio was significantly associated with a higher tumor recurrence rate (all p = 0.001; Fig. 2b–d).
Discussion
Serum AFP level and tumor burden are well-established parameters linked with post-operative tumor recurrence in HCC patients.4,8,13 Serum AFP concentrations often increase concomitantly with the increase of tumor burden. In order to accurately delineate tumor behavior after adjusting the confounding effect of tumor burden and determine the interaction between these two factors, we systematically investigated the role of serum AFP level per unit TTV for the prediction of tumor recurrence in this study. Our results show that the newly proposed AFP/TTV ratio had unique clinical characteristics and independently correlated with post-operative tumor recurrence in HCC patients. A higher ratio may be a useful marker to identify patients at increased risk of tumor recurrence.
In this study, serum AFP level and TTV were exclusively the only two independent factors linked with a higher AFP/TTV ratio in the multivariate analysis, indicating that other factors, such as age, gender, the severity of liver cirrhosis, and performance status are only associated factors and not directly relevant to the increased AFP/TTV ratio. An interesting finding is that serum AFP concentration markedly increased with increasing TTV, implying that the tumor behavior may change with a larger tumor burden, and the magnitude of the change was more prominent in patients with AFP/TTV ratio ≥ 1.5 in comparison to those with a lower ratio (Fig. 1b). The interplay between serum AFP level and TTV may have important clinical implications and provide useful prognostic information.
In the survival analysis, patients with AFP/TTV ≥ 1.5 had 49 % increased risk of tumor recurrence in comparison to patients with AFP/TTV < 1.5. Consistent with previous studies,4,5 our results infer that AFP expression represents a distinct tumor behavior which is closely linked with disease progression. Nevertheless, this finding does not necessarily exclude the individual prognostic value of AFP and TTV for outcome prediction as reported from previous studies.4,5,22 Importantly, the finding that the AFP/TTV ratio may predict tumor recurrence was more apparent in patients known to have high risk of tumor recurrence, characterized by macrovascular invasion, TTV ≥ 40 cm3, and main tumor size ≥4 cm. Interestingly, our data indicate that, as TTV increases, serum AFP level increases logarithmically (Fig. 1a), resulting in an increase of serum AFP exceeding the extent of increase of TTV. Consequently, serum AFP level and AFP/TTV are both determinant factors in tumor recurrence in patients with large tumor burden. In a similar way, as supported by a previous study,23 the extent of the increase of serum AFP also exceeds the increase of tumor burden in the subgroup of patients with macrovascular invasion. In this regard, the AFP/TTV ratio may have additional value in predicting tumor recurrence especially in patients with larger tumor burden and macrovascular invasion.
The 1-, 3-, and 5-year tumor recurrence rates in our study were 29 %, 55 %, and 68 %, respectively. Previous studies provided comparable results exhibiting 25–32 %, 40–53 %, and 52–81 % recurrence rates at 1, 3, and 5 years of surgical resection, respectively.4,5,24–26 The findings that serum INR of PT, number of tumor nodules, main tumor diameter, and presence of macrovascular invasion were crucial predictors for tumor recurrence are consistent with most published literatures and suggest that the stage of liver fibrosis, tumor burden, and vascular invasion are important determinants.4,5,27–29 Another novel finding of our study is that alcoholism was also identified an influential factor associated with tumor recurrence. Supporting evidence showed that alcohol use in patients with viral hepatitis is associated with an accelerated progression of liver damage, earlier development of cirrhosis, and higher incidence of HCC formation.30–35 In our previous study, patients with viral hepatitis and concomitant alcoholism were found to develop HCC 3–4 years earlier than patients without alcohol consumption.36 Taken together, de novo carcinogenesis may be an explanatory mechanism for a higher tumor recurrence rate in patients with alcoholism.
This study has a few possible limitations. HBV is the most common etiology of HCC in Taiwan. This feature is different from most Western countries, where HCV infection and alcoholism are the predominant causes of chronic liver disease. It is our concern whether our results can be readily applied to patients in the Western countries. Another limitation is that the calculation of TTV was based on the assumption that all tumor nodules were spherical. Therefore, TTV could be slightly overestimated in patients with non-spherical tumor nodules.
In conclusion, the aggressiveness of tumor behavior in HCC, as indicated by the interaction between serum AFP level and TTV, is closely associated with tumor occurrence. The AFP/TTV ratio, a newly proposed marker for predicting post-operative tumor recurrence in HCC, is a feasible surrogate and may provide additional information in selecting super-high-risk patients for tumor recurrence. Intensive post-operative surveillance program and appropriate preventive measures, such as adjuvant chemotherapy or targeted therapy, should be considered in these patients and warrant further studies to clarify.
Abbreviations
- AFP:
-
α-Fetoprotein
- AFP/TTV:
-
AFP-to-TTV ratio
- Anti-HCV:
-
Antibody against HCV
- BUN:
-
Blood urea nitrogen
- CT:
-
Computed tomography
- CTP:
-
Child–Turcotte–Pugh
- ECOG:
-
Eastern Cooperative Oncology Group
- HBsAg:
-
Hepatitis B surface antigen
- HBV:
-
Hepatitis B virus
- HCC:
-
Hepatocellular carcinoma
- HCV:
-
Hepatitis C virus
- HR:
-
Hazard ratio
- INR:
-
International normalized ratio
- MELD:
-
Model for end-stage liver disease
- MRI:
-
Magnetic resonance imaging
- OR:
-
Odds ratio
- PT:
-
Prothrombin time
- RR:
-
Risk ratios
- TTV:
-
Total tumor volume
References
Bosch FX, Ribes J, Diaz M, Cleries R. Primary liver cancer: worldwide incidence and trends. Gastroenterology 2004;127:S5-S16.
Bruix J, Llovet JM. Prognostic prediction and treatment strategy in hepatocellular carcinoma. Hepatology 2002;35:519–524.
Cunningham SC, Tsai S, Marques HP, Mira P, Cameron A, Barroso E, et al. Management of early hepatocellular carcinoma in patients with well-compensated cirrhosis. Ann Surg Oncol 2009;16:1820–1831.
Imamura H, Matsuyama Y, Tanaka E, Ohkubo T, Hasegawa K, Miyagawa S, et al. Risk factors contributing to early and late phase intrahepatic recurrence of hepatocellular carcinoma after hepatectomy. J Hepatol 2003;38:200–207.
Wu JC, Huang YH, Chau GY, Su CW, Lai CR, Lee PC, et al. Risk factors for early and late recurrence in hepatitis B-related hepatocellular carcinoma. J Hepatol 2009;51:890–897.
Shah SA, Cleary SP, Wei AC, Yang I, Taylor BR, Hemming AW, et al. Recurrence after liver resection for hepatocellular carcinoma: risk factors, treatment, and outcomes. Surgery 2007;141:330–339.
Andreou A, Vauthey JN, Cherqui D, Zimmitti G, Ribero D, Truty MJ, et al. Improved long-term survival after major resection for hepatocellular carcinoma: a multicenter analysis based on a new definition of major hepatectomy. J Gastrointest Surg 2012 (in press).
Cucchetti A, Piscaglia F, Caturelli E, Benvegnu L, Vivarelli M, Ercolani G, et al. Comparison of recurrence of hepatocellular carcinoma after resection in patients with cirrhosis to its occurrence in a surveilled cirrhotic population. Ann Surg Oncol 2009;16:413–422.
Hakeem AR, Young RS, Marangoni G, Lodge JP, Prasad KR. Systematic review: the prognostic role of alpha-fetoprotein following liver transplant for hepatocellular carcinoma. Aliment Pharmacol Ther 2012. doi:10.1111/j.1365-2036.2012.05060.x.
Iwadou S, Nouso K, Kuwaki K, Kobayashi Y, Nakamura S, Tanaka H, et al. Time-dependent analysis of predisposing factors for the recurrence of hepatocellular carcinoma. Liver Int 2010;30:1027–1032.
Ueno S, Tanabe G, Sako K, Hiwaki T, Hokotate H, Fukukura Y, et al. Discrimination value of the new western prognostic system (CLIP score) for hepatocellular carcinoma in 662 Japanese patients. Cancer of the Liver Italian Program. Hepatology 2001;34:529–534.
Hsu CY, Huang YH, Hsia CY, Su CW, Lin HC, Loong CC, et al. A new prognostic model for hepatocellular carcinoma based on total tumor volume: the Taipei Integrated Scoring System. J Hepatol 2010;53:108–117.
Truty MJ, Vauthey JN. Surgical resection of high-risk hepatocellular carcinoma: patient selection, preoperative considerations, and operative technique. Ann Surg Oncol 2010;17:1219–1225.
Hosaka T, Ikeda K, Kobayashi M, Hirakawa M, Kawamura Y, Yatsuji H, et al. Predictive factors of advanced recurrence after curative resection of small hepatocellular carcinoma. Liver Int 2009;29:736–742.
Toso C, Trotter J, Wei A, Bigam DL, Shah S, Lancaster J, et al. Total tumor volume predicts risk of recurrence following liver transplantation in patients with hepatocellular carcinoma. Liver Transpl 2008;14:1107–1115.
Toso C, Asthana S, Bigam DL, Shapiro AM, Kneteman NM. Reassessing selection criteria prior to liver transplantation for hepatocellular carcinoma utilizing the Scientific Registry of Transplant Recipients database. Hepatology 2009;49:832–838.
Bruix J, Sherman M, Llovet JM, Beaugrand M, Lencioni R, Burroughs AK, et al. Clinical management of hepatocellular carcinoma. Conclusions of the Barcelona-2000 EASL conference. European Association for the Study of the Liver. J Hepatol 2001;35:421–430.
Bruix J, Sherman M. Management of hepatocellular carcinoma. Hepatology 2005;42:1208–1236.
Grant BF, Dufour MC, Harford TC. Epidemiology of alcoholic liver disease. Semin Liver Dis 1988;8:12–25.
Wiesner R, Edwards E, Freeman R, Harper A, Kim R, Kamath P, et al. Model for end-stage liver disease (MELD) and allocation of donor livers. Gastroenterology 2003;124:91–96.
Kikuchi LO, Paranagua-Vezozzo DC, Chagas AL, Mello ES, Alves VA, Farias AQ, et al. Nodules less than 20 mm and vascular invasion are predictors of survival in small hepatocellular carcinoma. J Clin Gastroenterol 2009;43:191–195.
Portolani N, Coniglio A, Ghidoni S, Giovanelli M, Benetti A, Tiberio GA, et al. Early and late recurrence after liver resection for hepatocellular carcinoma: prognostic and therapeutic implications. Ann Surg 2006;243:229–235.
McHugh PP, Gilbert J, Vera S, Koch A, Ranjan D, Gedaly R. Alpha-fetoprotein and tumour size are associated with microvascular invasion in explanted livers of patients undergoing transplantation with hepatocellular carcinoma. HPB (Oxford) 2010;12:56–61.
Lei HJ, Chau GY, Lui WY, Tsay SH, King KL, Loong CC, et al. Prognostic value and clinical relevance of the 6th Edition 2002 American Joint Committee on Cancer staging system in patients with resectable hepatocellular carcinoma. J Am Coll Surg 2006;203:426–435.
Huang YH, Wu JC, Lui WY, Chau GY, Tsay SH, Chiang JH, et al. Prospective case-controlled trial of adjuvant chemotherapy after resection of hepatocellular carcinoma. World J Surg 2000;24:551–555.
Huo TI, Wu JC, Lui WY, Lee PC, Huang YH, Chau GY, et al. Diabetes mellitus is a recurrence-independent risk factor in patients with hepatitis B virus-related hepatocellular carcinoma undergoing resection. Eur J Gastroenterol Hepatol. 2003;15:1203–1208.
Taura K, Ikai I, Hatano E, Yasuchika K, Nakajima A, Tada M, et al. Influence of coexisting cirrhosis on outcomes after partial hepatic resection for hepatocellular carcinoma fulfilling the Milan criteria: an analysis of 293 patients. Surgery. 2007;142:685–694.
Hwang S, Ahn CS, Kim KH, Moon DB, Ha TY, Song GW, et al. Super-selection of a subgroup of hepatocellular carcinoma patients at minimal risk of recurrence for liver transplantation. J Gastrointest Surg 2011;15:971–981.
Huo TI, Huang YH, Wu JC. Percutaneous ablation therapy for hepatocellular carcinoma: current practice and look into future. J Chin Med Asso 2005;68:155–159.
Gramenzi A, Caputo F, Biselli M, Kuria F, Loggi E, Andreone P, et al. Review article: alcoholic liver disease—pathophysiological aspects and risk factors. Aliment Pharmacol Ther 2006;24:1151–1161.
Hassan MM, Hwang LY, Hatten CJ, Swaim M, Li D, Abbruzzese JL, et al. Risk factors for hepatocellular carcinoma: synergism of alcohol with viral hepatitis and diabetes mellitus. Hepatology 2002;36:1206–1213.
Yuan JM, Govindarajan S, Arakawa K, Yu MC. Synergism of alcohol, diabetes, and viral hepatitis on the risk of hepatocellular carcinoma in blacks and whites in the U.S. Cancer 2004;101:1009–1017.
Ohnishi K, Iida S, Iwama S, Goto N, Nomura F, Takashi M, et al. The effect of chronic habitual alcohol intake on the development of liver cirrhosis and hepatocellular carcinoma: relation to hepatitis B surface antigen carriage. Cancer 1982;49:672–677.
Shih WL, Chang HC, Liaw YF, Lin SM, Lee SD, Chen PJ, et al. Influences of tobacco and alcohol use on hepatocellular carcinoma survival. Int J Cancer 2012;131:2612–2621.
Brandon-Warner E, Walling TL, Schrum LW, McKillop IH. Chronic ethanol feeding accelerates hepatocellular carcinoma progression in a sex-dependent manner in a mouse model of hepatocarcinogenesis. Alcohol Clin Exp Res 2012;36:641–653.
Lee YH, Hsu CY, Hsia CY, Huang YH, Su CW, Chiou YY, et al. Alcoholism worsens the survival of patients with hepatitis B virus and C virus-related hepatocellular carcinoma. Hepatol Int 2012 doi:10.1007/s12072-012-9375-2.
Acknowledgments
All authors declare no conflict of interest. This study was supported by grants from the Center of Excellence for Cancer Research at Taipei Veterans General Hospital (DOH101-TD-C-111-007), Taiwan, from Taipei Veterans General Hospital (V101C-170), Taipei, Taiwan, and from the Ministry of Education, Aim for the Top University Plan (101AC-D101), Taiwan.
Disclosures
There are no conflicts of interest that are relevant to the manuscript for all authors.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Lee, YH., Hsu, CY., Huang, YH. et al. α-Fetoprotein-to-Total Tumor Volume Ratio Predicts Post-operative Tumor Recurrence in Hepatocellular Carcinoma. J Gastrointest Surg 17, 730–738 (2013). https://doi.org/10.1007/s11605-012-2081-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-012-2081-5