Abstract
The surface tension of ionic liquids is studied according to phenomenological scaling and the law of corresponding states. The reduced coordinates γ*–T*, where γ* represents the reduced surface tension and T* is the reduced temperature, are introduced for the prediction of the surface tension from the melting point up to boiling point. It has been shown that the correlation can be expressed as a unique straight-line plot with a linear correlation coefficient of 0.984 that requires only the melting and boiling point parameters and can predict the surface tension accurately.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ionic liquids (ILs) were typically composed of large organic cations and small organic/inorganic anions and usually considered as a kind of special organic salt. In the past decade, they have received increasing attention due to their promising practice applications in chemistry and engineering such as reaction, separation, and catalyzation [1, 2]. Liquid surface tension as equilibrium thermodynamic property is also important in the design of industrial applications. Surface tension of a liquid is related to the intermolecular interaction potential energy and the liquid interfacial microstructure. Despite numerous research works, investigation of the thermophysical properties of ILs, their temperature dependences, and their correlations are still open fields of research [3–8].
Very few works have systematically studied the qualitative and/or quantitative relationships between the structures of ILs and their fundamental properties, such as melting point, viscosity, density, thermal and electrochemical stability, solvent properties, and surface tension [9–11]. To better understand the nature of ionic liquids and rationally expand their applications, knowledge of their physical properties is required. At present, however, data for many other physicochemical properties of ionic liquids are in short supply or too unreliable to allow similar structure–property relationship studies. Recently, Deetlefs et al. [10] attempted to predict surface tension by using the quantitative structure–property relationship (QSPR) correlations previously proposed by Knotts et al. [11]. They attempted with limited success due to the lack of experimental data. Then, Gardas et al. [12, 13] applied the QSPR correlation of Knotts et al. to predict surface tensions of assembled large database for ionic liquids. Ghatee et al. [3] correlated a linear relation between logarithm of surface tension and fluidity involving the characteristic exponent.
In this work, we propose simple regularities for surface tension of ILs, which has been applied for alkali metals [14] and alkali halides [15]. The reliability and utility of the modification proposed herein are established by performing detailed comparisons of the experimental values from the melting temperature up to the boiling temperature.
Corresponding states theory
The corresponding states theory (CST) for simple spherical fluids was first proposed by van der Waals [16] in an empirical form and later demonstrated by Pitzer [17]. Following its initial empirical formulation by van der Walls, the corresponding states principle has evolved in order to deal with several non-spherical and polar molecules. Its success is grounded on the accuracy obtained in broad temperature and pressure conditions and its strong theoretical basis [17–23], while mathematically simple and using a minimum amount of experimental information. One of its advantages is that the same formalism can be used for different properties whereas the only changes are the definition of the reduced property and the equation used for the reference system, or the reference system itself. Since these models do not use any experimental data of the estimating property of the target fluid, they are also predictive. The only experimental information required from the target fluid are the critical properties and the Pitzer acentric factor. The application of corresponding states models extends from equilibrium properties such as vapor pressure [24–28], liquid density [26, 28–30], or surface tension [31–36].
Correlation
Empirical relationships for the surface tension of ILs are shown to follow from the principle of corresponding states and are defined as [14, 15]:
where γ and γ m are the surface tension at temperature T and the melting temperature, T m, respectively, and T b is the boiling temperature. Dimensionless quantities were introduced by choosing the corresponding quantities at the melting temperature.
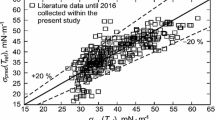
One distinct advantage of this set (γ m, T m) is that these are standard parameters that are readily available, as compared with critical parameters. Figure 1 shows the correlations suggested in this work. In Fig. 1, the solid line is the linear fit to the experimental data, which was obtained by linear regression as:
The linear correlation coefficient for Eq. (3) is 0.984. From Eq. (3), by using definitions (1) and (2) for the surface tension, γ, we can write:
Results and discussion
In Table 1, the absolute average percent deviation, AAD(%), was calculated in the range from the melting point to the boiling point.
The AAD(%), defined as,
where N represents the number of data points.
Reduced surface tension, as well as with the reduced temperature, can be expressed as a unique straight-line plot with a linear correlation coefficient of 0.984. In a database of 385 data points for 30 ILs, the mean percent deviation observed was only 2.98% (Table 1). We would like to illustrate the procedure by specific examples, [C5mim]NTf2, [C7mim]NTf2, [C12mim]NTf2, and [P6314]NTf2, that are not to be used in correlation. Table 2 shows the agreement with data reported from measurements. In Table 3, a comparison between the predicted surface tension values using Eq. (4) and the QSPR correlation [13] for the some ionic liquids is reported. It is apparent from Table 3 that our correlation results are in good overall agreement with the experimental data over the entire temperature range.
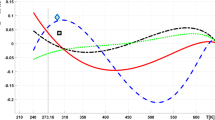
In the dimensionless representation γ*, there is a unique function for ionic liquids (Fig. 1). With dimensions, γ gives a curve for [C8mim]BF4, [C4mim]NTf2, [C4mim]Pf6, [C4mim]dca, resulting in good correlation with the experimental data (Fig. 2). The comparison of experimental values of surface tension at melting temperature γ m and calculated ones using Eq. (4) is shown in Fig. 3. A good agreement is obtained, indicating that expression provides a satisfactory description for the surface tension at melting temperature. In this work, we employ kT m as the characteristic energy, and no molecular parameters are involved. The proposed correlation has a simple form for easy calculation, requiring only the melting and boiling point parameters, and can predict the surface tension up to the boiling temperature accurately.
Comparison of experimental and correlated surface tension data for [C8mim]BF4 (black circle), [C4mim]NTf2 (black triangle), [C4mim]Pf6 (black square), [C4mim]dca (black diamond). Dots experimental data, Solid line predicted by Eq. (4)
The central idea of this work is the existence of a universal relation between dimensionless parameters formed using the physical quantities of interest and the applicability of CST to the estimation of surface tension. Thus, it is clear from the above results that whether a “universal” relation exists between the reduced surface tension and temperature depends on the particular parameter set used.
Most existing correlations for the prediction of the thermodynamic properties of ordinary fluids [16–23, 37] use the critical point data. The critical properties of ionic liquids cannot be determined since many of these compounds start to decompose as the temperature approaches the normal boiling point [38]. Moreover, the T b as a scaling parameter has not been measured, but the correlating methods for T b are available and estimates with high accuracy as compared with T c. One distinct advantage of this set (γ m, T m) is that these are standard parameters and are readily available. Thus, it would be interesting to see if they do well. Moreover, the effectiveness of T m, and T b as scaling parameters has been observed for liquid alkali metals [14]. Thus, we may classify those sets that suggest a universal relation, as desirable from a CST viewpoint. This does not mean that the other parameter sets have to be rejected altogether but only that they are not available within the context of CST. Also, the correlation shows how the successful empirical regularities can be obtained from a simple equation. As far as our results show, the present correlation could be applicable to other ionic liquids, but lack of experimental data hinders us from testing it.
References
Cole-Hamilton DJ (2003) Homogeneous catalysis new approaches to catalyst separation, recovery and recycling. Science 299:1702–1706
Chu X, Hu Y, Li J, Liang Q, Liu Y, Zhang X, Peng X, Yue W (2008) Desulfurization of diesel fuel by extraction with [BF4]-based ionic lquid. Chin J Chem Eng 16:881–884
Ghatee MH, Zare M, Zolghadr AR, Moosavi F (2010) Temperature dependence of viscosity and relation with the surface tension of ionic liquids. Fluid Phase Equilib 291:188–194
Restolho J, Serro AP, Mata JL, Saramago B (2009) Viscosity and surface tension of 1-ethanol-3-methylimidazolium tetrafluoroborate and 1-methyl-3-octylimidazolium tetrafluoroborate over a wide temperature range. J Chem Eng Data 54:950–955
Verevkin SP (2008) Predicting enthalpy of vaporization of ionic liquids: a simple rule for a complex property. Angew Chem Int Ed 47:5071–5074
Huo Y, Xia S, Zhang Y, Ma P (2009) Group contribution method for predicting melting points of imidazolium and benzimidazolium ionic liquids. Ind Eng Chem Res 48:2212–2217
Benjamin I (1996) Chemical reactions and solvation at liquid interfaces: a microscopic perspective. Chem Rev 96:1449–1475
Kuzmenko I, Raraport H, Kjaer K, Als-Nielsen J, Weissbuch I, Lahav M, Leiserowitz L (2001) Design and characterization of crystalline thin film architectures at the air–liquid interface: simplicity to complexity. Chem Rev 101:1659–1696
Katritzky AR, Lobanov VR, Karelson M (1995) QSPR: the correlation and quantitative prediction of chemical and physical properties from structure. Chem Soc Rev 24:279–287
Deetlefs M, Seddon KR, Shara M (2006) Predicting physical properties of ionic liquids. Phys Chem Chem Phys 8:642–649
Knotts TA, Wilding WV, Oscarson JL, Rowley RL (2001) Use of the DIPPR database for development of QSPR correlations: surface tension. J Chem Eng Data 46:1007–1012
Gardas RL, Coutinho JAP (2008) Extension of the Ye and Shreeve group contribution method for density estimation of ionic liquids in a wide range of temperatures and pressures. Fluid Phase Equilib 263:26–32
Gardas RL, Coutinho JAP (2008) Applying a QSPR correlation to the prediction of surface tensions of ionic liquids. Fluid Phase Equilib 265:57–65
Mousazadeh MH, Khanchi A, Ghanadi MM (2006) Corresponding states theory and thermodynamic properties of liquid alkali metals. JI CS 3:22–31
Ghatee MH, Mousazadeh MH, Boushehri A (1997) Corresponding states correlation for the surface tension of molten alkali halides. High Temp High Press 29:717–722
van der Waals JD (1894) Z Phys Chem 13:716–721
Pitzer KS (1939) Corresponding states for perfect liquids. J Chem Phys 7:583–590
Ely JF, Marrucho IM, Sengers JV, Kayser RF, Peters CJ, White HJ (2000) Equations of state for fluids and fluid mixture. Elsevier, Amsterdam
Guggenheim E (1945) The principle of corresponding states. J Chem Phys 13:253–261
Leland TW, Chappelear PS (1968) The corresponding states principle—a review of current theory and practice. Ind Eng Chem 60:15–43
Reed TM, Gubbins KE (1973) Applied statistical mechanics, chapter 11. McGraw-Hill, NewYork
Rowlinson JS, Swinton FL (1981) Liquids and liquid mixtures. Butterworths, London
Teja AS, Sandler SI (1980) A Corresponding States equation for saturated liquid densities. II. Applications to the calculation of swelling factors of CO2—crude oil systems. AIChE J 7:341–345
Morgan DL, Kobayashi R (1994) Extension of Pitzer CSP models for vapor pressures and heats of vaporization to long-chain hydrocarbons. Fluid Phase Equilib 94:51–58
Pitzer KS, Lippmann DZ, Curl RF, Huggins CM, Petersen DE (1995) The volumetric and thermodynamic properties of fluids. II. Compressibility factor, vapor pressure and entropy of vaporization. J Am Chem Soc 77:3433–3440
Poling BE, Prausnitz JM, O’Connell JP (2000) The properties of gases and liquids, 5th edn. McGraw-Hill, New York
Xiang HW (2002) The new simple extended corresponding-states principle: vapor pressure and second virial coefficient. Chem Eng Sci 57:1439–1449
Teja AS, Sandler SI, Patel NC (1981) A generalization of the corresponding states principle using two nonspherical reference fluids. Chem Eng J 21:21–28
Munõz F, Reich R (1983) New parameters for the Lee–Kesler correlation improve liquid density predictions. Fluid Phase Equilib 13:171–178
Teja AS (1980) A Corresponding States equation for saturated liquid densities. I. Applications to LNG. AIChE J 26:337–341
Rice P, Teja AS (1982) J Coll Interf Sci 86:158
Zuo Y, Stenby EH (1997) Corresponding-states and parachor models for the calculation of interfacial tensions. Can J Chem Eng 75:1130–1137
Queimada AJ, Silva FAE, Caço AI, Marrucho IM, Coutinho JAP (2003) Measurement and modeling of surface tensions of asymmetric systems: heptane, eicosane, docosane, tetracosane and their mixtures. Fluid Phase Equilib 214:211–221
Queimada AJ, Marrucho IM, Coutinho JAP (2001) Surface tension of pure heavy n-alkanes: a corresponding states approach. Fluid Phase Equilib 183–184:229–238
Queimada AJ, Caço AI, Marrucho IM, Coutinho JAP (2005) Surface tension of decane binary and ternary mixtures with eicosane, docosane, and tetracosane. J Chem Eng Data 50:1043–1046
Rolo LI, Caço AI, Queimada AJ, Marrucho IM, Coutinho JAP (2002) Surface tension of heptane, decane, hexadecane, eicosane, and some of their binary mixtures. J Chem Eng Data 47:1442–1445
Prakash SG, Ravi R, Chhabra RP (2004) Corresponding states theory and transport coefficients of liquid metals. Chem Phys 302:149–159
Valderrama JO, Rojas RE (2009) Critical properties of ionic liquids. Rev Ind Eng Chem Res 48:6890–6900
Kilaru P, Baker GA, Scovazzo P (2007) Density and surface tension measurements of imidazolium-, quaternary phosphonium-, and ammonium-based room-temperature ionic liquids: data and correlations. J Chem Eng Data 52:2306–2314
Wandschneider A, Lehmann JK, Heintz A (2008) Surface tension and density of pure ionic liquids and some binary mixtures with 1-propanol and 1-butanol. J Chem Eng Data 53:596–599
Sanchez LG, Espel JR, Onink F, Meindersma GW, De Haan AB (2009) Density, viscosity, and surface tension of synthesis grade imidazolium, pyridinium, and pyrrolidinium based room temperature ionic liquids. J Chem Eng Data 54:2803–2812
Ghatee MH, Zolghadr AR (2008) Surface tension measurements of imidazolium-based ionic liquids at liquid vapor equilibrium. Fluid Phase Equilib 263(2):168–175
Ahosseini A, Sensenich B, Weatherley LR, Scurto AM (2010) Phase equilibrium, volumetric, and interfacial properties of the ionic liquid, 1-hexyl-3-methylimidazolium bis(trifluoromethylsulfonyl)amide and 1-octene. J Chem Eng Data 55(4):1611–1617
Klomfar J, Souckova M, Patek J (2010) Surface tension measurements with validated accuracy for four 1-alkyl-3-methylimidazolium based ionic liquids. J Chem Thermodyn 42(3):323–329
Klomfar J, Souckova M, Patek J (2009) Surface tension measurements for four 1-alkyl-3-methylimidazolium-based ionic liquids with hexafluorophosphate anion. J Chem Eng Data 54:1389–1394
Pereiro AB, Santamarta F, Tojo E, Rodriguez A, Tojo J (2006) Temperature dependence of physical properties of ionic liquid 1, 3-dimethylimidazolium methyl sulfate. J Chem Eng Data 51:952–954
Pereiroa AB, Legidob JL, Rodriguez A (2007) Physical properties of ionic liquids based on 1-alkyl-3-methylimidazolium cation and hexafluorophosphate as anion and temperature dependence. J Chem Thermodyn 39:1168–1175
Pereiro AB, Verdia P, Tojo E, Rodriguez A (2007) Physical properties of 1-butyl-3-methylimidazolium methyl sulfate as a function of temperature. J Chem Eng Data 52:377–380
Freire MG, Carvalho PJ, Fernandes AM, Marrucho IM, Queimada AJ, Coutinho JAP (2007) Surface tensions of imidazolium based ionic liquids: anion, cation, temperature and water effect. J Colloid Interface Sci 314:621–630
Carvalho P, Neves CMSS, Coutinho JAP (2010) Surface tensions of bis(trifluoromethylsulfonyl)imide anion-based ionic liquids. J Chem Eng Data 55:3807–3812
Qing-Shan L, Miao Y, Pei-Fang Y, Xiu-Mei L, Zhi-Cheng T, Urs W (2010) Density and surface tension of ionic liquids [Cnpy][NTf2] (n = 2, 4, 5). J Chem Eng Data 55(11):4928–4930
Tariq M, Serro AP, Mata JL, Saramago B, Esperanc JMSS, Lopes JNC, Rebelo LPN (2010) High-temperature surface tension and density measurements of 1-alkyl-3-methylimidazolium bistriflamide ionic liquids. Fluid Phase Equilib 294:131–138
Carvalho PJ, Freire MG, Marrucho IM, Queimada AJ, Coutinho JAP (2008) Surface tensions for the 1-alkyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide ionic liquids. J Chem Eng Data 53:1346–1350
Wachter P, Schweiger HG, Wudy F, Gores HJ (2008) Efficient determination of crystallisation and melting points at low cooling and heating rates with novel computer controlled equipment. J Chem Thermodyn 40:1542–1547
Wasserscheid P, van Hal R, Bosmann A (2002) 1-n-Butyl-3-methylimidazolium ([bmim]) octylsulfate an even greener ionic liquid. Green Chem 4:400–404
Fredlake CP, Crosthwaite JM, Hert DG, Aki SNVK, Brennecke JF (2004) Thermophysical properties of imidazolium-based ionic liquids. J Chem Eng Data 49:954–964
Ngo HL, LeCompte K, Hargens L, McEwen AB (2000) Thermal properties of imidazolium ionic liquids. Thermochim Acta 97(102):357–358
MacFarlane DR, Meakin P, Amini N, Forsyth M (2001) Structural studies of ambient temperature plastic crystal ion conductors. J Phys Condens Matter 13:8257–8267
Sun J, Forsyth M, MacFarlane DR (1998) Room-temperature molten salts based on the quaternary ammonium ion. J Phys Chem B 102:8858–8864
Domanska U, Laskowska M (2008) Phase equilibria and volumetric properties of (1-ethyl-3-methylimidazolium ethylsulfate + alcohol or water) binary systems. J Solution Chem 37(9):1271–1287
Shevelyova MP, Zaitsau DH, Paulechka YU, Blokhin AV, Kabo GJ (2007) Solid–liquid equilibrium and activity coefficients for caprolactam + 1-hexyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide and cyclohexanone oxime + 1-hexyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide. J Chem Eng Data 52:1360–1365
Shimizu Y, Ohte Y, Yamamura Y, Saito K (2007) Effects of thermal history on thermal anomaly in solid of ionic liquid compound [C4mim][Tf2N]. Chem Lett 36:1484–1485
Domanska U, Morawski P (2007) Influence of high pressure on solubility of ionic liquids: experimental data and correlation. Green Chem 9:361–368
Tokuda H, Tsuzuki S, Susan MABH, Hayamizu K, Watanabe M (2006) How ionic are room-temperature ionic liquids? An indicator of the physicochemical properties. J Phys Chem B 110(39):19593–19600
Torrecilla JS, Rodríguez F, Bravo JL, Rothenberg G, Seddon KR, López-Martin I (2008) Optimising an artificial neural network for predicting the melting point of ionic liquids. Phys Chem Chem Phys 10:5826–5831
Zhang SJ, Sun N, He XZ, Lu XM, Zhang XP (2006) Physical properties of ionic liquids: database and evaluation. J Phys Chem Ref Data 35:1475–1517
Huddleston JG, Visser AE, Reichert WM, Willauer HD, Broker GA, Rogers RD (2001) Characterization and comparison of hydrophilic and hydrophobic room temperature ionic liquids incorporating the imidazolium cation. Green Chem 3:156–164
Arce A, Earle MJ, Katdare SP, Rodriguez H, Seddon KR (2007) Phase equilibria of mixtures of mutually immiscible ionic liquids. Fluid Phase Equilib 261:427–433
Acknowledgements
We are indebted to the Department of Chemistry, Nuclear Science Research School, Nuclear Science & Technology Research Institute (NSTRI) for support of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mousazadeh, M.H., Faramarzi, E. Corresponding states theory for the prediction of surface tension of ionic liquids. Ionics 17, 217–222 (2011). https://doi.org/10.1007/s11581-011-0536-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-011-0536-4