Abstract
The effect of noise on the pattern selection in a regular network of Hodgkin–Huxley neurons is investigated, and the transition of pattern in the network is measured from subexcitable to excitable media. Extensive numerical results confirm that kinds of travelling wave such as spiral wave, circle wave and target wave could be developed and kept alive in the subexcitable network due to the noise. In the case of excitable media under noise, the developed spiral wave and target wave could coexist and new target-like wave is induced near to the border of media. The averaged membrane potentials over all neurons in the network are calculated to detect the periodicity of the time series and the generated traveling wave. Furthermore, the firing probabilities of neurons in networks are also calculated to analyze the collective behavior of networks.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Noise plays an important role in changing the dynamics of media. For example, noise can induce and enhance the synchronization of neurons in network and coupled oscillators (Neiman and Russell 2002; Zhou and Kurths 2002; He et al. 2003; Xu et al. 2007; Kiss et al. 2003; Qu et al. 2012; Shi et al. 2008). Particularly, various of ordered spatiotemporal patterns and coherence resonance could be induced in the network by an optimized noise (Garcia-Ojalvo et al. 1993; Buceta et al. 2003; Perc 2005; Tang et al. 2012; Liu et al. 2010; Li et al. 2009; Du et al. 2010). More interesting, distinct transition of patterns could be induced by the fluctuation of bifurcation parameters (Vanag and Epstein 2001; Brusch et al. 2004; Xie et al. 2006; Ouyang and Felesselles 1996; Zhou and Ouyang 2000). Spiral wave is a special kind of spatiotemporal pattern, which is proved to exist in neocortex (Huang et al. 2004; Wu et al. 2008; Huang et al. 2010) and cardiac tissue (Garfinkel et al. 2000; Bursac et al. 2004). (Neiman and Russell 2002; Huang et al. 2010) show that spiral wave can play an active role in communicating signals in neuron systems. On the other hand, the appearance and breakup of the spiral wave often indicate harmful things in biological system. Modern medical science shows that the instability of the spiral wave in the cardiac tissue can possibly cause death due to ventricular fibrillation (Davidenko et al. 1992; Jalife 2000). Therefore, it is very important to study the evolution of spatiotemporal patterns in biological systems. A subexcitable medium is defined as one, in which a wave cannot grow in a limited time and space, i.e. a subexcitable medium cannot support any wave propagations, otherwise, it is defined as the excitable one (Hildebrand et al. 1995; Jung et al. 1998). Previous works show that noise can initiate and sustain wave behavior under subexcitable medium (Sagues et al. 2007; Hempel et al. 1999; Kadar and Wang 1998; Wang et al. 1999; Jia et al. 2004; Alonso et al. 2002a, b; Hou and Xin 2002). In the reference Jia et al. (2004), it is shown that the boundary of subexcitable BZ reaction medium is excitable and can be seen as a wave sources, but no waves propagate inside medium in the absence of noise. However, in the presence of a noisy electrical field, several wave fronts emerge and propagate forward in the medium. References (Alonso et al. 2002a, b; Hou and Xin 2002) show that spatiotemporal noise can sustain spiral wave propagation in the subexcitable media. References (Lindner et al. 2004; Garca-Ojalvo and Schimansky-Geier 2000; Garca-Ojalvo et al. 2001; Sendiña-Nadal et al. 1998; Ullner et al. 2003) summarize and describe the influence of noise on the spatiotemporal structure exhibited in an excitable medium. For example, it was reported that noise can induce pulse propagation (Garca-Ojalvo and Schimansky-Geier 2000; Garca-Ojalvo et al. 2001), and the propagation speed of planar pulses can be enhanced by noise (Sendiña-Nadal et al. 1998). In the reference (Garcia-Ojalvo and Schimansky-Geier 1998), it is shown that noise can induce complex spiral wave dynamics.
In this paper, the effect of Gaussian white noise on spatiotemporal dynamics of a two-dimensional square lattice Hodgkin–Huxley (H–H) neuron model (Hodgkin and Huxley 1952) is investigated. The numerical simulation results show that the formation and transition of different patterns can be controlled by noise in subexcitable and excitable media, respectively. The line wave cannot travel inside the subexcitable network of neurons in the absence of noise. However, when a Gaussian white noise is introduced into all neurons, it is found that line waves can be changed into spiral wave or target wave and can sustain propagation in the networks of neurons. In excitable neuronal networks, it is found that a sustained propagating spiral wave and a target wave can be converted into each other in the presence of noise. The results of this work show the more phenomena to prove that noise could enhance excitability of system than previous works.
Model and numerical simulation
The famous H–H model is often used to describe the electric activity of a neuron, and the nearest-neighbor coupling H–H neural network in the presence of noise is described as:
Integer subscripts i and j denote the positions of neurons in the two-dimensional network. V ij is the membrane potential of the neuron at the site (i,j). The variables m, n, and h describe the gating parameters of neuronal ion channels. D is the coupling coefficient, which has the same physical unit as conductance. The dimensionless S is the noise intensity. The Gaussian white noise ξ(t) with the statistical correlation <ξ(t)> = 0, and <ξ(t)ξ(t′)> = δ(t–t′), which holds the same physical unit as current, φ(T) is the temperature factor and is given by \( \varphi (T) = 3^{{{{(T - 6.3^{^\circ C} )} \mathord{\left/ {\vphantom {{(T - 6.3^{^\circ C} )} {10^\circ C}}} \right. \kern-0pt} {10^\circ C}}}} \). I ij = I 0 + I 1 is the external current, in which I 0 is the constant current, I 1 is external sinusoidal stimulus, and I 1 = sin (2π × 0.0013t) in all numerical simulations. Other parameters are constants in simulations. The membrane capacitance is assumed to be C m = 1 μF/cm2. The maximal potassium conductance is g K = 36 mS/cm2, the maximal sodium conductance is g Na = 120 mS/cm2, the leakage current conductance is g L = 0.3 mS/cm2. The reversal potentials are V K = −77 mV, V Na = 50 mV, and V L = −54.4 mV. Numerical simulation is carried out in a 100 × 100 neuronal network, and the 4th-order Runge–Kutta method with a time step h = 0.02 and no-flux boundary condition are adopted. The initial values of all neurons are taken as V ij = −32, m ij = −0.5, h ij = −0.12, n ij = −0.1(i = 41, 42, 43; j = 1,2,…,50); V ij = −10, m ij = 0, h ij = 0, n ij = 0(i = 44, 45, 46; j = 1, 2, … ,50); V ij = −61, m ij = 0.1, h ij = 0.47, n ij = 0.37(i = 47, 48, 49; j = 1, 2, … ,50); and V ij = −65, m ij = 0.1, h ij = 0.45, n ij = 0.4 for the other neurons. These initial values are selected to make a wave seed such that a wave could be developed in time in an excitable network.
In order to study the collective behavior of the network, the mean membrane potential and the firing probability of the neurons in the network are described by (Ma et al. 2008; Li et al. 2012)
where N 2 is the number of neurons and m is the number of firing neurons at a certain time k. The neuron is firing if the membrane potential V ij > x th, where x th = −55 is threshold of the membrane potential of the neuron.
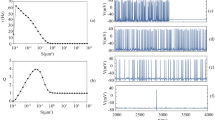
By selecting appropriate values of parameters D and I 0 and T, systems can become subexitable or excitable. The value of temperature is T = 6.0 °C in numerical simulations. The domains of the coupling coefficient D and the constant current I 0 for excitable and subexcitable systems are given in Fig. 1.
The effect of noise on patterns in the subexcitable neural network
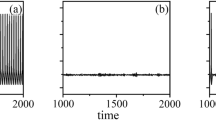
For coupling coefficient D = 0.25, and external current I 0 = 0.23, snapshots of the membrane potential of neurons are plotted in Fig. 2 (upper row). It is found that the selected initial values of variables make the line wave to form in the network, but the line wave fails to grow with time in the absence of noise (Fig. 2 upper row), which means that the system is subexcitable. If noise is imposed in all neurons, with noise intensity S = 0.5, it is found that the line wave evolves into a rotating spiral wave with time, as shown in Fig. 2 (bottom row). These results are in agreement with those reported in references (Alonso et al. 2002; Hou and Xin 2002).
If we change the parameters, such that the coupling coefficient D = 0.063 and the external current I 0 = 5, the line wave cannot propagate with time, as shown in Fig. 3 (the upper row), and the system is subexcitable in the absence of noise. By imposing noise with S = 0.19 on the networks of neurons, it is found that the circle waves appear and diffuse with time, as shown in Fig. 3 (the middle row). Increasing the intensity of noise to S = 0.20, the target wave is induced and propagates in the network, as shown in Fig. 3 (the third row).
The effect of noise on patterns in the excitable neural network
The same neural network can be excitable by changing the parameter values to I 0 = 4.0, D = 0.3. A stable rotating spiral wave can be induced and it can propagate with time in the network of neurons in the absence of noise. When noise is introduced into neurons in network with the noise intensity S = 0.6, it is found that the original spiral wave is replaced by the target wave, as seen in Fig. 4. These results indicate that different types of regular waves can be transformed into each other in excitable neural network by introduction of noise.
Furthermore, it is found that the wave source of initial wave in the original system without noise can be changed by noise.
If we choose D = 0.41 and I 0 = 5.0, the target wave can be generated and it propagates in the network in the absence of noise. The wave source is located near the centre of the network. The results are shown in Fig. 5a. By calculating the mean membrane potential of the network, it is found that the target wave could occupy the entire network after t > 100. The curves of the mean membrane potential and the firing probability are plotted in Fig. 5b, and it can be seen that the trends of two curves are almost identical.
By introducing noise with intensity S = 0.55 into the all neurons, it is observed that the U-shape wave appears first, and it diffuses outside with time till it encounters the boundary of the network of neurons, and then the system becomes nearly homogeneous. At this time, it is found that a new wave source appears at another site of the network, and new wave propagates with time, and the system becomes homogeneous after the wave encounters the boundary of the network of neurons at about t = 350, as shown in Fig. 6a. From the time series of the mean membrane potential of neurons in networks, as seen in Fig. 6b, it is found that the value of the mean membrane potential decreases from t = 50 to t = 70 and in this time domain the wave propagates in U-shape in network. Form t = 70 to t = 270, the value of the mean membrane potential increases with small fluctuation, which means that new wave appears and grows in the network. For t > 270, the value of the mean membrane potential begins to decrease. As the wave becomes sparse at about t = 350, and the value of the mean membrane potential is nearly stable at about −62, indicating that the system becomes homogeneous again. The curve of the firing probability for all neurons is plotted in Fig. 6b and it is found that the tendency of firing probability is coincident with the curve of the mean membrane potential.
Snapshots of the membrane potential of systems with D = 0.41, I 0 = 5.0,and S = 0.55, for t = 30 (a1), t = 50 (a2), t = 70 (a3), t = 80 (a4), t = 100 (a5), t = 130 (a6), t = 200 (a7), t = 300 (a8), t = 350 (a9) time units. b The curve of the mean membrane potential of all the neurons (solid); the curve of the firing probability of all neurons (dash)
If a noise intensity of S = 0.57 is imposed on all neurons in the network, another phenomenon is observed. First, the U-shape wave appears and diffuses outside with time, and the system becomes nearly homogeneous as the wave encounters the boundary of the network. This is also the case for S = 0.55. But the difference is that after becoming homogenous, two new wave sources appear at the same time, and new waves propagate outside until the system become homogenous again. The results are given in Fig. 7a. The curve of the mean membrane potential of neurons is plotted in Fig. 7b, and the trend of the curve is similar to the system for S = 0.55. The value of the mean membrane potential of neurons in network decreases when initial wave diffuses outside the system. When new wave forms and grows in the network, the value of mean membrane potential increases with small fluctuation. Lastly, the value of the mean membrane potential is nearly a stable value of about −62 at t = 350, and the system becomes homogeneous. The curve of the firing probability is plotted in Fig. 7b, and the trend of the firing probability is coincident with one of the mean membrane potential of neurons, too.
Snapshots of the membrane potential of systems with D = 0.41, I 0 = 5.0 and S = 0.57, for t = 30 (a1), t = 50 (a2) t = 70 (a3) t = 80 (a4) t = 100 (a5), t = 130 (a6), t = 150 (a7), t = 300 (a8), t = 350 (a9) time units. b The evolution of the mean membrane potential of all the neurons (solid); the curve of the firing probability of all neurons (dash)
Conclusions
In this work, the noise-induced formation and transition of varies patterns in subexcitable and excitable neuronal networks are investigated in a two-dimensional network described by the H–H model. The following novel phenomena are observed.
-
(1)
When appropriate initial and parameters values are selected, the line wave can develope, but cannot propagate in subexcitable neural networks in the absence of noise. By introducing suitable intensity of noise into all neurons, it is found not only a sustained propagating spiral wave can be generated, but also circle and target waves can be induced. Noise enhances excitability of a subexcitable system.
-
(2)
When systems are excitable, by selecting appropriate initial and parameters values, spiral and target waves can be formed with sustained propagation with time in the network of neurons in the absence of noise. After introducing appropriate intensity of noise into all neurons, it is found that the spiral wave and the target wave can be transformed into each other.
-
(3)
The most interesting phenomenon is that new wave sources can be formed in excitable system after imposing appropriate intensity of noise on all neurons in the network. Firstly, the sustained target wave is developed in the network of neurons in the absence of noise. Then, we impose noise on all neurons, and U-shape wave is produced and diffuses with time until the system becomes homogenous when U-shape wave encounters the boundary of the network. Lastly, the new wave sources appear at new sites in the network, and new waves propagate outside until the system become homogenous again. To analyze these phenomena, we calculate the mean membrane potential of all neurons in the network. It is found that the value of the mean membrane potential decreases with U-shape wave diffusing outside, then it increases with time when new wave forms and grows in the network. Lastly, the value of the mean membrane potential is nearly a stable value at a threshold time, and the system becomes homogeneous again. Furthermore, the curves of the firing probability are plotted. It is found that the trend of the firing probability is coincident with one of the mean membrane potential, which indicates that the results are reasonable and reliable.
The most important result is that the formation and transition of various patterns are shown in the same network of neurons by imposing appropriate strength of noise on the network. We think these interesting numerical results may have potential applications in practical system.
References
Alonso S, Sagués F, Sanchom JM (2002a) Excitability transitions and wave dynamics under spatiotemporal structured noise. Phys Rev E 65:066107
Alonso S, Sendiña-Nadal I, Pérez-Muñuzuri V, Sancho JM, Sagués F (2002) Regular wave propagation out of noise in chemical active media. Phys Rev Lett 87:078302
Brusch L, Nicola ME, Bar M (2004) Comment on antispiral waves in reaction-diffusion systems. Phys Rev Lett 92:89801
Buceta J, Ibanes M, et al (2003) Noise-driven mechanism for pattern formation. Phys Rev E 67:021113
Bursac N, Aguel F, Tung L (2004) Multiarm spirals in a two-dimensional cardiac substrate. In: Proceedings of the national academy of sciences of the United States of America, vol 101, no. 43, p 15530
Davidenko JM, Pertsov R, Salomonsz AV, Baxter W, Jalife J (1992) Stationary and drifting spiral waves of excitation in isolated cardiac muscle. Nature 355:349–351
Du Y, Lu QS, Wang RB (2010) Using interspike intervals to quantify noise effects on spike. Cogn Neurodyn 4:199–206
Garca-Ojalvo J, Schimansky-Geier L (2000) Excitable structures in stochastic bistable media. J Stat Phys 101:473–481
Garca-Ojalvo J, Sagues F, Sancho JM, Schimansky-Geier L (2001) Noise-enhanced excitability in bistable activator-inhibitor media. Phys Rev E 65:011105
Garcia-Ojalvo J, Schimansky-Geier L (1998) Noise-induced spiral dynamics in excitable media. Eur phys Lett 47:298
Garcia-Ojalvo J, Hernandez-Machado A, Sancho JM (1993) Effects of external noise on the Swift-Hohenberg equation. Phys Rev Lett 71:1542–1545
Garfinkel A, Kim YH, Voroshilovsky O, Qu Z, Kil JR, Lee MH, Karagueuzian HS, Weiss JN, Chen PS (2000) Preventing ventricular fibrillation by flattening cardiac restitution. In: Proceedings of the national academy of sciences of the United States of America, vol 97, no. 11, p 6061
He DH, Shi PL, Stone L (2003) Noise–induced synchronization in realistic models. Phys Rev E 67:027201
Hempel H, Schimansky-Geier L, Garcia-Ojalvo J (1999) Noise-sustained pulsating patterns and global oscillations in subexcitable media. Phys Rev Lett 82:3713–3716
Hildebrand M, Bar M et al (1995) Statistics of topological defects and spatiotemporal chaos in a reaction-diffusion system. Phys Rev Lett 75:1503–1506
Hodgkin AL, Huxley AF (1952) A quantitative description of membrane current and its application to conduction and excitation in nerve. J phys 117:500
Hou ZH, Xin HW (2002) Noise-sustained spiral waves: effect of spatial and physics review letter, temporal memory. Phys Rev Lett 89:280601
Huang X, Troy WC, Yang Q, Ma H, Laing CR, Schiff SJ, Wu JY (2004) Spiral waves in disinhibited mammalian neocortex. J Neurosci 24:9897–9902
Huang XY, Xu W, Liang J, Takagaki K, Gao X, Wu JY (2010) Spiral wave dynamics in neocortex. Neuron 68(5):978–990
Jalife J (2000) Ventricular fibrillation: mechanisms of initiation and maintenance. Ann Rev Physiol 62(1):25–50
Jia X, Liao HM, Zhou LQ, Ouyang Q (2004) Properties of wave propagations induced by temporal noise in a subexcitable medium. Phys D Nonlinear Phenom 199:194–200
Jung P, Cornell-Bell A, Moss F, Kadar S, Wang J, Showalter K (1998) Noise sustained waves in subexcitable media: from chemical waves to brain waves. Chaos 8(3):567–575
Kadar S, Wang J (1998) Noise-supported travelling waves in subexcitable media. Nature 391:770–772
Kiss IZ, Zhai Y, Hudson JL, Zhou C, Kurths J (2003) Noise enhanced phase synchronization and coherence resonance in sets of chaotic oscillators with weak global coupling. Chaos 13:267–278
Li YY, Zhang HM, Wei CL, Yang MH, Gu HG, Ren W (2009) Stochastic signal induced multiple spatial coherence resonances and spiral waves in excitable media. Chin Phys Lett 26(3):030504
Li YY, Jia B, Gu HG (2012) Multiple spatial coherence resonances induced by white gaussian noise in excitable network composed of Morris-Lecar model with class excitability. Acta Phys Sin 61(7):070504
Lindner B, Garça-Ojalvo J, Neiman A, Schimansky-Geier L (2004) Effects of noise in excitable systems. Phys Rep 392:321–424
Liu ZQ, Zhang HM, Li YY, Hua CC, Gu HG, Ren W (2010) Multiple spatial coherence resonance induced by stochastic signal in neuronal networks near a saddle-node bifurcation. Phys A 389:2642–2653
Ma J, Jia Y, Tang J, Yang LJ (2008) Breakup of spiral waves in the coupled Hindmarsh-Rose neurons Chin. Phys Lett 25:4325–4328
Neiman AB, Russell DF (2002) Synchronization of noise-induced bursts in noncoupled sensory neurons. Phys Rev Lett 88:138103
Ouyang Q, Felesselles JM (1996) Transition from spirals to defect turbulence driven by a convective instability. Nature 379:143–146
Perc M (2005) Spatial coherence resonance in excitable media. Phys Rev E 72:016207
Qu JY, Wang RB, Du Y, Cao JT (2012) Synchronization study in ring-like and grid-like neuronal network. Cogn Neurodyn 6:21–31
Sagues F, Sancho JM, Garca-Ojalvo J (2007) Spatiotemporal order out of noise. Rev Mod Phys 79:829–882
Sendiña-Nadal I, Muñuzuri AP, Vives D, Pérez-Muñuzuri V, Casademunt J, Ramírez-Piscina L, Sancho JM, Sagués F (1998) Wave propagation in a medium with disordered excitability. Phys Rev Lett 80:5437–5440
Shi X, Wang QY, Lu QS (2008) Firing synchronization and temporal order in noisy neuronal. Cogn Neurodyn 2:195–206
Tang Z, Li YY, Xi L, Jia B, Gu HG (2012) Spiral waves and multiple spatial coherence resonances induced by the colored noise in neuronal network. Commun Theor Phys 57(1):61–67
Ullner E, Zaikin A, Garca-Ojalvo J, Kurths J (2003) Noise-induced excitability in oscillatory media. Phys Rev Lett 91(18):180601
Vanag VK, Epstein IR (2001) Inwardly rotating spiral waves in a reaction-diffusion system. Science 294:835–837
Wang JC, Kádár S, Jung P, Showalter K (1999) Noise-driven avalanche behavior in subexcitable media. Phys Rev Lett 82:855–858
Wu JY, Huang XY, Zhang C (2008) Propagating waves of activity in the neocortex: what they are what they do. Neuroscientist 14(5):487–502
Xie FG, Xie DZ, et al (2006) Inwardly rotating spiral wave breakup in oscillatory reaction-diffusion media. Phys Rev E 74:026107
Wu Y, Xu JX, Jin WY, Hong L (2007) Detection of mechanism of noise-induced synchronization between two identical uncoupled neurons. Chin Phys Lett 24:3066–3069
Zhou C, Kurths J (2002) Noise-induced phase synchronization and synchronization transitions in chaotic oscillators. Phys Rev Lett 88:230602
Zhou LQ, Ouyang Q (2000) Experimental studies on long-wavelength instability and spiral breakup in a reaction-diffusion system. Phys Rev Lett 85:1650–1653
Acknowledgments
We thank Professor Huaguang Gu for useful discussions and helpful comments, and thank Professor Subhash Sinha for revising English writing. This work is supported by the National Natural Science Foundation of China (10972179 and 11172223), and New Faculty Research Foundation of XJTU.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, Y., Li, J., Liu, S. et al. Noise-induced spatiotemporal patterns in Hodgkin–Huxley neuronal network. Cogn Neurodyn 7, 431–440 (2013). https://doi.org/10.1007/s11571-013-9245-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11571-013-9245-1