Abstract
The use of multidetector CT (MDCT) represents a reality routinely used in several forensic institutes, for the numerous advantages that this diagnostic tool can provide; costs are becoming increasingly lower; data acquisition is always faster and once acquired may be revalued at any time. However, there are also some diagnostic limitations, for example, the visualization of the vascular system or a limited soft tissue contrast. In order to overcome these limitations, in recent years, contrast medium has been introduced in postmortem cases, with the development of several techniques of PMCT angiography (PMCTA) and standardized protocols to make them easily reproducible. The aim of this review is to highlight the advantages and pitfalls of PMCT and PMCTA in forensic investigation, taking into consideration the broad spectrum of applications both for natural and unnatural deaths and the numerous methods currently used. Secondly, in the light of the considerable progress in this field and the attempt to develop standardized protocols of PMCTA, the authors aim to evaluate the diagnostic value of PMCTA in comparison both to PMCT and conventional autopsy.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
With the discovery in 1895 of X-radiation by Dr. Röentgen of Würzburg (Germany), medical practitioners throughout the world were provided with an important new tool. It was also recognized as a potential means for medicolegal investigators [1]. Since it has been introduced, many improvements have been made in the field of radiological sciences, but only more recently have these been applied to forensic investigations, and the following of specific methods have been developed with the aim to support the forensic pathologist in establishing cause of death and in certain cases an attempt to substitute the conventional autopsy [2, 3]. In the field of Postmortem-Computed Tomography (PMCT), the first application reported in accredited literature dates back to 1983 in a case of a diving fatality, where it was possible to demonstrate the distribution of gas, by cerebral-computed tomography, in the head of a 20-year-old navy diver who died while diving at a depth of 43 m [4].
Currently, the use of Multidetector-Computed Tomography (MDCT) represents a reality routinely used in several forensic institutes, for the numerous advantages that this diagnostic tool can provide; costs are becoming increasingly lower; data acquisition is always faster and once acquired may be revalued at any time. However, there are also some diagnostic limitations, for example, the visualization of the vascular system or a limited soft tissue contrast. In order to overcome these limitations, in recent years, contrast medium has been introduced in postmortem cases, with the development of several techniques of PMCT angiography (PMCTA) [5–7] and standardized protocols to make them easily reproducible.
Presently, the most used PMCTA techniques are four, each of which has variants. One was developed by a research group of the University Center of Legal Medicine in Lausanne (Switzerland) [8]. They defined a protocol for high-quality multiphase PMCTA (MPMCTA) with the aim to obtain a better interpretation of the findings by completely filling the vascular system in order to decrease as much as possible the artifacts caused by perfusion. Postmortem circulation is established by using a pressure-controlled perfusion device (Virtangio®), which injects a contrast agent mixture composed of 6 % of Angiofil and paraffin oil into the femoral vessels which are cannulated on one side of the body. The MPMCTA protocol involves the performance of one native CT-scan followed by three angiographical phases. Variations of this technique have also been developed, for example, with the cannulation of the axillary artery and vein on one side of the body instead of the femoral artery and vein [9]. A different technique was developed even before the MPMCTA by Jackowski et al. and later modified by Ross et al. [10–12]. It differs in nearly all points from the MPMCTA. The only similarity is the cannulation of the femoral vessels and the fact that it is a whole body method. Otherwise, it uses a different perfusion devices (modified heart–lung machine, different perfusion protocol, in fact no standardized protocol exists), different liquids (use of contrast agents in watery solutions and different combinations of polyethylene glycol), and therefore, the interpretation of the images is very different. PMCTA using cardiopulmonary resuscitation (CPR) technique of chest compression immediately after death has also been evaluated [13]. This approach is preferred in Japan because it is less invasive and simpler than the cannulation of vessels. CPR is able to determine an increase of blood pressure thereby generating a slight cardiac output as a consequence of chest pressure. The application of CPR to PMCTA and the injection of the contrast media into peripheral veins is the same as in hospital procedure in vivo. Once the contrast agent has entered the heart cavities, it then goes into the body circulation thanks to CPR and images of the major arterial and venous system can be obtained [13, 14]. An alternative approach was applied in France in 2011 [15]. The femoral artery of a young man involved in a road accident was catheterized using ultrasound, and a pump was used for the injection of a mixture of iodinated contrast medium and water, which allowed the visualization of diverse principal arterial injuries.
The aim of this review is to highlight the advantages and pitfalls of PMCT and PMCTA in forensic investigation, taking into consideration the broad spectrum of applications both for natural and unnatural deaths and the numerous methods currently used. Secondly, in the light of the considerable progress in this field and the attempt to develop standardized protocols of PMCTA, the authors aim to evaluate the diagnostic value of PMCTA in comparison both to PMCT and conventional autopsy.
The databases
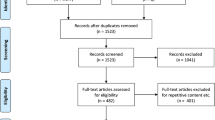
Some databases, from 1990 to December 2014, were searched: Medline, Cochrane Central, Scopus, Web of Science, ScienceDirect, EMBASE, and Google Scholar, using the following keywords: “virtopsy”, “virtual autopsy”, “minimally invasive autopsy”, “postmortem angiography”, “multiphase postmortem-computed tomography angiography (MPMCTA)”, “postmortem” with “imaging”, or “CT” or “CTA”. The 459 sources found after the initial screening in order to exclude duplicate sources and reviews, were examined according to the “inclusion criteria” by four independent physicians (two forensic pathologists and two forensic radiologists). Only papers selected by at least 3 physicians were included. The following inclusion criteria were used (one of the first two + at least other two):
-
-
Inclusion of case reports, series of cases, or retrospective studies
-
-
Research/original articles
-
-
Postmortem TC/TCA protocol used
-
-
Autopsy confirmation
-
-
Circumstantial data and/or clinical data
Seventy-one sources were identified. A comprehensive flow diagram is reported in Fig. 1.
All figures included in the present paper have extracted by authors from the archives of their Institutions, with authorization of the Ethical committee and consent of relatives of the deceased and here reported in anonymous form.
Discussion
Bleedings and hemorrhages
Acute bleedings and hemorrhages with a fatal outcome can be a tough challenge for forensic pathologists especially in identifying the source of bleeding. Moreover, forensic investigation must determine if the cause of bleeding is traumatic, an underlying medical condition or a combination of the two. [16–19]. PMCT (Fig. 2) and more recently PMCTA (Fig. 3) have provided a significant contribution for the detection of traumatic injuries (hematomas, lacerations, puncture wounds, crushing injuries, ballistic trauma etc.) or medical conditions in which the causes of bleeding not directly due to trauma (aneurysms, dissections etc.) [20–22].
Left-ventricle cardiac laceration due to gunshot. PMCTA. a No contrast phase: evidence of a large amount of left haemothorax associated with haemopericardium. The arterial phase confirms the extravasation of contrast material supported by rupture of the antero-lateral wall of the left ventricle. The ventricular lesion is also confirmed by MPR (arrows, b) and MIP reconstructions (c)
Palmiere et al. [23] have investigated the diagnostic value of MPMCTA (Virtangio®) in the detection and localization of the source of bleeding in 9 cases of acute hemorrhages. The results obtained show that in 8 cases, the MPMCTA identified the precise source of bleeding. The MPMCTA findings are summarized in Table 1. It is interesting to compare the MPMCTA results with the autopsy; only in three cases, the autopsy detected the exact source of bleeding (multiple laceration of the spleen, cerebellar arteriovenous malformation, and superior sagittal sinus) whereas for the remaining 5 cases, because smaller vessels were involved, it was not possible to identify the exact source of bleeding, and only hypotheses were formulated taking into consideration the presence of surrounding hematomas. The 9 cases reported by Palmiere et al. [23] concerned both bleedings due to traumatic causes (7 cases) and to natural causes (2 cases). Pomara et al. [24] reported a rare case of post-whiplash pseudoaneurysm of the right common carotid artery, which led to acute massive hemorrhage and the death of a 38-year-old man several days after the initial trauma. In this case, the application of PMCTA allowed the identification of the rupture of the pseudoaneurysm affecting the right common carotid artery with the contrast agent leaking into the mouth. The autopsy confirmed later the PMCTA findings and highlighted a large hemorrhagic clot extending to the right side of the neck and mediastinum.
A research group of Chiba University (Japan) [25] investigated the utility of postmortem cerebral angiography using multidetector row CT (MDCT) by injecting a contrast medium through a catheter inserted into the internal carotid and vertebral arteries of 10 subarachnoid hemorrhage cases. One of the most important aspects in the forensic investigation of subarachnoid hemorrhage is to determine whether the cause is intrinsic or extrinsic taking into consideration the legal consequences of the different causes of the subarachnoid hemorrhage [26, 27]. Although PMCTA has been shown to be able to detect aneurysms, however, it is sometimes difficult to identify the aneurysm and bleeding sites because of the large amount of contrast medium which can leak into the extravascular space. In order to overcome this limitation, the Japanese research group developed a “dynamic cerebral angiography” method, in which the same area is scanned several times during the injection of the contrast agent allowing real-time observation of the contrasted vasculature and capturing the exact moment when the contrast agent leaks from the hemorrhage site. This method although very useful in the detection of the exact source of bleeding shows some limitations: it is selective angiography for the cerebral arteries, not for the entire vascular system; therefore, systemic vascular conditions or pathologies with subsequent involvement of the cerebral vessels cannot be detected; secondly, this technique is unable to detect lesions at the proximal level of the internal carotid artery and vertebral artery due to the placement of the catheter [25].
Among natural causes of bleeding, aortic dissection is by far the most common and serious condition affecting the aorta. The ascendant aortic dissection represents a severe condition that may lead to death in a large number of cases, due to the rapidity of the pathological process. It is usually a consequence of chronic hypertension or other rare conditions (such as Marfan’s syndrome, aortic coarctation, and bicuspid aortic valve). Since dissection can involve any aortic segment, the disease can manifest itself in a variety of clinical manifestations [28]. In fact, when aortic dissection occurs, aortic branches occlusion may happen. In case of dissections of the ascending aorta, the major aortic branches are occluded, resulting in rapidly fatal complications such as cardiac tamponade, major stroke, or massive myocardial infarction [29–31]. If, on the one side, the evaluation in living subjects of cardiac tamponade due to a hemopericardium, using diagnostic imaging techniques with special regard to the CT, allows a good interpretation of the anatomy and the pathology of the pericardium [32, 33], on the other side, in postmortem investigations, only a few studies have used PMCT imaging to investigate cardiac tamponade in the last decade [34–38] (see Table 2) and the results obtained show the feasibility of using the PMCT technique to diagnose haemopericardium and cardiac tamponade in cadavers. However, despite the advantages of MDCT in terms of performance simplicity, data acquisition, and cost effectiveness, this technique has the disadvantage of limited organ parenchyma and vascular system visualization; therefore, a greater contribution for the detection of cardiac tamponade can be made using the PMCTA technique, application of which is presently very limited. Bello et al. [39] have recently reported a case involving a 72-year-old man, in which MPMCTA was helpful in defining the diagnosis, detecting a haemopericardium and the ruptured wall situated in the posterior part of the left ventricle. The autopsy was then performed, totally confirming the CT angiography findings.
Ischemic heart disease
Ischemic heart disease (IHD) as a consequence of atherosclerosis represents the most common cause of mortality in developed countries [40]. In postmortem investigation, the diagnosis of IHD is usually made after a complete autopsy, supported by macroscopic and microscopic examination of the heart, with the classical histological stainings and also the contribution of immunohistochemistry. However, in recent years, postmortem cardiac imaging has been introduced with both targeted coronary CT angiography techniques [41, 42] using minimally invasive approaches and MPMCTA involving the whole body [8], which allows the filling of the arterial and venous systems, including coronary arteries. The postmortem investigation of IHD differs in several aspects from the clinical one, because the application of certain techniques, such as electrocardiography and several laboratory analyses, after death is not possible [43].
Regarding postmortem coronary angiography, two English centers, Leicester [41] and Oxford [42], independently developed two separate postmortem coronary CTA techniques, both using the left carotid artery to insert a urinary catheter. A catheter balloon is then inflated in the ascending aorta, and the contrast medium is injected into the aortic root and as a consequence coronary arteries are filled. Some differences can be highlighted between the Leicester and the Oxford method; according to the first one [41], both air and contrast media are injected, whereas in the Oxford method only, the contrast agent is injected [42]. Moreover, in Leicester, a clinical CT power-contrast injector is used, whereas in Oxford, a manual injection of the contrast agent is performed.
Michaud et al. [22] evaluated postmortem MDCT and MDCT-angiography for the investigation of sudden cardiac death related to atherosclerotic coronary artery disease. For this purpose, 23 fatal cases suggestive of IHD were retrospectively selected. The results obtained show that native CT-scan was not significantly useful for the diagnosis of IHD, confirming the previous studies [44–46]. Only calcifications of coronary arteries were detected in 18 cases, although their presence in atherosclerotic plaques is a common finding, especially in elderly subjects. Moreover, the extent of calcification does not correlate with the degree of coronary artery stenosis and plaques involved in acute thrombosis show a lower degree of calcification than the ones associated with clinically stable angina [47, 48].
According to Michaud et al. [22], postmortem CT-angiography has allowed the obtaining of better images of coronary arteries than native CT, enabling the evaluation of stenoses and occlusions. An acute coronary thrombosis was found in 13 cases: 7 cases were associated to plaque rupture, whereas the remaining 6 to plaque erosion. In all 13 cases, PMCTA found “suspicious” segments of coronary arteries. The results reported in this study support the application of MPMCTA as a reasonable tool to use in order to exclude coronary artery stenosis and to highlight possible occlusions to point out the subsequent sampling for histology. In 5 of the 13 cases reported by Michaud et al. [22], a pathological enhancement of the myocardium, which is considered an indirect sign of myocardial ischemia, was found.
Pathological enhancement of the myocardium was also observed by Palmiere et al. [49] who investigated the detection of coronary thrombosis by using MPMCTA in 150 cases, according to the protocol of Grabherr et al. [8]. Pathological enhancement was found in some of the cases showing coronary luminal filling defects without collateral vessels, as well as in others presenting complete or incomplete coronary arterial luminal filling defects and the presence of collateral vessels. These findings are suggestive of myocardial lesions due to infarction. However, a limitation of this study [49] was that the authors have not correlated myocardial enhancements detected at angiography with the histological findings. Currently, the precise implication of myocardial enhancements is not completely understood because there could also be a correlation with postmortem changes. However, according to Palmiere et al. [49], myocardial enhancements detected at angiography seem to correspond well to the areas of infarction as long as the infarction is not in its acute phase.
Lastly, another point worthy of attention highlighted both by Michaud [22] and Palmiere [49] in both series of cases is that although PMCTA allows a reliable diagnosis of acute coronary thrombosis, the precise location and extent of suspected coronary thrombi cannot be exactly established before the PMCTA; therefore, their eventual dislodging due to the introduction of the contrast medium into the vascular system cannot be definitely excluded.
As stated by Michaud et al. [22], the advantage of MPMCTA is that there is no direct mechanical manipulation of the coronary arteries, because the site of cannulation is placed in one of the two inguinal regions and the perfusion pressure is very low (1.200 ml in 90 s for the arterial phase); therefore, the perfusion flow in the coronary arteries is slower than in vivo.
Pulmonary thromboembolism
Venous thromboembolism (VTE) includes pulmonary thromboemboli (PE) and deep venous thrombosis (DVT), typical of the pelvic and lower-extremity veins. The incidences of pulmonary embolism and DVT are difficult to calculate and are commonly underdiagnosed. However, not all pulmonary emboli are fatal [50, 51]. Pulmonary emboli have been estimated to occur in more than 600,000 patients per year in the United States and result in 50,000–200,000 deaths annually. Approximately 11 % of patients with an acute PE die within 1 h; this accounts for two-thirds of the fatalities caused by PE. The majority of subjects who died of a PE were never appropriately treated because the diagnosis was not made. The mortality rate of an acute untreated PE is 30–35 % [50–53].
The diagnosis of pulmonary thromboembolism may be burdened by serious legal and medical consequences. Moreover, the identification of pulmonary thromboembolism as a cause of sudden death may lead to the identification of a familial clotting disorder and could subsequently be lifesaving for relatives of the deceased [54]. Recently, a few reports of PMCTA for the diagnosis of PE have been reported in literature [9, 54, 55].
A research group of the Victorian Institute of Forensic Medicine in Melbourne, examined 13 cases where PT was clinically suspected. All subjects died in hospital, and they underwent PMCT pulmonary angiography (PMCTPA) and subsequent conventional full autopsy. A femoral vein was cannulated on one side of the body and the contrast agent injected according to a modified technique of Ross et al. [12]. It is interesting to take into consideration the criteria used by Burke et al. [54] for the interpretation of PMCTPA; all of them refer to “filling defects,” and they are summarized in Table 3.
In seven of the 13 cases examined by the Australian group, PMCTPA allowed the following diagnosis: PT to major vessels in 5 cases, massive PT in 1 case, and probable segmental PT in another case. The subsequent autopsy confirmed the PMCTPA findings in all cases. It is noteworthy to highlight that in the 13 cases reported by Burke and colleagues, there were no false negatives; the six cases where the PMCTPA excluded the presence of PT, were all confirmed successively following autopsy.
An Italian group of the University of Foggia developed an alternative approach suitable in cases of fatal PE [9]; they modified the standard procedure established by the Technical Working Group Postmortem Angiography Methods [8], which uses the femoral vessels on one side of the corpse as an access point to the vascular system by isolating and then cannulating the axillary artery and vein on one side of the corpse. The cannulas were then normally connected to the tube system of the perfusion device. This alternative approach allows, in comparison to the traditional one [9], which excludes the vascular tree below the cannula insertion where lesions such as thrombosis of the superficial and deep venous system remain elusive, a reliable recognition of a filling defect or inhomogeneous opacification, facilitating therefore a visual inspection of the exact site of the suspected venous thrombus. This method makes it at the same time easier and more possible for the forensic pathologist to check the correct site in the venous system of the lower extremities and identify the venous thrombus for subsequent histological examination [9].
Further applications of PMCT and PMCTA in forensic investigation
Vogel et al. evaluated in two different studies [56, 57] the most significant findings provided by PMCT and PMCTA after transvascular cardiac interventions [56] and cardiac surgery [57]. For this purpose, the authors retrospectively reviewed their archive of PMCT and PMCTA cases. In those cases, in which the death of a patient occurs during the surgical intervention or shortly after, is very important to establish if the cause of death is due to a complication of the surgical procedure, a medical error or if it was not related to surgery. A postmortem CT and/or CTA may help to address these issues. Regarding transvascular interventions, Vogel et al. [56] examined PMCT and PMCTA findings after coronary angiography, coronary angioplasty (PTCA), stent placement, transarterial valve implantation (TAVI), mitral clips, transvascular annuloplasty, and pacemaker placement. PMCT detected previous contrast medium injections, the presence of blood (e.g., in pleura, mediastinum, pericardium, groin) and its amount, and the presence or absence of air bubbles in vessels. PMCTA added further information to PMCT findings, such as the source of a bleeding, the passage of blood through abnormally compromised ostia, the occlusion of coronary arteries, covered and free perforation, and dysfunctions of implanted valves.
PMCT has found in the new millennium several other applications [58–84] in forensic investigation, e.g., for the detection of spine injuries [60], gunshot wounds [61], strangulation [59] (Fig. 4), drowning [63], rib fractures [67], blunt force traumas [69], body packers [75], and many others [76–84].
In Table 4, some of the reports published in literature are summarized during the last 10 years, trying to highlight benefits and disadvantages of the application of PMCT in forensic practice.
Conclusions
The use of PMCT in forensic investigation has become routine in several Institutes of Legal Medicine. MDCT is a rapid and easy way to look inside the body and document findings. Moreover, it provides the chance to revise stored data at any time, even after cremation or burial of the corpse. Currently, the management of a MDCT unit is easier, and its maintenance costs are more affordable, making it a valid tool available in numerous Forensic Science Centers [85–88].
The diagnostic value of MDCT compared to full autopsy has been broadly investigated, and numerous papers have been published in this field. However, if, on one the hand, native CT is able to detect major vascular lesions (e.g., aortic rupture), on the other hand, its main limitation is represented by its low ability to visualize the vascular system together with the soft tissue. The recent introduction of some techniques of PMCTA with the injection of a contrast medium into the vessels has allowed the visualization of the whole vascular system in a minimally invasive way [7–12, 85]. Some standardized protocols have been developed and published; their application would allow making the procedure more easily reproducible and the operators aware of possible artifacts that its application can generate. In this regard, Bruguier and colleagues [89] have recently published a study in which they have evaluated technique-related artifacts in 54 cases in which the MPMCTA was performed according to the protocol proposed by Grabherr et al. [8]. The identification and categorization of these artifacts according to their type, anatomical location, and timing of appearance during the angiography are of utmost importance, because the MPMCTA technique was developed with the aim of applying it in routine forensic investigations. The results obtained by this preliminary study [88] show essentially three groups of artifacts/pitfalls which may be misinterpreted for pathological findings; they are reassumed in Table 5. The identification of these artifacts and “an understanding of their meaning” are fundamental in order to guarantee a proper interpretation of PMCTA images.
However, if on the one side, the application of PMCTA protocols has numerous unquestionable advantages, on the other side, some disadvantages need to be discussed; the application of certain protocols requires the use of further equipment to be coupled to MDCT and the employment of dedicated personnel which result in costs that small institutions cannot always financially cope with.
PMCTA techniques are relatively new, and much more research is needed in this field. Presently, PMCTA still seems to be far from replacing the conventional autopsy, but it may represent a valid diagnostic complementary tool with encouraging prospects in the near future. As was already stated a few years ago, “…Legal systems around the world must accept the admissibility of imaging evidence in determining the cause and manner of death. Radiologists will also need special training in postmortem imaging, as cadaver imaging is very different from imaging living patients. However, digital autopsy is and will remain merely an aid in the practice of forensic medicine and one that is not always available and that cannot be considered an alternative to conventional postmortem procedures” [90]. This is, then and now, our thought and we trust that many of us share it.
References
Eckert WG, Garland N (1984) The history of the forensic applications in radiology. Am J Forensic Med Pathol 5:53–56
Roberts IS, Benamore RE, Benbow EW, Lee SH, Harris JN, Jackson A, Mallett S, Patankar T, Peebles C, Roobottom C, Traill ZC (2012) Post-mortem imaging as an alternative to autopsy in the diagnosis of adult deaths: a validation study. Lancet 379:136–142
Wichmann D, Obbelode F, Vogel H, Hoepker WW, Nierhaus A, Braune S, Sauter G, Pueschel K, Kluge S (2012) Virtual autopsy as an alternative to traditional medical autopsy in the intensive care unit: a prospective cohort study. Ann Intern Med 156:123–130
Krantz P, Holtås S (1983) Postmortem computed tomography in a diving fatality. J Comput Assist Tomogr 7:132–134
Grabherr S, Grimm J, Dominguez A, Vanhaebost J, Mangin P (2014) Advances in post-mortem CT-angiography. Br J Radiol 87(1036):20130488
Jackowski C, Sonnenschein M, Thali MJ, Aghayev E, von Allmen G, Yen K, Dirnhofer R, Vock P (2005) Virtopsy: postmortem minimally invasive angiography using cross section techniques—implementation and preliminary results. J Forensic Sci 50:1175–1186
Grabherr S, Gygax E, Sollberger B, Ross S, Oesterhelweg L, Bolliger S, Christe A, Djonov V, Thali MJ, Dirnhofer R (2008) Two-step postmortem angiography with a modified heart-lung machine: preliminary results. AJR Am J Roentgenol 190:345–351
Grabherr S, Doenz F, Steger B, Dirnhofer R, Dominguez A, Sollberger B, Gygax E, Rizzo E, Chevallier C, Meuli R, Mangin P (2011) Multi-phase post-mortem CT angiography: development of a standardized protocol. Int J Legal Med 125:791–802
Pomara C, Bello S, Grilli G, Guglielmi G, Turillazzi E (2014) Multi-phase postmortem CT angiography (MPMCTA): a new axillary approach suitable in fatal thromboembolism. Radiol Med. doi:10.1007/s11547-014-0467-z
Jackowski C, Bolliger S, Aghayev E, Christe A, Kilchoer T, Aebi B, Périnat T, Dirnhofer R, Thali MJ (2006) Reduction of postmortem angiography-induced tissue edema by using polyethylene glycol as a contrast agent dissolver. J Forensic Sci 51:1134–1137
Jackowski C, Persson A, Thali MJ (2008) Whole body postmortem angiography with a high viscosity contrast agent solution using polyethylene glycol as contrast agent dissolver. J Forensic Sci 53:465–468
Ross S, Spendlove D, Bolliger S, Christe A, Oesterhelweg L, Grabherr S, Thali MJ, Gygax E (2008) Postmortem whole-body CT angiography: evaluation of two contrast media solutions. AJR Am J Roentgenol 190:1380–1389
Iizuka K, Sakamoto M, Kawasaki H, Miyoshi T, Komatsuzaki A, Kikuchi S (2009) Examination of the usefulness of the contrast-enhanced Post mortem CT imaging diagnosis. Innervision 24:89–92
Sakamoto N, Senoo S, Kamimura Y, Uemura K (2009) Case report: cardiopulmonary arrest on arrival case which underwent contrast-enhanced postmortem CT. J Jap Assoc Acute Med 30:114–115
Jolibert M, Cohen F, Bartoli C, Boval C, Vidal V, Gaubert JY, Moulin G, Petit P, Bartoli JM, Leonetti G, Gorincour G (2011) Postmortem CT-angiography: feasibility of US-guided vascular access. J Radiol 92:446–449
Becker CD, Poletti PA (2005) The trauma concept: the role of MDCT in the diagnosis and management of visceral injuries. Eur Radiol 15(Suppl 4):D105–D109
Scalici E, Indorato F, Portelli F, Savì T, Maresi E, Busardò FP (2014) A fatal iatrogenic right vertebral injury after transoral odontoidectomy and posterior cervical stabilization for a type II odontoid fracture. J Forensic Leg Med 22:41–44
Busardò FP, Frati P, Carbone I, Pugnetti P, Fineschi V (2015) Iatrogenic left common iliac artery and vein perforation during lumbar discectomy: a fatal case. Forensic Sci Int 246:e7–e11
Nguyen D, Platon A, Shanmuganathan K, Mirvis SE, Becker CD, Poletti PA (2009) Evaluation of single-pass continuous whole-body 16-MDCT protocol for patients with polytrauma. AJR Am J Roentgenol 192:3–10
Ruder TD, Ketterer T, Preiss U, Bolliger M, Ross S, Gotsmy WF, Ampanozi G, Germerott T, Thali MJ, Hatch GM (2011) Suicidal knife wound to the heart: challenges in reconstructing wound channels with post-mortem CT and CT-angiography. Leg Med (Tokyo) 13:91–94
Kominato Y, Fujikura T, Hata Y, Matsui K, Takizawa H (2004) A case of postoperative hemorrhage after a hysterectomy in which a bleeding point of the left uterine artery was identified by postmortem angiography. Leg Med (Tokyo) 6:187–189
Michaud K, Grabherr S, Doenz F, Mangin P (2012) Evaluation of postmortem MDCT and MDCT-angiography for the investigation of sudden death related atherosclerotic coronary artery disease. Int J Cardiovasc Imaging 28:1807–1822
Palmiere C, Binaghi S, Doenz F, Bize P, Chevallier C, Mangin P, Grabherr S (2012) Detection of hemorrhage source: the diagnostic value of post-mortem CT-angiography. Forensic Sci Int 222:33–39
Pomara C, Bello S, Serinelli S, Fineschi V (2015) A rare and lethal case of right common carotid pseudoaneurysm following whiplash trauma. Forensic Sci Med Pathol 11:69–73
Inokuchi G, Yajima D, Hayakawa M, Motomura A, Chiba F, Torimitsu S, Makino Y, Iwase H (2014) Postmortem dynamic cerebral angiography for detecting aneurysm and bleeding sites in cases of subarachnoid hemorrhage. Forensic Sci Med Pathol 10:487–495
Aghayev E, Thali M, Jackowski C, Sonnenschein M, Yen K, Vock P, Dirnhofer R (2004) Virtopsy—fatal motor vehicle accident with head injury. J Forensic Sci 49:809–813
Ikegaya H, Yajima D, Iwase H (2008) Vertebral artery rupture in a sudden death case after mild trauma. Am J Forensic Med Pathol 29:276–278
Lardi C, Lobrinus JA, Doenz F, Fracasso T, Augsburger M, Mangin P, Grabherr S (2014) Acute aortic dissection with carotid and coronary malperfusion: from imaging to pathology. Am J Forensic Med Pathol 35:157–162
Fleischmann D, Mitchell RS, Miller DC (2008) Acute aortic syndromes: new insights from electrocardiographically gated computed tomography. Semin Thorac Cardiovasc Surg 20:340–347
Hiratzka LF, Bakris GL, Beckman JA et al (2010) Guidelines for the diagnosis and management of patients with thoracic aortic disease: executive summary. JACC 55:1509–1544
Hagan PG, Nienaber CA, Isselbacher EM et al (2000) The international registry of acute aortic dissection (IRAD): new insights into an old disease. JAMA 283:897–903
Oyama N, Oyama N, Komuro K, Nambu T, Manning WJ, Miyasaka K (2004) Computed tomography and magnetic resonance imaging of the pericardium: anatomy and pathology. Magn Reson Med Sci 3:145–152
Restrepo CS, Lemos DF, Lemos JA, Velasquez E, Diethelm L, Ovella TA, Martinez S, Carrillo J, Moncada R, Klein JS (2007) Imaging findings in cardiac tamponade with emphasis on CT. Radiographics 27:1595–1610
Ebert LC, Ampanozi G, Ruder TD, Hatch G, Thali MJ, Germerott T (2012) CT based volume measurement and estimation in cases of pericardial effusion. J Forensic Leg Med 19:126–131
Filograna L, Flach PM, Bolliger SA, Thali MJ (2014) The role of post-mortem CT (PMCT) imaging in the diagnosis of pericardial tamponade due to hemopericardium: a case report. Leg Med Tokyo 16:150–153
Filograna L, Hatch G, Ruder T, Ross SG, Bolliger SA, Thali MJ (2013) The role of post-mortem imaging in a case of sudden death due to ascending aorta aneurysm rupture. Forensic Sci Int 228:e76–e80
Huang P, Wan L, Qin Z, Zhang J, Liu N, Zou D, Chen Y (2012) Post-mortem MSCT diagnosis of acute pericardial tamponade caused by blunt trauma to the chest in a motor-vehicle collision. Rom J Leg Med 20:117–122
Shiotani S, Watanabe K, Kohno M, Ohashi N, Yamazaki K, Nakayama H (2004) Postmortem computed tomographic (PMCT) findings of pericardial effusion due to acute aortic dissection. Radiat Med 22:405–407
Bello S, Neri M, Grilli G, Pascale N, Pomara C, Riezzo I, Turillazzi E, Fineschi V (2014) Multi-phase post-mortem ct-angiography (MPMCTA) is a very significant tool to explain cardiovascular pathologies. A sudden cardiac death case. Exp Clin Cardiol 20:1419–1430
Chugh SS, Reinier K, Teodorescu C, Evanado A, Kehr E, Al Samara M, Mariani R, Gunson K, Jui J (2008) Epidemiology of sudden cardiac death: clinical and research implications. Prog Cardiovasc Dis 51:213–228
Saunders SL, Morgan B, Raj V, Robinson CE, Rutty GN (2011) Targeted post-mortem computed tomography cardiac angiography: proof of concept. Int J Legal Med 125:609–616
Roberts IS, Benamore RE, Peebles C, Roobottom C, Traill ZC (2011) Technical report: diagnosis of coronary artery disease using minimally invasive autopsy: evaluation of a novel method of post-mortem coronary CT angiography. Clin Radiol 66:645–650
Michaud K, Grabherr S, Jackowski C, Bollmann MD, Doenz F, Mangin P (2014) Postmortem imaging of sudden cardiac death. Int J Legal Med 128:127–137
Jeffery AJ (2010) The role of computed tomography in adult post-mortem examinations: an overview. Diagn Histopathol 16:546–551
O’Donnell C (2010) An image of sudden death: utility of routine post-mortem computed tomography scanning in medico-legal autopsy practice. Diagn Histopathol 16:552–555
Weustink AC, Hunink MGM, van Dijke CF, Renken NS, Krestin GP, Oosterhuis JW (2009) Minimally invasive autopsy: an alternative to conventional autopsy? Radiology 250:897–904
Oudkerk M, Stillman A, Halliburton S, Kalender W, Mohlenkamp S, McCollough C, Vliegenthart R, Shaw L, Stanford W, Taylor A, van Ooijen P, Wexler L, Raggi P (2008) Coronary artery calcium screening: current status and recommendations from the European society of cardiac radiology and North American society for cardiovascularimaging. Int J Cardiovasc Imaging 24:645–671
Sanz J, Moreno PR, Fuster V (2010) The year in atherothrombosis. J Am Coll Cardiol 55:1487–1498
Palmiere C, Lobrinus JA, Mangin P, Grabherr S (2013) Detection of coronary thrombosis after multi-phase postmortem CT-angiography. Leg Med (Tokyo) 15:12–18
Tsokos M (2005) Forensic pathology reviews, vol 3. Humana Press Inc., Totowa, p 286
Consensus-Conference (2002) College of American pathologists consensus conference XXXVI: diagnostic issues in thrombophilia. Arch Pathol Lab Med 126:1277–1433
Wood KE (2002) Major pulmonary embolism: review of a pathophysiologic approach to the golden hour of hemodynamically significant pulmonary embolism. Chest 121:877–905
White RH (2003) The epidemiology of venous thromboembolism. Circulation 107:14–18
Burke MP, Bedford P, Baber Y (2014) Can forensic pathologists diagnose pulmonary thromboembolism on postmortem computed tomography pulmonary angiography? Am J Forensic Med Pathol 35:124–131
Kikuchi K, Kawahara K, Tsuji C, Tajima Y et al (2010) Post mortem contrast-enhanced computed tomography in a case of sudden death from acute pulmonary thromboembolism. Exp Ther Med 1:503–505
Vogel B, Heinemann A, Gehl A, Hasegawa I, Höpker WW, Poodendaen C, Tzikas A, Gulbins H, Reichenspurner H, Püschel K, Vogel H (2013) Post-mortem computed tomography (PMCT) and PMCT-angiography after transvascular cardiac interventions. Arch Med Sadowej Kryminol. 63:255–266
Vogel B, Heinemann A, Tzikas A, Poodendaen C, Gulbins H, Reichenspurner H, Püschel K, Vogel H (2013) Post-mortem computed tomography (PMCT) and PMCT-angiography after cardiac surgery. Possibilities and limits. Arch Med Sadowej Kryminol 63:155–171
Sogawa N, Michiue T, Ishikawa T, Kawamoto O, Oritani S, Maeda H (2014) Postmortem volumetric CT data analysis of pulmonary air/gas content with regard to the cause of death for investigating terminal respiratory function in forensic autopsy. Forensic Sci Int 241:112–117
Maiese A, Gitto L, dell’Aquila M, Bolino G (2014) When the hidden features become evident: the usefulness of PMCT in a strangulation-related death. Leg Med (Tokyo) 16:364–366
Oshima T, Hayashida M, Ohtani M, Hashimoto M, Takahashi S, Ishiyama K, Otani T, Koga M, Sugawara M, Mimasaka S (2014) Spinal hyperostosis as an important sign indicating spine injuries on postmortem computed tomography. Leg Med (Tokyo) 16:197–200
Maiese A, Gitto L, De Matteis A, Panebianco V, Bolino G (2014) Post mortem computed tomography: useful or unnecessary in gunshot wounds deaths? Two case reports. Leg Med (Tokyo) 16:357–363
Lee WI, Lee J, Bassed R, O’Donnell C (2014) Post-mortem CT findings in a case of necrotizing cellulitis of the floor of the mouth (Ludwig angina). Forensic Sci Med Pathol 10:109–113
Lo Re G, Vernuccio F, Galfano MC, Picone D, Milone L, La Tona G, Argo A, Zerbo S, Salerno S, Procaccianti P, Midiri M, Lagalla R (2014) Role of virtopsy in the post-mortem diagnosis of drowning. Radiol Med. doi:10.1007/s11547-014-0438-4
Oshima T, Yoshikawa H, Dewa K, Ohtani M, Nakauchi K, Mimasaka S (2014) Four cases of orbital hyperdensity identified by postmortem computed tomography. Leg Med (Tokyo). doi:10.1016/j.legalmed.2014.08.001
Flach PM, Egli TC, Bolliger SA, Berger N, Ampanozi G, Thali MJ, Schweitzer W (2014) “Blind spots” in forensic autopsy: improved detection of retrobulbar hemorrhage and orbital lesions by postmortem computed tomography (PMCT). Leg Med (Tokyo) 16:274–282
Sieswerda-Hoogendoorn T, Soerdjbalie-Maikoe V, Maes A, van Rijn RR (2013) The value of post-mortem CT in neonaticide in case of severe decomposition: description of 12 cases. Forensic Sci Int 233:298–303
Schulze C, Hoppe H, Schweitzer W, Schwendener N, Grabherr S, Jackowski C (2013) Rib fractures at postmortem computed tomography (PMCT) validated against the autopsy. Forensic Sci Int 233:90–98
Noda Y, Yoshimura K, Tsuji S, Ohashi A, Kawasaki H, Kaneko K, Ikeda S, Kurokawa H, Tanigawa N (2013) Postmortem computed tomography imaging in the investigation of nontraumatic death in infants and children. Biomed Res Int 2013:327903
Daly B, Abboud S, Ali Z, Sliker C, Fowler D (2013) Comparison of whole-body post mortem 3D CT and autopsy evaluation in accidental blunt force traumatic death using the abbreviated injury scale classification. Forensic Sci Int 225:20–26
Sano R, Hirasawa S, Awata S, Kobayashi S, Shimada T, Takei H, Takahashi Y, Kominato Y (2013) Use of postmortem computed tomography to reveal acute subdural hematoma in a severely decomposed body with advanced skeletonization. Leg Med (Tokyo) 15:32–34
Makino Y, Shimofusa R, Hayakawa M, Yajima D, Inokuchi G, Motomura A, Iwase H (2013) Massive gas embolism revealed by two consecutive postmortem computed-tomography examinations. Forensic Sci Int 231:e4–e10
Ampanozi G, Schwendener N, Krauskopf A, Thali MJ, Bartsch C (2013) Incidental occult gunshot wound detected by postmortem computed tomography. Forensic Sci Med Pathol 9:68–72
Okuda T, Shiotani S, Hayakawa H, Kikuchi K, Kobayashi T, Ohno Y (2013) A case of fatal cervical discoligamentous hyperextension injury without fracture: correlation of postmortem imaging and autopsy findings. Forensic Sci Int 225:71–74
Bin Abdul Rashid SN, Krauskopf A, Vonlanthen B, Thali MJ, Ruder TD (2013) Sudden death in a case of sickle cell anemia: post-mortem computed tomography and autopsy correlation from a radiologist’s perspective. Leg Med (Tokyo) 15:213–216
Bin Abdul Rashid SN, Rahim AS, Thali MJ, Flach PM (2013) Death by ‘ice’: fatal methamphetamine intoxication of a body packer case detected by postmortem computed tomography (PMCT) and validated by autopsy. Forensic Sci Med Pathol 9:82–87
Fischer F, Grimm J, Kirchhoff C, Reiser MF, Graw M, Kirchhoff S (2012) Postmortem 24-h interval computed tomography findings on intrahepatic gas development and changes of liver parenchyma radiopacity. Forensic Sci Int 214:118–123
Høyer CB, Nielsen TS, Nagel LL, Uhrenholt L, Boel LW (2012) Investigation of a fatal airplane crash: autopsy, computed tomography, and injury pattern analysis used to determine who was steering the plane at the time of the accident. A case report. Forensic Sci Med Pathol 8:179–188
Flach PM, Ross SG, Bolliger SA, Ampanozi G, Hatch GM, Schön C, Thali MJ, Germerott T (2012) Massive systemic fat embolism detected by postmortem imaging and biopsy. J Forensic Sci 57:1376–1380
Ishida M, Gonoi W, Hagiwara K, Takazawa Y, Akahane M, Fukayama M, Ohtomo K (2011) Intravascular gas distribution in the upper abdomen of non-traumatic in-hospital death cases on postmortem computed tomography. Leg Med (Tokyo) 13:174–179
Thali YA, Bolliger SA, Hatch GM, Ampanozi G, Thali MJ, Ruder TD (2011) Death by biscuit—exhumation, post-mortem CT, and revision of the cause of death one year after interment. Leg Med (Tokyo) 13:142–144
Berens S, Ketterer T, Kneubuehl BP, Thali MJ, Ross S, Bolliger SA (2011) A case of homicidal intraoral gunshot and review of the literature Forensic. Sci Med Pathol 7:209–212
Uhrenholt L, Boel LW (2010) Contributions from forensic imaging to the investigation of upper cervical fractures. J Forensic Sci 55:1598–1602
Sochor MR, Trowbridge MJ, Boscak A, Maino JC, Maio RF (2008) Postmortem computed tomography as an adjunct to autopsy for analyzing fatal motor vehicle crash injuries: results of a pilot study. J Trauma 65:659–665
Shiotani S, Kohno M, Ohashi N, Yamazaki K, Nakayama H, Watanabe K, Oyake Y, Itai Y (2004) Non-traumatic postmortem computed tomographic (PMCT) findings of the lung. Forensic Sci Int 139:39–48
Chevallier C, Doenz F, Vaucher P, Palmiere C, Dominguez A, Binaghi S, Mangin P, Grabherr S (2013) Postmortem computed tomography angiography vs. conventional autopsy: advantages and inconveniences of each method. Int J Legal Med 127:981–989
Dirnhofer R, Jackowski C, Vock P et al (2006) VIRTOPSY: minimally invasive, imaging-guided virtual autopsy. Radiographics 26(5):1305–1333
Poulsen K, Simonsen J (2007) Computed tomography as a routine in connection with medico-legal autopsies. Forensic Sci Int 171(2–3):190–197
Flach PM, Gascho D, Schweitzer W, Ruder TD, Berger N, Ross SG, Thali MJ, Ampanozi G (2014) Imaging in forensic radiology: an illustrated guide for postmortem computed tomography technique and protocols. Forensic Sci Med Pathol 10:583–606
Bruguier C, Mosimann PJ, Vaucher P, Uské A, Doenz F, Jackowski C, Mangin P, Grabherr S (2013) Multi-phase postmortem CT angiography: recognizing technique-related artefacts and pitfalls. Int J Legal Med 127:639–652
Pomara C, Fineschi V, Scalzo G, Guglielmi G (2009) Virtopsy versus digital autopsy: virtuous autopsy. Radiol Med 114:1367–1382
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
This article does not contain any studies with human participants or animals performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Busardò, F.P., Frati, P., Guglielmi, G. et al. Postmortem-computed tomography and postmortem-computed tomography–angiography: a focused update. Radiol med 120, 810–823 (2015). https://doi.org/10.1007/s11547-015-0559-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-015-0559-4