Abstract
As the fourth most important food crop, potato plays a key role in food safety and economic development of the world. Harvested potato tubers can be stored for a long time, but sprouting and cold-induced sweetening (CIS) can seriously affect the quality of tubers during storage. One of the key pathways involved in CIS is starch degradation, in which both α-amylase and β-amylase play important roles. However, each amylase belongs to an extensive gene family and it is not clear which genes are the key regulators. In this study, we identified genes most likely regulating starch degradation. We first selected candidate genes from the public potato genome database and then investigated their expression patterns associated with reducing sugars and amylase activities. The results showed that the activity of α-amylase was mainly caused by StAmy23 and the activity of β-amylase was mainly caused by StBAM1 and StBAM7. In addition, α-amylase and β-amylase may play important roles in starch degradation of the tubers stored at low temperature and during sprouting, and the amylase activity may be regulated by the amylase inhibitor in cold-stored tubers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Potato (Solanum tuberosum L.) is the fourth most important crop behind the cereal species rice (Oryza sativa), wheat (Triticum aestivum), and maize (Zea mays) and plays crucial roles in food security and economic development in the world. Tubers contain high-quality proteins and substantial amounts of vitamins, minerals, and trace elements. Therefore, there is a huge demand for potato tubers all year round. However, potato is grown in moderate climates, so continuous cultivation throughout the year is impossible, which makes tuber storage essential.
During storage, sprouting seriously affects potato quality since tubers need a large amount of water and nutrients to break bud dormancy. Previous studies have shown that bud dormancy can be regulated by changing hormones (Horvath et al. 2003; Rohde et al. 2007). The maintenance of tuber bud dormancy seems to involve abscisic acid (ABA) (Suttle 1995; Biemelt et al. 2000), whereas bud dormancy release possibly needs gibberellins (GA) and cytokinins (Suttle 2004; Hartmann et al. 2011). Starch degradation is also involved in the process of dormancy and sprouting, but the relationship between amylase activity and sprouting is not clear. Bailey et al. (1978) reported that a transient increase in α-amylase occurred at the time of sprouting. Biemelt et al. (2000) found an increase of both α-amylase and β-amylase in the sub-eye tissue after the onset of sprouting. However, Davies and Viola (1988) reported a decrease in total amylase activity during sprouting.
When potato tubers are stored under cold conditions post harvest, sprouting is inhibited but reducing sugars (RS) can accumulate, a process known as cold-induced sweetening (CIS). RS can cause a nonenzymatic Maillard reaction at a high frying temperature, which leads to unacceptable dark-colored products (Talburt et al. 1975). More concernedly, this reaction can also generate carcinogen acrylamide (Shepherd et al. 2010). The carbohydrate metabolic pathways of potato tubers have been extensively investigated, and some genes encoding the enzymes involved in the CIS pathway of potato tubers have been studied. In order to clarify the key pathways of CIS, the expression pattern of 188 genes from the cDNA microarray was analysed and results showed that amylolysis, sucrose decomposition, and glycolysis pathways play important roles in potato CIS (Chen et al. 2012).
Starch degradation occurs hydrolytically or phosphorolytically. The hydrolytic pathway involves enzymes like α-amylase and β-amylase, while the phosphorolytic pathway involves starch phosphorylase (Preiss 1982; Solomos and Mattoo 2005). It was found that the activity of starch-degrading enzymes was increased in the process of potato tuber CIS (Cottrell et al. 1993) although, previously, there were some arguments for a predominant pathway (Morrell and Rees 1986) and incompatible results between potato genotypes (Claassen et al. 1993; Hill et al. 1996). It has been shown that a potato RING finger gene SbRFP1 plays an important role in inhibiting β-amylase, which consequently slows down starch degradation and the accumulation of reducing sugars in cold-stored tubers (Zhang et al. 2013). With the release of genome information, genes encoding enzymes related to starch degradation are now available. However, their functions in potato CIS are still unclear.
In this study, we selected genes encoding enzymes related to starch degradation and analysed their expression patterns. We found a few amylase genes that may play important roles in starch degradation and further discussed the regulation mechanisms of the amylase activity.
Methods and Materials
Genes Related to Starch Degradation
Genes encoding key enzymes in the process of starch degradation were tested in this study. Two α-amylase genes, seven β-amylase genes, five isoamylase genes, two starch phosphorylase genes, and two amylase inhibitor genes were selected from the Potato Genome Sequence Consortium (PGSC) database and the National Center of Biotechnology Information (NCBI, http://www.ncbi.nlm.nih.gov/) database. Amino acid sequences of the β-amylase and isoamylase genes of Arabidopsis thaliana were selected from the NCBI database. The phylogenetic tree was constructed using CLUSTAL W (2.0) (Tompson et al. 1997) with neighbor-joining 1,000 bootstrap analysis. The tree obtained was viewed using TreeView software (version 1.6.6).
Plant Materials and Tuber Storage
CIS-tolerant potato genotypes (Solanum berthaultii, AC030-06, and AC142-01) and CIS-sensitive genotypes (AC035-01, AC041-03, and E3) were used in the present research (Liu et al. 2010). The plants were grown at 20–25 °C in diameter of 24 cm plastic pots in the greenhouse of Huazhong Agricultural University (Wuhan, China) with 16 h light per day, supplemented with mercury lamps. Leaves and stems were sampled at tuber formation stage about 45 days after planting. When mature tubers were harvested, a portion of them was kept at 20 °C in darkness. After about 75 days of storage, tubers without visible outgrowths were considered to be before sprouting and tubers with outgrowths longer than 0.5 cm were considered to be after sprouting. Tuber discs 1.5 cm in diameter around the buds were sampled from tubers both before and after sprouting. For the analysis of cold storage, tubers were kept at 20 °C for 10 days for the skin set. Then, they were divided into two groups: one group was stored at 4 °C in darkness as the cold treatment and the other at 20 °C for comparison. Tubers were sampled at 0, 5, 15, 30, and 60 days after storage, immediately peeled and frozen in liquid nitrogen, and then stored at −80 °C for further molecular and biochemical analyses.
RNA Isolation and Real-Time qRT-PCR
RNA isolation and reverse transcription were performed as described previously (Liu et al. 2010). The real-time reverse-transcription PCR (qRT-PCR) was performed to quantify the transcripts on the CFX96TM real-time PCR system (Bio-Rad, USA). A total volume of 20 μL PCR contained 1 μL of cDNA, 0.6 μL primers (10 μmol L−1) for specific gene, and 1× SsoAdvancedTM SYBR® Green supermix (Bio-Rad, USA). The amplification program was 95 °C for 30 s, 40 cycles of 95 °C for 5 s, and 56 °C for 30 s. Relative quantification of each individual gene expression was calculated with the 2−ΔCT method as described in the manufacturer’s introduction. Ef1α of potato (GenBank accession no. AB061263) was used as a control (Nicot et al. 2005). Primer Express v2.0 was employed for primer design, and all genes’ sequences blasted from the above-mentioned databases were referenced for primer design to affirm their specificity. Information and primers of the genes tested in this research are listed in Table 1.
Assessment of Enzyme Activity and Sugar Contents
Activities of amylases were quantified as described by Li et al. (2005). The amount of reducing sugars was determined as previously described (Ou et al. 2013). The data were shown as means of three repeats. Each test was repeated three times. Correlation analysis of the data was conducted using the Microsoft Excel program (Microsoft Office XP, 2010).
Results
Selection of Candidate Genes Related to Starch Degradation
Genes related to starch degradation were selected from the PGSC database and the NCBI database, including two α-amylase genes, StAmy1 (PGSC0003DMC400035807) and StAmy23 (PGSC0003DMC400017447) (Rentzsch et al. 2012); seven β-amylase genes (StBAM1, StBAM2, StBAM3, StBAM4, StBAM5, StBAM6, and StBAM7); five isoamylase genes (StISA1, StISA2, StISA3, StISA4, and StISA5); two starch phosphorylase genes, pho1 (Brisson et al. 1989) and pho2 (Lorberth et al. 1998), which represent cytosolic and plastidic starch phosphorylase, respectively; and two putative amylase inhibitor genes, StAI1 (PGSC0003DMC400063289) and SbAI from EST C20-3-E15 (GeneBank ID: HS989783) (Chen 2012). The β-amylase genes and isoamylase genes in potato were respectively clustered with the genes identified in A. thaliana. The cluster showed that StBAM1, StBAM2, StBAM4, StBAM5, StBAM6, and StBAM7 were most identical to AtBAM1, AtBAM3, AtBAM5, AtBAM7, AtBAM8, and AtBAM9, respectively, while StBAM3 had no homologs in Arabidopsis (Fig. 1a). StISA1, StISA2, and StISA3 were most identical to AtISA1, AtISA2, and AtISA3, respectively, while StISA4 and StISA5 were grouped together without Arabidopsis homologs (Fig. 1b).
Phylogenetic relationships of β-amylase genes (a) and isoamylase genes (b). Amino acid sequences of β-amylase genes are selected from Solanum tuberosum (StBAM1, PGSC0003DMC400002800; StBAM2, PGSC0003DMC400035625; StBAM3, PGSC0003DMC400021443; StBAM4, PGSC0003DMC400045472; StBAM5, PGSC0003DMC400000368; StBAM6, PGSC0003DMC400041754; StBAM7, PGSC0003DMC400018848) and Arabidopsis thaliana (AtBAM1, GenBank:AEE76832; AtBAM2, GenBank: AEE81889; AtBAM3, GenBank: AEE83848; AtBAM4, GenBank: AED96669; AtBAM5, GenBank: AEE83568; AtBAM6, GenBank: AEC08663; AtBAM7, GenBank: AEC10613; AtBAM8, GenBank: AED95229; AtBAM9, GenBank: AED92597). Amino acid sequences of the isoamylase genes are selected from Solanum tuberosum (StISA1, PGSC0003DMC400035985; StISA2, PGSC0003DMC400001818; StISA3, PGSC0003DMC400012889; StISA4, PGSC0003DMC400046519; StISA5, PGSC0003DMC400030763) and Arabidopsis thaliana (AtISA1, GenBank: AAQ56791; AtISA2, GenBank: AEE27557; AtISA3, GenBank: AEE82713)
Expression Analysis of Genes Related to Starch Degradation
The transcripts of genes related to starch degradation in different potato organs of AC030-06 and E3 were estimated by qRT-PCR. Results showed that the transcripts of the α-amylase gene StAmy23 were much higher than those of StAmy1 in the leaves, stems, and tubers at different storage times. In addition, StAmy23 had the highest expression in tubers before sprouting, and the expression decreased after sprouting. Moreover, the expression of StAmy23 increased when the tubers were stored at 4 °C for 30 days (Fig. 2a, d). These data indicate that StAmy23 may play roles in potato tubers during cold storage and sprouting.
For β-amylase, seven candidate genes were selected from the potato database (PGSC) and their expression was tested in AC030-06 and E3 tubers. Results showed that the expression of StBAM1 and StBAM7 was much higher than that of the rest of the candidate genes and increased sharply after cold storage, but there were no significant differences between the tubers before and after sprouting (Fig. 2b, e). The data indicated that the activity of β-amylase could be mainly caused by StBAM1 and StBAM7. We also tested the expression of amylase inhibitors and found that the expression of SbAI was much higher than that of StAI1 in all the organs tested (Fig. 2c, f), which indicated that the activity of amylase inhibitors was mainly caused by SbAI.
Five isoamylase genes were selected from the PGSC database, and their expression was tested. The results showed that two genes, StISA4 and StISA5, had much lower expression compared with the other three genes, StISA1, StISA2, and StISA3, while the expression patterns of these three genes in tubers during storage were inconsistent between AC030-06 and E3 (Fig. 3a, c). For pho1 and pho2, the expression levels were similar in AC030-06 tubers before and after cold storage (Fig. 3b). In E3, the expression of pho1 decreased after tubers were stored at 4 °C for 30 days and increased during sprouting, while the expression of pho2 decreased during both cold storage and sprouting (Fig. 3d). These results indicate that isoamylase and starch phosphorylase genes may not play important roles in potato tubers during cold storage and sprouting.
Analysis of StAmy23, StBAM1, StBAM7, and SbAI in Potato Tubers During Cold Storage
According to the above gene expression analysis, StAmy23, StBAM1, StBAM7, and SbAI were considered as important genes regulating tuber starch degradation (Fig. 2). In order to analyze their functions in cold-stored tubers, six potato genotypes which have a well-defined resistance to potato CIS were employed, including CIS-resistant genotypes S. berthaultii, AC030-06, and AC142-01 and CIS-sensitive genotypes AC035-01, AC041-03, and E3 (Liu et al. 2010). The expression of StAmy23, StBAM1, StBAM7, and SbAI in the cold-stored tubers was tested by qRT-PCR with a potato housekeeping gene ef1α (GenBank accession no. AB061263) as an internal control. The results showed that the expression of StAmy23 in S. berthaultii and AC030-06 was higher than in the other four genotypes (Fig. 4a). The expression of StBAM1 in tubers stored at 4 °C for 5 days was the highest in all tested genotypes and decreased afterwards (Fig. 4b). The expression of StBAM7 in tubers was increased after cold storage for 30 or 60 days (Fig. 4c). However, the expression levels of the three amylase genes in CIS-resistant tubers showed no significant differences compared to the CIS-sensitive tubers (Fig. 4a–c). The expression of amylase inhibitor gene SbAI in CIS-resistant tubers was much higher than that in CIS-sensitive tubers (Fig. 4d). To look into the relationship between amylase genes and CIS, the RS content (Liu et al. 2010) was plotted against the expression level of StAmy23, StBAM1, StBAM7, and SbAI in the cold-stored tubers of six different genotypes. A significant negative correlation between the RS content and the expression of SbAI was established (Fig. 4h), while no significant correlations between the RS content and the expression of StAmy23, StBAM1, and StBAM7 were observed (Fig. 4e–g). These results led to a conclusion that the roles of amylases in potato CIS may be regulated by the amylase inhibitor SbAI.
The relationship of StAmy23, StBAM1, StBAM7, SbAI, and potato CIS (a–d). Expression patterns of StAmy23, StBAM1, StBAM7, and SbAI in six potato tubers during 4 °C storage, respectively. e–h Correlation of the reducing sugar content and the relative expression of StAmy23, StBAM1, StBAM7, and SbAI, respectively. **P < 0.01, significant level; ns no significance
The activity of amylase in the six potato genotype tubers stored at 4 and 20 °C was analysed. In tubers stored at 20 °C, the amylase activity was at a low level in all six genotypes (Fig. 5b). When tubers were stored at 4 °C, the activity was increased sharply in E3 (from 0.12 to 0.69 stored for 30 days) and was increased in AC041-03 tubers stored for 60 days, but it was low in the other four genotypes (Fig. 5a). The amylase activity was not consistent with the expression of StAmy23, StBAM1, and StBAM7 (Fig. 4a–c), so they may be regulated by regulators such as amylase inhibitors.
Analysis of Amylase Activity in Potato Tubers During Sprouting
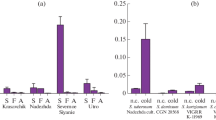
In order to identify the contribution of amylases to potato tubers during sprouting, reducing sugar amounts and amylase activities were measured. The RS amounts in tubers were decreased by about 67.3 and 46.2% after sprouting in E3 and AC030-06, respectively (Fig. 6a). The amylase activity in tubers was decreased by about 77.7% after sprouting in E3, while it was slightly increased in AC030-06 (Fig. 6b).
Discussion
Starch degradation is an important process in plants, especially in starch-excess organs like potato tubers. It is also important for potato tubers stored after harvest. However, genes related to starch degradation in potato CIS remain to be clarified. In this study, we selected candidate genes involved in starch degradation according to the available potato information and tested their expression. Our data indicate that StAmy23, StBAM1, and StBAM7 may play roles in tubers stored at low temperature and in the process of sprouting, and their activity may be regulated by amylase inhibitor gene SbAI.
Many enzymes were involved in the process of starch degradation, such as α-amylase, β-amylase, starch phosphorylase, and so on, but they may play different roles. When potato tubers were stored at 4 °C for 2 weeks, α-amylase activity and reducing sugar content increased sharply and showed significant deviation between different cultivars (Cottrell et al. 1993). We found that the StAmy23 silencing tubers showed a significant reduction in the accumulation of reducing sugars when stored at 4 °C for 15 days (data not shown). β-Amylase genes were considered to play key roles in the process of starch degradation, and different members may be expressed in different cellular compartments and play distinct roles (Valerio et al. 2011; Fulton et al. 2008). Antisense plants of a chloroplast-targeted β-amylase gene, PCT-BMY1 (StBAM2 in this paper), showed a starch-excess phenotype in leaves but not in cold-storage tubers (Scheidig et al. 2002). In the present research, two β-amylase genes, StBAM1 and StBAM7, out of the seven candidates showed higher expression levels, and their expression in tubers stored at 4 °C for 30 days was much higher than that in tubers stored at 4 °C for 0 day (Fig. 2b, e). This indicates that StBAM1 and StBAM7 may play roles in potato tubers during cold storage. In contrast, the starch phosphorylase and isoamylase may not be important in potato CIS according to our results (Fig. 3) although it was suggested that the increase in phosphorylase activity acts as a triggering event in potato CIS (Claassen et al. 1993).
The expression of StAmy23, StBAM1, and StBAM7 in CIS-resistant potato genotypes is higher than that in the CIS-sensitive potato genotype (Fig. 4a–c). But, their amylase activity is low during cold storage (Fig. 5a). SbAI is a putative amylase inhibitor, and here, we show that its expression is higher in cold-stored tubers of CIS-resistant genotypes than that of CIS-sensitive ones (Fig. 4d). It demonstrates that the amylase activity may be regulated by SbAI. However, the regulation mechanism of SbAI and the roles of StAmy23, StBAM1, StBAM7, and SbAI in potato starch degradation are still not clear. As we know, different amylase genes may be located in different cellular compartments, plastidic or cytosolic, and their location may affect their functions. StAmy23 and StBAM1 were located in cytosolic (data not shown), and the localization of StBAM7 and SbAI needs further investigation. In this study, we found that the amylase activity was only higher in one of the three CIS-sensitive genotypes (Fig. 5a), implying that there could be more pathways involved in regulating the potato CIS, such as sucrose cleavage. Invertase is the key enzyme in turning sucrose to reducing sugars, and suppression of acid invertase by silencing of StvacINV1 resulted in a strong decrease in RS accumulation in cold-stored tubers (Bhaskar et al. 2010; Liu et al. 2010).
Starch degradation can provide energy for potato to tuber dormancy breaking. In this study, we found that the expression of StAmy23 (Fig. 2a, d) and reducing sugar (Fig. 6a) contents in both AC030-06 and E3 tubers was significantly lower after sprouting. It suggested that the α-amylase gene StAmy23 may play important roles in sprouting. In addition, the StAmy23 RNAi-transgenic tubers sprouted later than the wild-type control (data not published). The amylase activity of AC03-06 tubers was slightly higher after sprouting (Fig. 6b) accompanied by a lower reducing sugar content in the present research (Fig. 6a). The reason could be that other pathways may also affect the accumulation of reducing sugars in tubers except for starch degradation (Malone et al. 2006).
In conclusion, our data indicate that several key regulators are involved in potato starch degradation. The activity of α-amylase is mainly caused by StAmy23, and StAmy23 plays an important role in the process of potato CIS and tuber sprouting. The activity of β-amylase is mainly caused by StBAM1 and StBAM7, and they may play roles only in potato CIS. Our data also indicate that the activity of amylases may be significantly regulated by the amylase inhibitor.
References
Bailey K, Phillips I, Pitt D (1978) The role of buds and gibberellin in dormancy and the mobilization of reserve materials in potato tubers. Ann Bot 42:649–657
Bhaskar PB, Wu L, Busse JS, Whitty BR, Hamernik AJ, Jansky SH, Buell CR, Bethke PC, Jiang J (2010) Suppression of the vacuolar invertase gene prevents cold-induced sweetening in potato. Plant Physiol 154:939–948
Biemelt S, Hajirezaei M, Hentschel E, Sonnewald U (2000) Comparative analysis of abscisic acid content and starch degradation during storage of tubers harvested from different potato varieties. Potato Res 43:371–382
Brisson N, Giroux H, Zollinger M, Camirand A, Simard C (1989) Maturation and subcellular compartmentation of potato starch phosphorylase. Plant Cell 11:559–566
Chen X (2012) Identification and profiling of the genes associated with cold-induced sweetening in Solanum berthaultii. Dissertation, Huazhong Agricultural University
Chen X, Song B, Liu J, Yang J, He T, Lin Y, Zhang H, Xie C (2012) Modulation of gene expression in cold-induced sweetening resistant potato species Solanum berthaultii exposed to low temperature. Mol Genet Genomics 287:411–421
Claassen PA, Budde MA, Van Calker MH (1993) Increase in phosphorylase activity during cold-induced sugar accumulation in potato tubers. Potato Res 36:205–217
Cottrell J, Duffus C, Paterson L, Mackay G, Allison M, Bain H (1993) The effect of storage temperature on reducing sugar concentration and the activities of three amylolytic enzymes in tubers of the cultivated potato, Solanum tuberosum L. Potato Res 36:107–117
Davies H, Viola R (1988) The effect of gibberellic acid on starch breakdown in sprouting tubers of Solanum tuberosum L. Ann Bot 61:689–693
Fulton DC, Stettler M, Mettler T, Vaughan CK, Li J, Francisco P, Gil M, Reinhold H, Eicke S, Messerli G, Dorken G, Halliday K, Smith AM, Smith SM, Zeeman SC (2008) β-AMYLASE4, a noncatalytic protein required for starch breakdown, acts upstream of three active β-amylases in Arabidopsis chloroplasts. Plant Cell 20:1040–1058
Hartmann A, Senning M, Hedden P, Sonnewald U, Sonnewald S (2011) Reactivation of meristem activity and sprout growth in potato tubers require both cytokinin and gibberellin. Plant Physiol 155:776–796
Hill LM, Reimholz R, SchrÖder R, Nielsen TH, Stitt M (1996) The onset of sucrose accumulation in cold-stored potato tubers is caused by an increased rate of sucrose synthesis and coincides with low levels of hexose-phosphates, an activation of sucrose phosphate synthase and the appearance of a new form of amylase. Plant Cell Environ 19:1223–1237
Horvath DP, Anderson JV, Chao WS, Foley ME (2003) Knowing when to grow: signals regulating bud dormancy. Trends Plant Sci 8:534–540
Li W, Shao Y, Chen W (2005) Improved method for determining amylase activity. Plant Physiol Commun 41:655–656
Liu X, Song B, Zhang H, Li X-Q, Xie C, Liu J (2010) Cloning and molecular characterization of putative invertase inhibitor genes and their possible contributions to cold-induced sweetening of potato tubers. Mol Genet Genomics 284:147–159
Lorberth R, Ritte G, Willmitzer L, Kossmann J (1998) Inhibition of a starch-granule-bound protein leads to modified starch and repression of cold sweetening. Nat Biotechnol l16:473–477
Malone JG, Mittova V, Ratcliffe RG, Kruger NJ (2006) The response of carbohydrate metabolism in potato tubers to low temperature. Plant Physiol 47:1309–1322
Morrell S, Rees TA (1986) Control of the hexose content of potato tubers. Phytochemistry 25:1073–1076
Nicot N, Hausman JF, Hoffmann L, Evers D (2005) Housekeeping gene selection for real-time RT-PCR normalization in potato during biotic and abiotic stress. J Exp Bot 56:2907–2914
Ou Y, Song B, Liu X, Lin Y, Zhang H, Li M, Fang H, Liu J (2013) Profiling of StvacINV1 expression in relation to acid invertase activity and sugar accumulation in potato cold-stored tubers. Potato Res 56:1–9
Preiss J (1982) Regulation of the biosynthesis and degradation of starch. Ann Rev Plant Physiol 33:431–454
Rentzsch S, Podzimska D, Voegele A, Imbeck M, Müller K, Linkies A, Leubner-Metzger G (2012) Dose-and tissue-specific interaction of monoterpenes with the gibberellin-mediated release of potato tuber bud dormancy, sprout growth and induction of α-amylases and β-amylases. Planta 235:137–151
Rohde A, Ruttink T, Hostyn V, Sterck L, Van Driessche K, Boerjan W (2007) Gene expression during the induction, maintenance, and release of dormancy in apical buds of poplar. J Exp Bot 58:4047–4060
Scheidig A, Fröhlich A, Schulze S, Lloyd JR, Kossmann J (2002) Downregulation of a chloroplast-targeted β-amylase leads to a starch-excess phenotype in leaves. Plant J 30:581–591
Shepherd LVT, Bradshaw JE, Dale MFB, McNicol JW, Pont SDA, Mottram DS, Davies HV (2010) Variation in acrylamide producing potential in potato: segregation of the trait in a breeding population. Food Chem 123:568–573
Solomos T, Mattoo AK (2005) Starch-sugar metabolism in potato (Solanum tuberosum L.) tubers in response to temperature variations. In: Razdan MK and Mattoo AK (ed) Genetic Improvement of Solanaceous Crops, 1st edn. Plymouth: 209–234
Suttle JC (1995) Postharvest changes in endogenous ABA levels and ABA metabolism in relation to dormancy in potato tubers. Physiol Plant 95:233–240
Suttle JC (2004) Involvement of endogenous gibberellins in potato tuber dormancy and early sprout growth: a critical assessment. J Plant Physiol 161:157–164
Talburt WF, Schwimmer S, Burr HK (1975) Structure and chemical composition of the potato tuber. In: Talburt WF and Smith O (eds) Potato processing, 2nd edn. AVI publishing, Westport, Connecticut, pp 11–46.
Tompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The Clustal X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882
Valerio C, Costa A, Marri L, Issakidis-Bourguet E, Pupillo P, Trost P, Sparla F (2011) Thioredoxin-regulated beta-amylase (BAM1) triggers diurnal starch degradation in guard cells, and in mesophyll cells under osmotic stress. J Exp Bot 62:545–555
Zhang H, Liu X, Liu J, Ou Y, Lin Y, Li M, Song B, Xie C (2013) A novel RING finger gene, SbRFP1, increases resistance to cold-induced sweetening of potato tubers. FEBS Lett 587:749–755
Acknowledgments
We thank Prof. Dr. Uwe Sonnewald and Dr. Jingcai Li for useful discussion and Stephen Reid and Dr. Juan Du for language editing. This research was supported by grants from the National Science Foundation of China (31171602) and the National High Technology Research and Development Program of China (2009AA10Z103).
Author information
Authors and Affiliations
Corresponding author
Additional information
Huiling Zhang and Juan Hou contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhang, H., Hou, J., Liu, J. et al. Amylase Analysis in Potato Starch Degradation During Cold Storage and Sprouting. Potato Res. 57, 47–58 (2014). https://doi.org/10.1007/s11540-014-9252-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11540-014-9252-6