Abstract
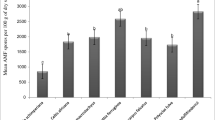
The plants in the Gurbantunggut Desert located in Xinjiang, China are drought adapted species and grow in generally nutrient-poor soils. A survey was conducted in the desert to determine the arbuscular mycorrhizal (AM) status associated with the desert plants which belong to 23 species from 11 families of annuals and perennials. Roots from all plants were examined for the presence of internal and external hyphae, vesicles and coils/arbuscules to determine the status and extent of mycorrhizal colonization. Of the plant species surveyed, 14 (61%) were found to form AM associations, 5 (22%) were possible AM colonized species, and 4 were non-mycorrhizal plants. The proportions of annuals and shrubs forming AM were significantly lower than those of perennials and herbs, respectively. Spore density varied from 5 to 21 per 20 g in soil of root zone. 14 AM fungal taxa in 3 genera were isolated and identified, 10 of which belonged to Glomus, 3 to Acaulospora, and 1 to Archaeospora. Glomus was the dominant genus in all genera identified. G. deserticola and G. etunicatum were the most common taxa isolated, with occurrence frequencies of 77.4% and 74.8%, and relative abundances of 14.4% and 15.5%, respectively.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Wang X Q, Lei J Q. The characteristics of erosion and deposition on the longitudinal dune surface in Gurbantunggut Desert. Arid Zone Res, 1998, 15(1): 35–39
Wang X Q, Jiang J, Lei J Q, et al. The distribution of ephemeral vegetation on the longitudinal dune surface and its stabilization significance in the Gurbantunggut Desert. Acta Geog Sin, 2003, 58: 598–605
Smith S E, Read D J. Mycorrhizal Symbiosis. London: Academic Press, 1997. 1–378
Call C A, McKell C M. Field establishment of four-wing saltbush (Atriplex canescens) in processed oil shale and disturbed native soil as influenced by vesicular-arbuscular mycorrhizae. Great Bas Natural, 1984, 442: 363–371
Cui M, Nobel P S. Nutrient status, water uptake and gas exchange for three desert succulents infected with mycorrhizal fungi. New Phytol, 1992, 122: 643–649
Davies J F T, Potter J R, Linderman R G. Drought resistance of mycorrhizal pepper plants independent of leaf P concentration—response in gas exchange and water relations. Physiol Plant, 1993, 87: 45–53
Davies J F T, Svenson S E, Cole J C, et al. Non-nutritional stress acclimation of mycorrhizal woody plants exposed to drought. Tree Physiol, 1996, 16: 985–993
Subramanian K S, Charest C, Dwyer L M, et al. Effects of arbuscular mycorrhizae on leaf water potential, sugar content, and P content during drought and recovery of maize. Can J Bot, 1997, 75: 1582–1591
Grime J P, Mackey J M L, Hillier S H, et al. Floristic diversity in a model system using experimental microcosms. Nature, 1987, 328: 420–422
Gange A C, Brown V K, Farmer L M. A test of mycorrhizal benefit in an early successional plant community. New Phytol, 1990, 115: 85–91
van der Heijden M G A, Klironomos J N, Ursic M, et al. Mycorrhizal fungal diversity determines plant biodiversity, ecosystem variability and productivity. Nature, 1998, 396: 69–72
Hartnett D C, Wilson G W T. Mycorrhizae influence plant community structure and diversity in tallgrass prairie. Ecology, 1999, 80: 1187–1195
Klironomos J N, McCune J, Hart M, et al. The influence of arbuscular mycorrhizae on the relationship between plant diversity and productivity. Ecol Lett, 2000, 3: 137–141
O’Connor P J, Smith S E, Smith F A. Arbuscular mycorrhizas influence plant diversity and community structure in semiarid herbland. New Phytol, 2002, 154: 209–218
Dhillion S S, Gardsjord T L. Arbuscular mycorrhizas influence plant diversity, productivity, and nutrients in boreal grasslands. Can J Bot, 2004, 82: 104–114
O’Connor P J, Smith S E, Smith F A. Arbuscular mycorrhizal associations in the Simpson Desert. Aus J Bot, 2001, 49: 493–499
Titus J H, Titus P J, Nowak R S, et al. Arbuscular mycorrhizae of Mojave Desert plants. Western North Am Natural, 2002, 62: 327–334
Collier S C, Yarnes C T, Herman R P. Mycorrhizal dependency of Chihuahuan Desert plants is influenced by life history strategy and root morphology. J Arid Environ, 2003, 55: 223–229
Ferrol N, Calvente R, Cano C, et al. Analysing arbuscular mycorrhizal fungal diversity in shrub-associated resource islands from a desertification threatened semiarid Mediterranean ecosystem. Appl Soil Ecol, 2004, 25: 123–133
Biemann B, Linderman R G. Quantifying vesicular arbuscular mycorrhizae: A proposed methods towards standardization. New Phytol, 1981, 87: 63–67
Koske R E, Tessier B. A convenient, permanent slide mounting medium. Newsl Mycol Soc Am, 1983, 34: 59
McGonigle T P, Miller M H, Evans D G, et al. A new method which gives an objective measure of colonization of roots by vesicular arbuscular mycorrhizal fungi. New Phytol, 1990, 115: 495–501
Dalpe Y. Vesicular-arbuscular mycorrhiza, collection. In: Carter M R, ed. Soil Sampling and Methods of Analysis. Boca Raton: Lewis Publishers, 1993, 287–301
Morton J B, Redecker D. Two new families of Glomales, Achaeosporaceae and Paraglomaceae, with two new genera Achaeospora and Paraglomus, based on concordant molecular and morphological characters. Mycologia, 2001, 93: 181–195
Gange A C, Brown V K, Sinclair G S. Vesicular-arbuscular mycorrhizal fungi: A determinant of plant community structure in early succession. Funct Ecol, 1993, 7: 616–622
Francis R, Read D J. The contributions of mycorrhizal fungi to the determination of plant community structure. Plant Soil, 1994, 159: 11–25
Azcón-Aguilar C, Barea J M. Applying mycorrhiza biotechnology to horticulture: Significance and potentials. Sci Hort, 1997, 68: 1–24
Hartnett D C, Wilson G W T. The role of mycorrhizas in plant community structure and dynamics: Lessons from grasslands. Plant Soil, 2002, 244: 319–331
Moraes R M, De Andrade Z, Bedir E, et al. Arbuscular mycorrhiza improves acclimatization and increases lignan content of micropropagated mayapple (Podophyllum peltatum L.). Plant Sci, 2004, 166: 23–29
Allen M F. Formation of vesicular-arbuscular mycorrhizae in Atriplex gardneri (Chenopodiaceae): Seasonal response in a cold desert. Mycologia, 1983, 75: 773–776
Allen M F, Allen E B, Friese C F. Responses of the nonmycotropic plant Salsola kali to invasion by vesicular-arbuscular mycorrhizal fungi. New Phytol, 1989, 111: 45–49
Aguilera L E, Gutierrez J R, Moreno R J. Vesiculo arbuscular mycorrhizae associated with saltbushes Atriplex spp. (Chenopodiaceae) in the Chilean arid zone. Rev Chil Hist Nat, 1998, 71: 291–302
Siguenza C, Espejel I, Allen E B. Seasonality of mycorrhizae in coastal sand dunes of Baja California. Mycorrhiza, 1996, 6: 151–157
He X L, Mouratov S, Steinberger Y. Temporal and spatial dynamics of vesicular-arbuscular mycorrhizal fungi under the canopy of Zygophyllum dumosum Boiss. in the Negev Desert. J Arid Environ, 2002, 52: 379–387
Miller R M. Some occurrences of vesicular-arbuscular mycorrhiza in natural and disturbed ecosystems of the Red Desert. Can J Bot, 1979, 57: 619–623
Reeves F B, Wagner D, Moorman T, et al. The role of endomycorrhizae in revegetation practices in the semi-arid West (I): Comparison of incidence of mycorrhizae in severely disturbed vs natural environments. Am J Bot, 1979, 66: 6–13
Bethlenfalway G J, Dakessian S, Pacovsky R S. Mycorrhizae in a southern California desert: Ecological implications. Can J Bot, 1984, 62: 519–524
Dhillion S S, Vidiella P E, Aquilera L E, et al. Mycorrhizal plants and fungi in the fog-free Pacific coastal desert of Chile. Mycorrhiza, 1995, 5: 381–386
Rose S L. Vesicular-arbuscular endomycorrhizal associations of some desert plants of Baja California. Can J Bot, 1981, 59: 1056–1060
Pond E C, Menge J A, Jarrell W M. Improved growth of tomato in salinized soil by vesicular-arbuscular mycorrhizaal fungi collected from saline soils. Mycologia, 1984, 76: 74–84
Stahl P D, Christensen M. Mycorrhizal fungi associated with Agropyron and Bouteloua in Wyoming sagebrush-grasslands. Mycologia, 1982, 74: 877–885
Stutz J, Morton J B. Successive pot cultures reveal high species richness of indigenous arbuscular endomycorrhizal fungi in arid ecosystems. Can J Bot, 1996, 74: 1883–1889
Zhao Z W, Xia Y M, Qin X Z, et al. Arbuscular mycorrhizal status of plants and the spore density of arbuscular mycorrhizal fungi in the tropical rain forest of Xishuangbanna, southwest China. Mycorrhiza, 2001, 11: 159–162
Shi Z Y, Chen Y L, Feng G, et al. Arbuscular mycorrhizal fungi associated with the Meliaceae on Hainan island, China. Mycorrhiza, 2006, 16(2): 81–87
Uhlmann E, Görke C, Petersen A, et al. Arbuscular mycorrhizae from semiarid region of Namibia. Can J Bot, 2004, 82: 645–653
Lamont B. Mechanisms for enhancing nutrient uptake in plants, with particular reference to Mediterranean South Africa and Western Australia. Bot Rev, 1982, 48: 597–689
Pande M, Tarafdar J C. Arbuscular mycorrhizal fungal diversity in neem-based agroforestry systems in Rajasthan. Appl Soil Ecol, 2004, 26: 233–241
Koske R E, Halvorson W L. Ecological studies of vesiculararbuscular mycorrhizae in a barrier sand dune. Can J Bot, 1981, 59: 1413–1422
Logan V S, Clark P J, Allaway W G. Mycorrhizas and root attributes of plants of coastal sand-dunes of New South Wales. Aus J Plant Physiol, 1989, 16: 141–146
Jehne W, Thompson C H. Endomycorrhizae in plant colonization on coastal sand-dunes at Cooloola, Queensland. Aus J Ecol, 1981, 6: 221–230
Sylvia D M. Spatial and temporal distribution of vesiculararbuscular mycorrhizal fungi associated with Uniola paniculata in Florida foredunes. Mycologia, 1986, 78: 728–734
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Tian, C., Shi, Z., Chen, Z. et al. Arbuscular mycorrhizal associations in the Gurbantunggut Desert. CHINESE SCI BULL 51 (Suppl 1), 140–146 (2006). https://doi.org/10.1007/s11434-006-8218-8
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11434-006-8218-8